Abstract
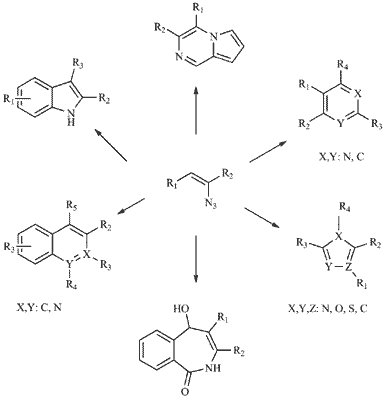
Vinyl azides have emerged as important synthons in recent years and a variety of |useful synthetic methodologies have been achieved in the construction of the nitrogen heterocycles, such as indoles, pyrroles, imidazoles, pyrazoles, pyridines and pyrimidines. The unstable azide group and the presence of neighboring unsaturated double bond accentuate the high reactive ity of vinyl azides with other electrophiles and nucleophiles in various manners. One of the important features of vinyl azides is that they can decompose to vinyl nitrenes or highly strained three-membered cyclic imines, 2H-azirines. These two three-atom intermediates can easily undergo tandem reactions with 1,3-dipoles to generate the nitrogen-containing heterocycles. Vinyl azide can also act as a two-atom synthon while azide group serves as a leaving group in the cycloaddition reactions. What’s more, when vinyl azide is connected with a carbonyl group in the α-position of azide, the resulted α-azidovinylketone can react with the dinucleophiles, leading to the amino substituted ring. The purpose of this review is to present the synthetic approaches achieved both in our group and other groups.
Keywords: 2H-azirine, nitrene, nitrogen heterocycles, synthon, vinyl azides, α-azidovinylketone.
Current Organic Chemistry
Title:Vinyl Azides as Versatile Synthons for the Synthesis of Nitrogen-containing Heterocycles
Volume: 19 Issue: 9
Author(s): Hangcheng Ni, Guolin Zhang and Yongping Yu
Affiliation:
Keywords: 2H-azirine, nitrene, nitrogen heterocycles, synthon, vinyl azides, α-azidovinylketone.
Abstract: Vinyl azides have emerged as important synthons in recent years and a variety of |useful synthetic methodologies have been achieved in the construction of the nitrogen heterocycles, such as indoles, pyrroles, imidazoles, pyrazoles, pyridines and pyrimidines. The unstable azide group and the presence of neighboring unsaturated double bond accentuate the high reactive ity of vinyl azides with other electrophiles and nucleophiles in various manners. One of the important features of vinyl azides is that they can decompose to vinyl nitrenes or highly strained three-membered cyclic imines, 2H-azirines. These two three-atom intermediates can easily undergo tandem reactions with 1,3-dipoles to generate the nitrogen-containing heterocycles. Vinyl azide can also act as a two-atom synthon while azide group serves as a leaving group in the cycloaddition reactions. What’s more, when vinyl azide is connected with a carbonyl group in the α-position of azide, the resulted α-azidovinylketone can react with the dinucleophiles, leading to the amino substituted ring. The purpose of this review is to present the synthetic approaches achieved both in our group and other groups.
Export Options
About this article
Cite this article as:
Ni Hangcheng, Zhang Guolin and Yu Yongping, Vinyl Azides as Versatile Synthons for the Synthesis of Nitrogen-containing Heterocycles, Current Organic Chemistry 2015; 19 (9) . https://dx.doi.org/10.2174/1385272819666150320001329
| DOI https://dx.doi.org/10.2174/1385272819666150320001329 |
Print ISSN 1385-2728 |
| Publisher Name Bentham Science Publisher |
Online ISSN 1875-5348 |
Call for Papers in Thematic Issues
Advances of Heterocyclic Chemistry with Pesticide Activity
Global food safety and security will continue to be a global concern for the next 50 years and beyond. Plant diseases have had a significant impact on food safety and security throughout the entire food chain, from primary production to consumption. While conventional chemical pesticides have been traditionally used for ...read more
Calculation design of covalent/metal organic framework based catalysts
This research area combines theoretical computation and screening with machine learning for the design of covalent/metal organic framework-based catalysts, bridging the disciplines of organic chemistry, physical chemistry, computational chemistry, materials science, and machine learning. It covers several critical aspects: designing and synthesizing organic catalysts for improved performance, applying computational methods ...read more
Carbohydrates conversion in biofuels and bioproducts
Biomass pretreatment, hydrolysis, and saccharification of carbohydrates, and sugars bioconversion in biofuels and bioproducts within a biorefinery framework. Carbohydrates derived from woody biomass, agricultural wastes, algae, sewage sludge, or any other lignocellulosic feedstock are included in this issue. Simulation, techno-economic analysis, and life cycle analysis of a biorefinery process are ...read more
Catalytic C-H bond activation as a tool for functionalization of heterocycles
The major topic is the functionalization of heterocycles through catalyzed C-H bond activation. The strategies based on C-H activation not only provide straightforward formation of C-C or C-X bonds but, more importantly, allow for the avoidance of pre-functionalization of one or two of the cross-coupling partners. The beneficial impact of ...read more
Related Journals
 65
65 2
2
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
- Announcements
Related Articles
-
Drug Repurposing for the Treatment of Staphylococcal Infections
Current Pharmaceutical Design Recent Advances in Pretargeted Radioimmunotherapy
Current Medicinal Chemistry Sphingosine Kinases Signalling in Carcinogenesis
Mini-Reviews in Medicinal Chemistry Dual Inhibition of PI3-Kinase and mTOR in Renal Cell Carcinoma
Current Cancer Drug Targets Sleep and the Immune System
Current Immunology Reviews (Discontinued) Application of RNA Interference for the Control of Female Reproductive Functions
Current Pharmaceutical Design Synthesis of [DTPA-bis(D-ser)] Chelate (DBDSC): An Approach for the Design of SPECT Radiopharmaceuticals Based on Technetium
Current Radiopharmaceuticals Insights into the Structural Features Essential for JAK2 Inhibition and Selectivity
Current Medicinal Chemistry Predictive Efficacy Biomarkers of Programmed Cell Death 1/Programmed Cell Death 1 Ligand Blockade Therapy
Recent Patents on Anti-Cancer Drug Discovery Targeting Protein Degradation in the Nervous System
Current Medicinal Chemistry - Central Nervous System Agents Busulphan in Blood and Marrow Transplantation: Dose, Route, Frequency and Role of Therapeutic Drug Monitoring
Current Clinical Pharmacology Midkine: A Promising Molecule for Drug Development to Treat Diseases of the Central Nervous System
Current Pharmaceutical Design The Pathogenic Subpopulation of Th17 Cells in Obesity
Current Pharmaceutical Design Recent Developments in Phototherapy: Treatment Methods and Devices
Recent Patents on Inflammation & Allergy Drug Discovery The Mechanism by which the Phenothiazine Thioridazine Contributes to Cure Problematic Drug-Resistant Forms of Pulmonary Tuberculosis: Recent Patents for “New Use”
Recent Patents on Anti-Infective Drug Discovery CD26/Dipeptidyl Peptidase IV as a Novel Therapeutic Target for Cancer and Immune Disorders
Mini-Reviews in Medicinal Chemistry In Silico Identification of Human miR 3654 and its Targets Revealed its Involvement in Prostate Cancer Progression
MicroRNA Targeting the Fibroblast Growth Factor Receptors for the Treatment of Cancer
Anti-Cancer Agents in Medicinal Chemistry Graphene and Graphene Oxide as a Docking Station for Modern Drug Delivery System
Current Drug Delivery Strategies for In Vivo siRNA Delivery in Cancer
Mini-Reviews in Medicinal Chemistry