Abstract
Background: Invasive tumor growth and metastasis require the formation of a chaotic network of blood vessels that nourish cancer cells and provide them with a direct access to peripheral circulation, which is essential for them to diffuse throughout the organism. Tumor vascularization is sustained by multiple mechanisms, including sprouting angiogenesis and vasculogenesis, which consists in the recruitment of endothelial progenitor cells (EPCs) from either the bone marrow or the arterial wall. Tumor endothelium presents significant morphologic, genetic and functional differences as compared to normal cells.
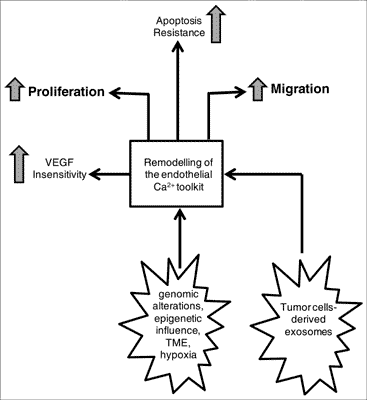
Methods: Recent work has shown that the Ca2+ signaling machinery is heavily remodeled in both tumor endothelial cells (TECs) and tumor-associated EPCs (T-EPCs). The rearrangement of the Ca2+ toolkit in TECs and T-EPCs leads to a dramatic improvement of their pro-angiogenic activity and increases cancer resistance to chemotherapeutics and anti-angiogenic treatments. Herein, the major changes involved in this process are analyzed. Results: The major changes observed include the reduction in the endogenous Ca2+ reservoir, the upregulation of store-operated Ca2+ entry or TRPC5 and the alteration in TRPV4 levels. Additionally, a growing number of studies revealed that other transporters involved in Ca2+ homeostasis are aberrantly expressed in TECs, including Na+/H+ exchanger, chloride intracellular channels, and nicotinic acetylcholine receptors. This emerging field of investigation is, therefore, coming of age because of its tremendous therapeutic potential. Conclusions: Understanding the impact of Ca2+ dysregulation in tumor vascularization is mandatory to circumvent patient refractoriness to current anti-cancer regimens.Keywords: Tumor vascularization, tumor endothelial cells, endothelial progenitor cells, Ca2+ signaling, proliferation, migration, vessel normalization, resistance to apoptosis.