Abstract
Background: Nowadays, in course of the drug design and discovery much attention is paid to the physicochemical parameters of a drug candidate, in addition to their biological activity. Disadvantageous physicochemical parameters can hinder the success of a drug candidate.
Objective: Lovering et al. introduced the Fsp3 character as a measure of carbon bond saturation, which is related to the physicochemical paramethers of the drug. The pharmaceutical research focuses on the synthesis of compounds with high Fsp3 character.
Methods: To improve the physicochemical properties (clogP, solubility, more advantageous ADME profile, etc.) of drug-candidate molecules one possibility is the replacement of all-carbon aromatic systems with bioisoster heteroaromatic moieties, e.g. with one or two nitrogen atom containing systems, such as pyridines and pyridazines, etc. The other option is to increase the Fsp3 character of the drug candidates. Both of these aspects were considered in the design the new spiro[cycloalkanepyridazinones], the synthesis of which is described in the present study.
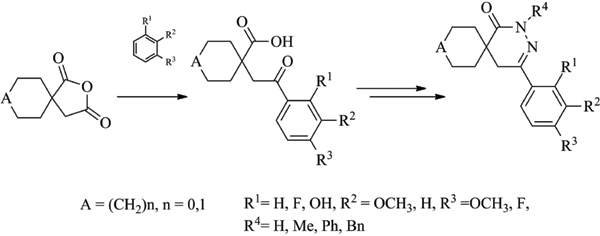
Results: Starting from 2-oxaspiro[4.5]decane-1,3-dione or 2-oxaspiro[4.4]nonane-1,3-dione, the corresponding ketocarboxylic acids were obtained by Friedel-Crafts reaction with anisole or veratrole. The ketocarboxylic acids were treated by hydrazine, methylhydrazine or phenylhydrazine to form the pyridazinone ring. N-Alkylation reaction of the pyridazinones resulted in the formation of further derivatives with high Fsp3 character.
Conclusion: A small compound library was obtained incorporating compounds with high Fsp3 characters, which predicts advantageous physico-chemical parameters (LogP, ClogP and TPSA) for potential applications in medicinal chemistry.
Keywords: Pyridazinone, spiro[cycloalkane-pyridazinone], Fsp3 character, Friedel-Crafts reaction, spiro, ketocarboxylic acid, hydrazine, phenylhydrazine.
[PMID: 11259830]
[http://dx.doi.org/10.1016/j.ddtec.2004.11.007] [PMID: 24981612]
[http://dx.doi.org/10.1021/jm020017n] [PMID: 12036371]
[http://dx.doi.org/10.1021/jm701122q] [PMID: 18232648]
[http://dx.doi.org/10.1021/jm0492002] [PMID: 15857122]
[http://dx.doi.org/10.1602/neurorx.2.4.541] [PMID: 16489364]
[http://dx.doi.org/10.1021/jm901241e] [PMID: 19827778]
[http://dx.doi.org/10.1039/c2md20347b]
[http://dx.doi.org/10.1016/j.drudis.2009.07.014] [PMID: 19729075]
[http://dx.doi.org/10.1016/j.drudis.2010.11.014] [PMID: 21129497]
[http://dx.doi.org/10.1039/C1MD00074H]
[http://dx.doi.org/10.1016/j.ejmech.2016.07.061] [PMID: 27484513]
[http://dx.doi.org/10.1016/j.ejogrb.2006.02.015] [PMID: 16621226]
[http://dx.doi.org/10.1016/j.ijcard.2011.07.022] [PMID: 21784540]
[http://dx.doi.org/10.1016/0006-2952(86)90085-7] [PMID: 3954800] ; bContreras JM, Rival YM, Chayer S, Bourguignon JJ, Wermuth CG. Aminopyridazines as acetylcholinesterase inhibitors. J Med Chem 1999; 42(4): 730-41.
[http://dx.doi.org/10.1021/jm981101z] [PMID: 10052979]
[http://dx.doi.org/10.1007/s10517-009-0638-4] [PMID: 19902096]
[http://dx.doi.org/10.1016/0002-9149(91)90356-P] [PMID: 1715125]
[http://dx.doi.org/10.1139/cjc-2015-0176]
[http://dx.doi.org/10.1016/0954-6111(95)90214-7] [PMID: 7644776]
[http://dx.doi.org/10.1111/j.1939-1676.2012.01026.x] [PMID: 23078651]
[http://dx.doi.org/10.1023/A:1015894300247] [PMID: 1808615]
[http://dx.doi.org/10.1021/jm051263c] [PMID: 16789729]
[http://dx.doi.org/10.1021/jm060265+] [PMID: 16913726]
[http://dx.doi.org/10.1021/jm00173a010] [PMID: 2146392]
[http://dx.doi.org/10.1039/CT9211901199]
[http://dx.doi.org/10.1055/s-1993-25861]
[http://dx.doi.org/10.1021/jm010837k] [PMID: 11472205]
[http://dx.doi.org/10.1021/jm010838c] [PMID: 11472206]
[http://dx.doi.org/10.1021/jo01338a043]