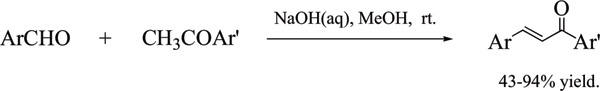Abstract
Background: Although the expression levels of many P450s differ between tumour and corresponding normal tissue, CYP1B1 is one of the few CYP subfamilies which is significantly and consistently overexpressed in tumours. CYP1B1 has been shown to be active within tumours and is capable of metabolising a structurally diverse range of anticancer drugs. Because of this, and its role in the activation of procarcinogens, CYP1B1 is seen as an important target for anticancer drug development.
Objective: To synthesise a series of chalcone derivatives based on the chemopreventative agent DMU-135 and investigate their antiproliferative activities in human breast cancer cell lines which express CYP1B1 and CYP1A1.
Method: A series of chalcones were synthesised in yields of 43-94% using the Claisen-Schmidt condensation reaction. These were screened using a MTT assay against a panel of breast cancer cell lines which have been characterised for CYP1 expression.
Result: A number of derivatives showed promising antiproliferative activities in human breast cancer cell lines which express CYP1B1 and CYP1A1, while showing significantly lower toxicity towards a non-tumour breast cell line with no CYP expression. Experiments using the CYP1 inhibitors acacetin and α-naphthoflavone provided supporting evidence for the involvement of CYP1 enzymes in the bioactivation of these compounds.
Conclusion: Chalcones show promise as anticancer agents with evidence suggesting that CYP1 activation of these compounds may be involved.
Keywords: Antiproliferative, cancer, chalcones, CYP1A1, CYP1B1, prodrug.