Abstract
Background: Cyclin-dependent kinase 4 (CDK4) and the human epidermal growth factor receptor 2 (HER2) are two of the most promising targets in oncology research. Thus, a series of computational approaches have been applied to the search for more potent inhibitors of these cancerrelated proteins. However, current approaches have focused on chemical analogs while predicting the inhibitory activity against only one of these targets, but never against both.
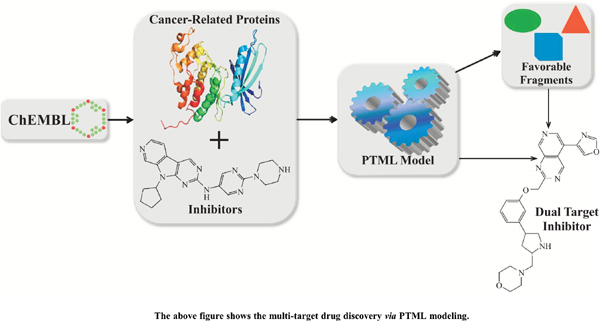
Aims: We report the first perturbation model combined with machine learning (PTML) to enable the design and prediction of dual inhibitors of CDK4 and HER2.
Methods: Inhibition data for CDK4 and HER2 were extracted from ChEMBL. The PTML model relied on artificial neural networks to allow the classification/prediction of molecules as active or inactive against CDK4 and/or HER2.
Results: The PTML model displayed sensitivity and specificity higher than 80% in the training set. The same statistical metrics had values above 75% in the test set. We extracted several molecular fragments and estimated their quantitative contributions to the inhibitory activity against CDK4 and HER2. Guided by the physicochemical and structural interpretations of the molecular descriptors in the PTML model, we designed six molecules by assembling several fragments with positive contributions. Three of these molecules were predicted as potent dual inhibitors of CDK4 and HER2, while the other three were predicted as inhibitors of at least one of these proteins. All the molecules complied with Lipinski’s rule of five and its variants.
Conclusion: The present work represents an encouraging alternative for future anticancer chemotherapies.
Keywords: Artificial neural network, Cancer, Fragment, Pseudo-linear equation, PTML model, Local contributions, Virtual design.
[http://dx.doi.org/10.3322/caac.21442] [PMID: 29313949]
[http://dx.doi.org/10.1038/d41586-018-07188-1] [PMID: 30459368]
[http://dx.doi.org/10.3389/fphar.2018.00245] [PMID: 29623040]
[http://dx.doi.org/10.1371/journal.pone.0184360] [PMID: 29016607]
[http://dx.doi.org/10.1111/ecc.12618] [PMID: 28004440]
[http://dx.doi.org/10.1016/j.tcb.2018.07.002] [PMID: 30061045]
[http://dx.doi.org/10.1016/j.ctrv.2014.02.008] [PMID: 24656976]
[http://dx.doi.org/10.1016/j.jtho.2015.10.025] [PMID: 26723242]
[http://dx.doi.org/10.1016/j.lungcan.2014.12.018] [PMID: 25601485]
[http://dx.doi.org/10.1016/S0140-6736(16)32417-5] [PMID: 27939064]
[http://dx.doi.org/10.1517/14656566.2014.870555] [PMID: 24369047]
[http://dx.doi.org/10.1093/annonc/mdy155] [PMID: 29718092]
[http://dx.doi.org/10.1093/annonc/mdx002] [PMID: 28119295]
[http://dx.doi.org/10.1016/j.jmgm.2018.04.001] [PMID: 29680736]
[http://dx.doi.org/10.1016/j.compbiolchem.2017.11.005] [PMID: 29153893]
[http://dx.doi.org/10.1371/journal.pone.0093704] [PMID: 24722522]
[http://dx.doi.org/10.1038/aps.2013.105] [PMID: 24122012]
[http://dx.doi.org/10.1016/j.compbiolchem.2018.04.002] [PMID: 29702367]
[http://dx.doi.org/10.1080/1062936X.2017.1284898] [PMID: 28235391]
[http://dx.doi.org/10.1016/j.jmgm.2015.06.008] [PMID: 26188796]
[http://dx.doi.org/10.4155/fmc.14.136] [PMID: 25531966]
[http://dx.doi.org/10.1007/s00044-017-1936-4]
[http://dx.doi.org/10.1021/acs.jcim.5b00630] [PMID: 26960000]
[http://dx.doi.org/10.2174/1568026615666150506144814] [PMID: 25961517]
[http://dx.doi.org/10.1016/j.biosystems.2015.04.007] [PMID: 25916548]
[http://dx.doi.org/10.1021/ci400716y] [PMID: 24521170]
[http://dx.doi.org/10.1021/acs.molpharmaceut.9b00538] [PMID: 31426639]
[http://dx.doi.org/10.1021/acs.jcim.9b00034] [PMID: 30802402]
[http://dx.doi.org/10.1016/j.chemolab.2014.08.006]
[http://dx.doi.org/10.1080/17435390.2017.1379567] [PMID: 28937298]
[http://dx.doi.org/10.2217/nnm.14.96] [PMID: 25600965]
[http://dx.doi.org/10.1021/acs.chemrestox.8b00266] [PMID: 30868869]
[http://dx.doi.org/10.1021/acs.jcim.8b00631] [PMID: 30408958]
[http://dx.doi.org/10.1038/s41598-017-13691-8] [PMID: 29038520]
[http://dx.doi.org/10.1021/acs.jcim.6b00458] [PMID: 28414908]
[http://dx.doi.org/10.1021/acs.jproteome.7b00477] [PMID: 28922600]
[http://dx.doi.org/10.1016/j.ejmech.2013.08.035] [PMID: 24445280]
[http://dx.doi.org/10.1021/acs.chemrestox.9b00154] [PMID: 31327231]
[http://dx.doi.org/10.2174/15680266113139990114] [PMID: 23889053]
[http://dx.doi.org/10.1021/acschemneuro.8b00083] [PMID: 29791132]
[http://dx.doi.org/10.2174/1389450116666151102095243] [PMID: 26521774]
[http://dx.doi.org/10.1016/j.neuropharm.2015.12.019] [PMID: 26721628]
[http://dx.doi.org/10.1016/j.bmc.2013.01.035] [PMID: 23415089]
[http://dx.doi.org/10.1021/acschemneuro.9b00302] [PMID: 31618004]
[http://dx.doi.org/10.1021/cn400111n] [PMID: 23855599]
[http://dx.doi.org/10.1016/j.ejmech.2011.02.072] [PMID: 21447431]
[http://dx.doi.org/10.1021/acs.molpharmaceut.0c00308] [PMID: 32459098]
[http://dx.doi.org/10.1038/s41598-020-62279-2] [PMID: 32210335]
[http://dx.doi.org/10.1021/acscombsci.8b00090] [PMID: 30240186]
[http://dx.doi.org/10.2174/1871520611313050013] [PMID: 23272967]
[http://dx.doi.org/10.1007/s11030-017-9731-1] [PMID: 28194627]
[http://dx.doi.org/10.1093/nar/gkr777] [PMID: 21948594]
[http://dx.doi.org/10.1021/ci200260t] [PMID: 21978256]
[http://dx.doi.org/10.1016/j.chembiol.2003.09.002] [PMID: 14522049]
[http://dx.doi.org/10.1002/9783527628766]
[http://dx.doi.org/10.1021/jp026238m]
[http://dx.doi.org/10.1002/9783527613106]
[http://dx.doi.org/10.2174/15680266113139990130] [PMID: 23895092]
[http://dx.doi.org/10.2174/1573406411309020009] [PMID: 22762164]
[http://dx.doi.org/10.1007/s10822-015-9893-9] [PMID: 26721261]
[http://dx.doi.org/10.1007/s10822-016-9895-2] [PMID: 26830599]
[http://dx.doi.org/10.3390/ijms150917035] [PMID: 25255029]
[http://dx.doi.org/10.1016/0005-2795(75)90109-9] [PMID: 1180967]
[http://dx.doi.org/10.1098/rspl.1895.0041]
[http://dx.doi.org/10.3390/molecules17054791] [PMID: 22534664]
[http://dx.doi.org/10.1080/1062936X.2020.1818617] [PMID: 32967475]
[http://dx.doi.org/10.1021/ci00025a021]
[http://dx.doi.org/10.1021/acscombsci.7b00039] [PMID: 28437091]
[http://dx.doi.org/10.1021/acsomega.8b02419] [PMID: 30555986]
[http://dx.doi.org/10.2174/1568026620666200607190951] [PMID: 32515311]
[http://dx.doi.org/10.1007/s11030-018-9890-8] [PMID: 30421269]
[http://dx.doi.org/10.1021/ci049714+] [PMID: 15667143]
[http://dx.doi.org/10.1016/S0169-409X(00)00129-0] [PMID: 11259830]
[http://dx.doi.org/10.1021/jm020017n] [PMID: 12036371]