Abstract
Introduction: Miuraenamides belong to natural marine compounds with interesting biological properties.
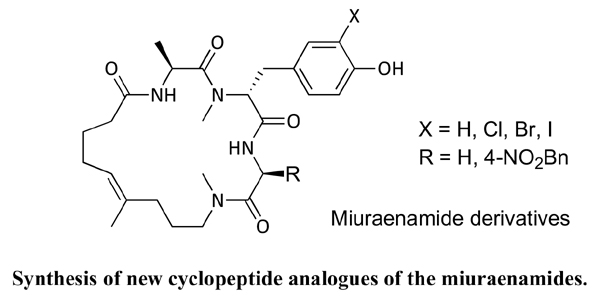
Materials and Methods: Miuraenamides initiate polymerization of monomeric actin and therefore show high cytotoxicity by influencing the cytoskeleton. New derivatives of the miuraenamides have been synthesized containing an N-methylated amide bond instead of the more easily hydrolysable ester in the natural products.
Results: Incorporation of an aromatic side chain onto the C-terminal amino acid of the tripeptide fragment also led to highly active new miuraenamides.
Conclusion: In this study, we showed that the ester bond of the natural product miuraenamide can be replaced by an N-methyl amide. The yields in the cyclization step were high and generally much better than with the corresponding esters. On the other hand, the biological activity of the new amide analogs was lower compared to the natural products, but the activity could significantly be increased by incorporation of a p-nitrophenyl group at the C-terminus of the peptide fragment.
Keywords: Actin, cyclodepsipeptides, cyclopeptides, cytoskeleton, cytotoxic compounds, myxobacteria, natural products.
(b) Hamada, Y.; Shioiri, T. Recent progress of the synthetic studies of biologically active marine cyclic peptides and depsipeptides. Chem. Rev., 2005, 105(12), 4441-4482.
[http://dx.doi.org/10.1021/cr0406312] [PMID: 16351050]
(c) von Nussbaum, F.; Brands, M.; Hinzen, B.; Weigand, S.; Häbich, D. Antibacterial natural products in medicinal chemistry-Exodus or revival? Angew. Chem. Int. Ed. Engl., 2006, 45(31), 5072-5129.
[http://dx.doi.org/10.1002/anie.200600350] [PMID: 16881035]
(d) Desriac, F.; Jégou, C.; Balnois, E.; Brillet, B.; Le Chevalier, P.; Fleury, Y. Antimicrobial peptides from marine proteobacteria. Mar. Drugs, 2013, 11(10), 3632-3660.
[http://dx.doi.org/10.3390/md11103632] [PMID: 24084784]
[http://dx.doi.org/10.1016/S0040-4039(00)84645-6]
(b) Braekman, J.C.; Daloze, D.; Moussiaux, B.; Riccio, R. Jaspamide from the marine sponge Jaspis johnstoni. J. Nat. Prod., 1987, 50, 994-995.
[http://dx.doi.org/10.1021/np50053a048]
[http://dx.doi.org/10.1021/jo00390a023]
(b) De Silva, E.D.; Andersen, R.J.; Allen, T.M. Geodiamolides C to F, new cytotoxic cyclodepsipeptides from the marine sponge Pseudaxinyssa sp. Tetrahedron Lett., 1990, 31, 489-492.
[http://dx.doi.org/10.1016/0040-4039(90)87015-R]
[http://dx.doi.org/10.1016/S0040-4039(00)80273-7]
(b) Kato, S.; Hamada, Y.; Shioiri, T. A practical synthesis of the peptide part of jaspamide (jasplakinolide), a cyclodepsipeptide from a marine sponge. Tetrahedron Lett., 1988, 29, 6465-6466.
(c) Grieco, P.A.; Perez-Medrano, A. Total synthesis of the mixed peptide-polypropionate based cyclodepsipeptide (+)-geodiamolide B. Tetrahedron Lett., 1988, 29, 4225-4228.
(d) White, J.D.; Amedio, J.C., Jr Total synthesis of geodiamolide A, a novel cyclodepsipeptide of marine origin. J. Org. Chem., 1989, 54, 736-738.
[http://dx.doi.org/10.1007/BF00689969] [PMID: 1505079]
[PMID: 8195116]
(b) Senderowicz, A.M.J.; Kaur, G.; Sainz, E.; Laing, C.; Inman, W.D.; Rodríguez, J.; Crews, P.; Malspeis, L.; Grever, M.R.; Sausville, E.A.; Duncan, K.L. Jasplakinolide’s inhibition of the growth of prostate carcinoma cells in vitro with disruption of the actin cytoskeleton. J. Natl. Cancer Inst., 1995, 87(1), 46-51.
[http://dx.doi.org/10.1093/jnci/87.1.46] [PMID: 7666463]
[http://dx.doi.org/10.1002/jcp.21432] [PMID: 18330887]
[http://dx.doi.org/10.1016/S0040-4020(98)00157-4]
[http://dx.doi.org/10.1038/ja.2006.55] [PMID: 17025014]
[http://dx.doi.org/10.1016/S0076-6879(09)04803-4] [PMID: 19374979]
(b) Weissman, K.J.; Müller, R. Myxobacterial secondary metabolites: bioactivities and modes-of-action. Nat. Prod. Rep., 2010, 27(9), 1276-1295.
[http://dx.doi.org/10.1039/c001260m] [PMID: 20520915]
[http://dx.doi.org/10.1039/b612668p] [PMID: 18033578]
(b) Wenzel, S.C.; Müller, R. Myxobacteria--‘microbial factories’ for the production of bioactive secondary metabolites. Mol. Biosyst., 2009, 5(6), 567-574.
[http://dx.doi.org/10.1039/b901287g] [PMID: 19462013]
(c) Wenzel, S.C.; Müller, R. The biosynthetic potential of myxobacteria and their impact in drug discovery. Curr. Opin. Drug Discov. Devel., 2009, 12(2), 220-230.
[PMID: 19333867]
(d) Birkett, S.L.; Loits, D.A.; Wimala, S.; Rizzacasa, M.A. Synthesis of myxobacteria metabolites. Pure Appl. Chem., 2012, 84, 1421-1433.
[http://dx.doi.org/10.1351/PAC-CON-11-11-19]
[http://dx.doi.org/10.3390/md8092466] [PMID: 20948900]
(b) García-Ruiz, C.; Sarabia, F. Chemistry and biology of bengamides and bengazoles, bioactive natural products from Jaspis sponges. Mar. Drugs, 2014, 12(3), 1580-1622.
[http://dx.doi.org/10.3390/md12031580] [PMID: 24646945]
(c) Dávila-Céspedes, A.; Hufendiek, P.; Crüsemann, M.; Schäberle, T.F.; König, G.M. Marine-derived myxobacteria of the suborder Nannocystineae: An underexplored source of structurally intriguing and biologically active metabolites. Beilstein J. Org. Chem., 2016, 12, 969-984.
[http://dx.doi.org/10.3762/bjoc.12.96] [PMID: 27340488]
[http://dx.doi.org/10.1038/ja.2006.55] [PMID: 17025014]
[http://dx.doi.org/10.1002/asia.200700233] [PMID: 18022981]
[http://dx.doi.org/10.1021/cb1003459] [PMID: 21322638]
(b) Wang, S.; Crevenna, A.H.; Ugur, I.; Marion, A.; Antes, I.; Kazmaier, U.; Hoyer, M.; Lamb, D.C.; Gegenfurtner, F.; Kliesmete, Z.; Ziegenhain, C.; Enard, W.; Vollmar, A.; Zahler, S. Actin stabilizing compounds show specific biological effects due to their binding mode. Sci. Rep., 2019, 9(1), 9731.
[http://dx.doi.org/10.1038/s41598-019-46282-w] [PMID: 31278311]
[http://dx.doi.org/10.1002/anie.201808028]
[http://dx.doi.org/10.1002/anie.200900406]
(b) Ullrich, A.; Herrmann, J.; Müller, R.; Kazmaier, U. Synthesis and biological evaluation of pretubulysin and derivatives. Eur. J. Org. Chem., 2009, 6367-6378.
[http://dx.doi.org/10.1002/ejoc.200900999]
(c) Chai, Y.; Pistorius, D.; Ullrich, A. Weissman, K. J.; Kazmaier, U.; Müller, R. Discovery of 23 novel natural tubulysins from Angiococcus disciformis An d48 and Cystobacter SBCb004. Chem. Biol., 2010, 17, 296-309.
[http://dx.doi.org/10.1016/j.chembiol.2010.01.016] [PMID: 20338521]
(d) Kazmaier, U.; Ullrich, A.; Hoffmann, J. Synthetic approaches towards tubulysins and their derivatives Open. Nat. Prod. J., 2013, 6, 12-30.
[http://dx.doi.org/10.2174/1874848101306010012]
[http://dx.doi.org/10.1371/journal.pone.0037416]
(b) Kubisch, R.; von Gamm, M.; Braig, S.; Ullrich, A.; Burkhart, J.L.; Colling, L.; Hermann, J.; Scherer, O.; Müller, R.; Werz, O.; Kazmaier, U.; Vollmar, A.M. Simplified pretubulysin derivatives and their biological effects on cancer cells. J. Nat. Prod., 2014, 77(3), 536-542.
[http://dx.doi.org/10.1021/np4008014] [PMID: 24437936]
[http://dx.doi.org/10.1039/c2mb25144b] [PMID: 22722320]
[http://dx.doi.org/10.1002/cbic.201300140] [PMID: 23959765]
(b) Becker, D.; Kazmaier, U. Synthesis of simplified halogenated chondramide derivatives as new actin-binding agents. Eur. J. Org. Chem., 2015, 2591-2602.
[http://dx.doi.org/10.1002/ejoc.201403577]
(c) Becker, D.; Kazmaier, U. Synthesis and biological evaluation of dichlorinated chondramide derivatives. Eur. J. Org. Chem., 2015, 4198-4213.
[http://dx.doi.org/10.1002/ejoc.201500369]
[http://dx.doi.org/10.1002/anie.200801010] [PMID: 18624308]
(b) Ma, C.I.; Diraviyam, K.; Maier, M.E.; Sept, D.; Sibley, L.D. Synthetic chondramide A analogues stabilize filamentous actin and block invasion by Toxoplasma gondii. J. Nat. Prod., 2013, 76(9), 1565-1572.
[http://dx.doi.org/10.1021/np400196w] [PMID: 24020843]
(c) Menhofer, M.H.; Kubisch, R.; Schreiner, L.; Zorn, M.; Foerster, F.; Mueller, R.; Raedler, J.O.; Wagner, E.; Vollmar, A.M.; Zahler, S. The actin targeting compound Chondramide inhibits breast cancer metastasis via reduction of cellular contractility. PLoS One, 2014, 9(11)e112542
[http://dx.doi.org/10.1371/journal.pone.0112542] [PMID: 25391145]
[http://dx.doi.org/10.1002/anie.201411212]
[http://dx.doi.org/10.1021/acs.joc.6b02061]
[http://dx.doi.org/10.1021/jo00238a005]