Abstract
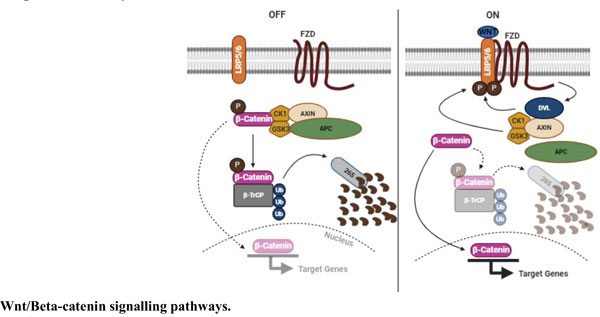
In the present review, an attempt has been made to summarize the development of various Tankyrase inhibitors focussing on Wnt/beta-Catenin pathways along with other cancer targets. The last decade witnessed a plethora of research related to the role of various genetic and epigenetic events that are responsible for the progression of multistage cancers. As a result, the discovery of various signalling pathways responsible for the development of different types of cancers has resulted in the development of molecularly targeted anticancer agents. Out of the many signalling pathways, the Wnt/beta-Catenin pathways have attracted the attention of many research groups owing to their involvement in cell proliferation, role in apoptosis induction, cellular differentiation and also cell migration. The abnormal activation of this pathways has been documented in a variety of tumour cells. Another crucial factor that makes this pathway attractive to the researches is its direct involvement with poly ADP ribose polymerases. Tankyrases are poly ADP (Adenosine Diphosphate) ribose polymerases that have the capacity to inhibit Wnt/beta-Catenin pathways and become an attractive target for anticancer drugs.
Keywords: Wnt-beta catenin, cancer, colorectal, tankyrases, telomere, cell death.
[http://dx.doi.org/10.1186/1471-2164-6-139] [PMID: 16202152]
[http://dx.doi.org/10.2174/1381612820666140630101525] [PMID: 24975604]
[http://dx.doi.org/10.3892/ol.2018.9551] [PMID: 30546421]
[http://dx.doi.org/10.1007/s12253-011-9369-8] [PMID: 21455637]
[http://dx.doi.org/10.1515/CCLM.2007.133] [PMID: 17617028]
[http://dx.doi.org/10.1371/journal.pone.0064296] [PMID: 23691191]
[http://dx.doi.org/10.1111/j.1365-2567.2009.03090.x] [PMID: 19689742]
[http://dx.doi.org/10.1016/j.canlet.2004.05.010] [PMID: 15500951]
[http://dx.doi.org/10.3892/or.16.6.1261] [PMID: 17089047]
[http://dx.doi.org/10.1007/s00262-007-0423-z] [PMID: 18026951]
[PMID: 8615664]
[http://dx.doi.org/10.1016/S0960-0760(01)00101-7] [PMID: 11595501]
[http://dx.doi.org/10.1186/bcr288] [PMID: 11305948]
[http://dx.doi.org/10.1016/j.biochi.2007.07.012] [PMID: 17825467]
[http://dx.doi.org/10.1038/cdd.2011.101] [PMID: 21818122]
[http://dx.doi.org/10.1038/nm.2799] [PMID: 22673992]
[http://dx.doi.org/10.1186/s13045-017-0471-6] [PMID: 28476164]
[http://dx.doi.org/10.1111/bph.14038] [PMID: 28910490]
[http://dx.doi.org/10.1038/79039] [PMID: 10966653]
[http://dx.doi.org/10.1016/j.canlet.2004.08.040] [PMID: 15837535]
[http://dx.doi.org/10.1038/sj.onc.1206817] [PMID: 14562050]
[http://dx.doi.org/10.1016/j.cell.2011.10.045] [PMID: 22153076]
[http://dx.doi.org/10.2174/138161213804581837] [PMID: 23016862]
[http://dx.doi.org/10.1016/j.cell.2011.10.046] [PMID: 22153077]
[http://dx.doi.org/10.1038/ncb2222] [PMID: 21478859]
[http://dx.doi.org/10.18632/oncotarget.16754] [PMID: 28422739]
[http://dx.doi.org/10.1002/cmdc.201500580] [PMID: 26781775]
[http://dx.doi.org/10.1083/jcb.200108079] [PMID: 11739401]
[http://dx.doi.org/10.1074/jbc.M203916200] [PMID: 12080061]
[http://dx.doi.org/10.1186/s13046-021-01950-6] [PMID: 33910596]
[http://dx.doi.org/10.1128/MCB.01014-06] [PMID: 17283041]
[http://dx.doi.org/10.1016/j.str.2016.07.014] [PMID: 27594684]
[http://dx.doi.org/10.1111/febs.12320] [PMID: 23648170]
[http://dx.doi.org/10.1038/cr.2012.52] [PMID: 22473005]
[http://dx.doi.org/10.1074/jbc.M007635200] [PMID: 10988299]
[http://dx.doi.org/10.1042/BJ20050885] [PMID: 16076287]
[http://dx.doi.org/10.1158/0008-5472.CAN-12-4562] [PMID: 23539443]
[http://dx.doi.org/10.1158/1078-0432.CCR-14-3081] [PMID: 26224873]
[http://dx.doi.org/10.1371/journal.pone.0048670] [PMID: 23144924]
[http://dx.doi.org/10.18632/oncotarget.8626] [PMID: 27070088]
[http://dx.doi.org/10.3390/molecules25071680] [PMID: 32268564]
[http://dx.doi.org/10.3892/ijo.2014.2406] [PMID: 24789807]
[http://dx.doi.org/10.1186/s12929-017-0329-9] [PMID: 28376884]
[http://dx.doi.org/10.1634/theoncologist.2015-0057] [PMID: 26306903]
[http://dx.doi.org/10.1073/pnas.87.12.4519] [PMID: 2162045]
[http://dx.doi.org/10.1016/j.tox.2018.09.003] [PMID: 30205152]
[http://dx.doi.org/10.1080/19420862.2018.1515565] [PMID: 30183492]
[http://dx.doi.org/10.1038/nature08356] [PMID: 19759537]
[http://dx.doi.org/10.1002/cam4.170] [PMID: 24403055]
[http://dx.doi.org/10.1186/1756-9966-32-100] [PMID: 24308762]
[http://dx.doi.org/10.1158/1078-0432.CCR-16-1179] [PMID: 27663586]
[http://dx.doi.org/10.1186/1471-2407-13-211] [PMID: 23621985]
[http://dx.doi.org/10.1111/cas.13805] [PMID: 30238564]
[http://dx.doi.org/10.1158/0008-5472.CAN-11-3336] [PMID: 22440753]
[http://dx.doi.org/10.1021/jm400807n] [PMID: 23844574]
[http://dx.doi.org/10.1021/acs.jmedchem.7b00137] [PMID: 28591512]