Abstract
Background: To address multidrug resistance, we developed engineered Cationic Antimicrobial Peptides (eCAPs). Lead eCAP WLBU2 displays potent activity against drug-resistant bacteria and effectively treats lethal bacterial infections in mice, reducing bacterial loads to undetectable levels in diverse organs.
Objective: To support the development of WLBU2, we conducted a mass balance study.
Methods: CD1 mice were administered 10, 15, 20 and 30 mg/kg of QDx5 WLBU2 or a single dose of [14C]-WLBU2 at 15 mg/kg IV. Tolerability, tissue distribution and excretion were evaluated with liquid scintillation and HPLC-radiochromatography.
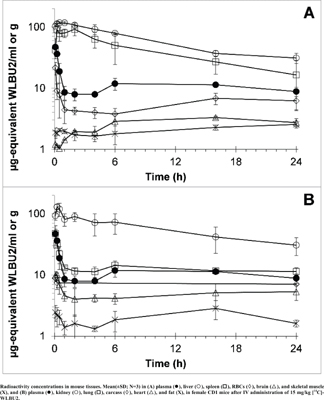
Results: The maximum tolerated dose of WLBU2 is 20 mg/kg IV. We could account for greater than >96% of the radioactivity distributed within mouse tissues at 5 and 15 min. By 24h, only ~40-50% of radioactivity remained in the mice. The greatest % of the dose was present in liver, accounting for ~35% of radioactivity at 5 and 15 min, and ~ 8% of radioactivity remained at 24h. High radioactivity was also present in kidneys, plasma, red blood cells and lungs, while less than 0.2% of radioactivity was present in brain, fat, or skeletal muscle. Urinary and fecal excretion accounted for 12.5 and 2.2% of radioactivity at 24h.
Conclusion: WLBU2 distributes widely to mouse tissues and is rapidly cleared with a terminal radioactivity half-life of 22 h, a clearance of 27.4 mL/h/kg, and a distribution volume of 0.94 L/kg. At 2-100 μg-eq/g, the concentrations of 14C-WLBU2 appear high enough in the tissues to account for the inhibition of microbial growth.
Keywords: Engineered cationic peptide, radioactivity, pharmacokinetics, mass balance, antimicrobial, disposition.