Abstract
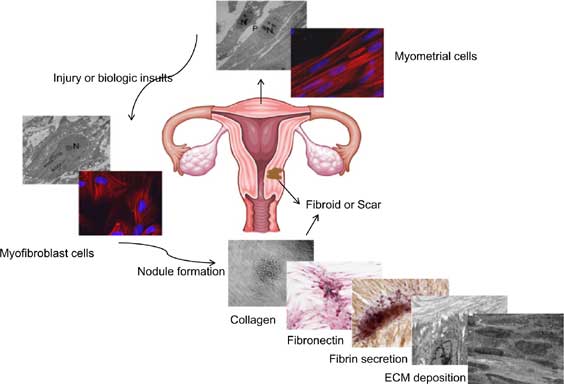
Utilizing both primary myometrial cells and a myometrial cell line, we show here that myometrial cells undergo transition to a myofibroblast-like phenotype after a biological insult of 72 hours serum starvation and serum add-back (SB: 1% to 10% FBS). We also found that thrombospondin-1 was increased and that the transforming growth factor-beta (TGFB)-SMAD3/4 pathway was activated. This pathway is a key mediator of fibrosis and extracellular matrix (ECM) deposition. Applying the same insult supplemented with TGFB3 (1-10ng/ml) and ascorbic acid (100μg/ml) in the serum add-back treatment, we further demonstrated that cells migrated into nodules containing collagen and fibronectin. The number of cellnodules was inversely related to the percentage serum add-back. Using transmission electron microscopy we demonstrated myofibroblast-like cells and fibril-like structures in the extracellular spaces of the nodules. This study is the first direct evidence of induction of myofibroblast transdifferentiation in cultured myometrial cells which is related to the increase of thrombospondin-1 (THBS1) and the activation of TGFBSMAD 3 / 4 pathways. Combined, these observations provide biochemical and direct morphological evidence that fibrotic responses can occur in cultured myometrial cells. The findings are the first to demonstrate uterine healing mechanisms at a molecular level. Our data support the concept that fibrosis may be an initial event in formation of fibroid which exhibits signaling pathways and molecular features of fibrosis and grow by both cellular proliferation and altered extracellular matrix accumulation. Our data assists in further understanding of myometrium tissue remodeling during gestation and postpartum.
Keywords: Uterine fibroids, myometrial cell injury and repair, fibrosis, myofibroblasts, nodules, TGFB3.