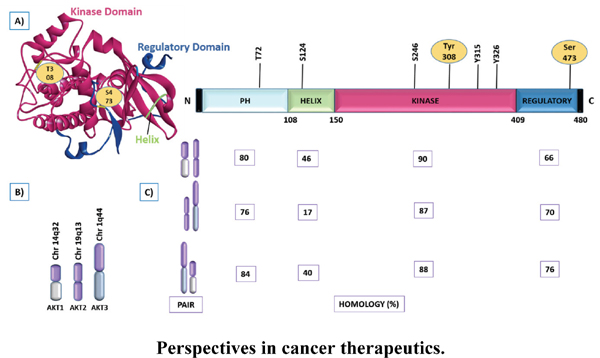
Abstract
Background: AKT/PKB is an important enzyme with numerous biological functions, and its overexpression is related to carcinogenesis. AKT stimulates different signaling pathways that are downstream of activated tyrosine kinases and phosphatidylinositol 3-kinase, hence functions as an important target for anti-cancer drugs.
Objective: In this review article, we have interpreted the role of AKT signaling pathway in cancer and the natural inhibitory effect of Thymoquinone (TQ) in AKT and its possible mechanisms.
Method: We have collected the updated information and data on AKT, its role in cancer and the inhibitory effect of TQ in AKT signaling pathway from Google Scholar, PubMed, Web of Science, Elsevier, Scopus, and many more.
Results: Many drugs are already developed, which can target AKT, but very few among them have passed clinical trials. TQ is a natural compound, mainly found in black cumin, which has been found to have potential anti-cancer activities. TQ targets numerous signaling pathways, including AKT, in different cancers. In fact, many studies revealed that AKT is one of the major targets of TQ. The preclinical success of TQ suggests its clinical studies on cancer.
Conclusion: This review article summarizes the role of AKT in carcinogenesis, its potent inhibitors in clinical trials, and how TQ acts as an inhibitor of AKT and TQ’s future as a cancer therapeutic drug.
Keywords: AKT signaling pathway, carcinogenesis, PIK3, PTEN, thymoquinone, cancer therapeutics.
[http://dx.doi.org/10.1002/ijc.31937] [PMID: 30350310]
[PMID: 25057339]
[http://dx.doi.org/10.1155/2014/761608] [PMID: 25295272]
[http://dx.doi.org/10.18632/oncotarget.17206] [PMID: 28881699]
[http://dx.doi.org/10.3389/fphar.2017.00295] [PMID: 28659794]
[http://dx.doi.org/10.1155/2018/4010629] [PMID: 29651429]
[http://dx.doi.org/10.4103/2008-7802.180412] [PMID: 27141285]
[http://dx.doi.org/10.1002/ijc.21205] [PMID: 15906362]
[http://dx.doi.org/10.1016/j.cell.2007.06.009] [PMID: 17604717]
[http://dx.doi.org/10.1038/nrd2926] [PMID: 19644473]
[http://dx.doi.org/10.1007/s11033-019-04929-x] [PMID: 31313132]
[http://dx.doi.org/10.1073/pnas.211430998] [PMID: 11572954]
[http://dx.doi.org/10.1016/S0065-230X(05)94002-5] [PMID: 16095999]
[http://dx.doi.org/10.4161/cc.5.6.2561] [PMID: 16582622]
[http://dx.doi.org/10.1093/jnci/djv171] [PMID: 26071042]
[http://dx.doi.org/10.1016/j.ctrv.2003.07.007] [PMID: 15023437]
[http://dx.doi.org/10.1074/jbc.M406731200] [PMID: 15262962]
[http://dx.doi.org/10.1101/gad.13.22.2905] [PMID: 10579998]
[http://dx.doi.org/10.3390/ijms16023202] [PMID: 25648320]
[http://dx.doi.org/10.1038/sj.onc.1209085] [PMID: 16288292]
[http://dx.doi.org/10.1186/2045-3701-4-59] [PMID: 25309720]
[PMID: 8510938]
[http://dx.doi.org/10.4161/cc.9.3.10508] [PMID: 20081374]
[http://dx.doi.org/10.1073/pnas.89.19.9267] [PMID: 1409633]
[http://dx.doi.org/10.1073/pnas.93.8.3636] [PMID: 8622988]
[http://dx.doi.org/10.1002/(SICI)1098-2744(199802)21:2<81:AID-MC1>3.0.CO;2-R] [PMID: 9496907]
[http://dx.doi.org/10.1006/bbrc.1996.1280] [PMID: 8780719]
[http://dx.doi.org/10.1016/0888-7543(88)90114-0] [PMID: 3384441]
[http://dx.doi.org/10.1016/S0959-437X(98)80062-2] [PMID: 9529606]
[http://dx.doi.org/10.1016/S0002-9440(10)61714-2] [PMID: 11485901]
[PMID: 9679957]
[http://dx.doi.org/10.1146/annurev.biochem.68.1.965] [PMID: 10872470]
[http://dx.doi.org/10.1016/S0092-8674(00)81780-8] [PMID: 9778245]
[http://dx.doi.org/10.1074/jbc.C100462200] [PMID: 11533044]
[http://dx.doi.org/10.1172/JCI16885] [PMID: 12843127]
[http://dx.doi.org/10.1126/science.292.5522.1728] [PMID: 11387480]
[http://dx.doi.org/10.1128/MCB.25.5.1869-1878.2005] [PMID: 15713641]
[http://dx.doi.org/10.1517/13543776.2011.587959] [PMID: 21635152]
[http://dx.doi.org/10.1038/nrd1902] [PMID: 16341064]
[http://dx.doi.org/10.1038/nature04869] [PMID: 16724053]
[http://dx.doi.org/10.4161/cbt.3.3.703] [PMID: 15034304]
[http://dx.doi.org/10.1038/sj.onc.1209088] [PMID: 16288295]
[http://dx.doi.org/10.1111/nyas.13422] [PMID: 28891094]
[http://dx.doi.org/10.1093/carcin/bgl151] [PMID: 16950795]
[http://dx.doi.org/10.15430/JCP.2014.19.1.23] [PMID: 25337569]
[http://dx.doi.org/10.3892/ol.2017.6526] [PMID: 28927043]
[http://dx.doi.org/10.1007/s11010-008-9722-8] [PMID: 18259841]
[http://dx.doi.org/10.1080/09674845.2011.11730326] [PMID: 21706917]
[http://dx.doi.org/10.18632/oncotarget.18770] [PMID: 29207615]
[http://dx.doi.org/10.1371/journal.pone.0136447] [PMID: 26317520]
[http://dx.doi.org/10.1002/hed.24910] [PMID: 28945300]
[http://dx.doi.org/10.1158/0008-5472.CAN-03-3241] [PMID: 14744754]
[http://dx.doi.org/10.1093/jnci/95.4.291] [PMID: 12591985]
[http://dx.doi.org/10.1158/1541-7786.MCR-12-0558] [PMID: 23319332]
[http://dx.doi.org/10.3892/etm.2015.2637] [PMID: 26622460]
[http://dx.doi.org/10.1038/modpathol.3800525] [PMID: 16341149]
[http://dx.doi.org/10.1038/nm1052] [PMID: 15156201]
[PMID: 11948129]
[http://dx.doi.org/10.1073/pnas.0602567103] [PMID: 16682621]
[http://dx.doi.org/10.1038/sj.onc.1209096] [PMID: 16288293]
[http://dx.doi.org/10.1200/JCO.2005.07.168] [PMID: 15735123]
[http://dx.doi.org/10.1158/0008-5472.CAN-04-1399] [PMID: 15466193]
[http://dx.doi.org/10.1016/j.ccr.2005.11.005] [PMID: 16338658]
[http://dx.doi.org/10.1016/j.advenzreg.2006.01.004] [PMID: 16854453]
[http://dx.doi.org/10.1038/nature12634] [PMID: 24132290]
[http://dx.doi.org/10.1038/nature12881] [PMID: 24390348]
[http://dx.doi.org/10.1038/nature12113] [PMID: 23636398]
[PMID: 22873098]
[http://dx.doi.org/10.1016/j.ijrobp.2009.03.004] [PMID: 19480971]
[http://dx.doi.org/10.1517/13543784.2010.520701] [PMID: 20846000]
[http://dx.doi.org/10.2174/156802607781696864] [PMID: 17692025]
[http://dx.doi.org/10.3816/CGC.2007.n.031] [PMID: 18272025]
[http://dx.doi.org/10.1002/cncr.27668] [PMID: 22674198]
[http://dx.doi.org/10.1186/1756-8722-7-1] [PMID: 24387695]
[http://dx.doi.org/10.1200/JCO.2011.35.5263] [PMID: 22025163]
[http://dx.doi.org/10.1007/s10637-015-0212-z] [PMID: 25637165]
[http://dx.doi.org/10.1158/1535-7163.MCT-10-0760] [PMID: 21191045]
[http://dx.doi.org/10.18632/oncotarget.6153] [PMID: 26497682]
[http://dx.doi.org/10.1371/journal.pone.0100880] [PMID: 24978597]
[http://dx.doi.org/10.1016/j.radonc.2006.07.021] [PMID: 16916558]
[http://dx.doi.org/10.1021/jm8004527] [PMID: 18800763]
[http://dx.doi.org/10.1158/1078-0432.CCR-08-1253] [PMID: 19118049]
[http://dx.doi.org/10.4155/fmc.09.5] [PMID: 21426073]
[http://dx.doi.org/10.1016/j.semcancer.2019.07.009] [PMID: 31323288]
[http://dx.doi.org/10.1158/2159-8290.CD-RW2017-093]
[http://dx.doi.org/10.1158/2159-8290.CD-16-0512] [PMID: 27872130]
[http://dx.doi.org/10.1002/ijc.30457] [PMID: 27699769]
[http://dx.doi.org/10.1158/1538-7445.AM2017-139]
[http://dx.doi.org/10.1016/j.bmcl.2008.04.074] [PMID: 18479914]
[http://dx.doi.org/10.1182/blood.V120.21.942.942]
[http://dx.doi.org/10.1093/carcin/bgt365] [PMID: 24282290]
[http://dx.doi.org/10.1158/1535-7163.MCT-17-0823] [PMID: 29695636]
[http://dx.doi.org/10.1093/carcin/bgx082] [PMID: 29029040]
[http://dx.doi.org/10.1093/jnci/dji377] [PMID: 16288123]
[http://dx.doi.org/10.3892/or.2012.2195] [PMID: 23254561]
[http://dx.doi.org/10.1016/j.freeradbiomed.2012.01.026] [PMID: 22366652]
[http://dx.doi.org/10.1016/j.taap.2013.05.031] [PMID: 23747687]
[http://dx.doi.org/10.1016/j.bbrc.2016.12.114] [PMID: 27998773]
[http://dx.doi.org/10.3892/or.2017.5662] [PMID: 28534975]
[http://dx.doi.org/10.3892/mmr.2017.7612] [PMID: 28983625]
[http://dx.doi.org/10.2174/1568009617666170315162932] [PMID: 28302032]
[http://dx.doi.org/10.1038/srep37997] [PMID: 27897231]
[http://dx.doi.org/10.1016/j.canlet.2015.08.030] [PMID: 26454217]
[http://dx.doi.org/10.1016/S0731-7085(98)00300-8] [PMID: 10698539]
[http://dx.doi.org/10.1371/journal.pone.0012124] [PMID: 20711342]
[http://dx.doi.org/10.1007/s10495-009-0421-z] [PMID: 19882352]
[http://dx.doi.org/10.1158/0008-5472.CAN-07-1483] [PMID: 17699783]
[http://dx.doi.org/10.1016/j.drudis.2020.07.019] [PMID: 32721537]
[http://dx.doi.org/10.1016/j.lfs.2013.09.009] [PMID: 24044882]
[http://dx.doi.org/10.3748/wjg.v23.i7.1171] [PMID: 28275297]
[http://dx.doi.org/10.3892/or.2014.3059] [PMID: 24603952]
[http://dx.doi.org/10.1007/s12094-014-1206-6] [PMID: 25060568]
[http://dx.doi.org/10.1371/journal.pone.0046641] [PMID: 23077516]
[http://dx.doi.org/10.1111/jfbc.12793] [PMID: 31353586]
[http://dx.doi.org/10.1007/s12038-017-9708-3] [PMID: 29229873]
[http://dx.doi.org/10.1007/s11010-016-2697-y] [PMID: 27032769]
[http://dx.doi.org/10.1016/j.mrfmmm.2010.10.007] [PMID: 21040738]
[http://dx.doi.org/10.1371/journal.pone.0061342] [PMID: 23613836]
[http://dx.doi.org/10.1007/s13277-015-4307-0] [PMID: 26500095]
[http://dx.doi.org/10.1007/s10620-014-3394-x] [PMID: 25344906]
[http://dx.doi.org/10.1155/2016/1407840] [PMID: 28105374]
[PMID: 22490899]
[http://dx.doi.org/10.3390/ijms17040474] [PMID: 27043539]
[http://dx.doi.org/10.1016/j.bmcl.2013.03.003] [PMID: 23562242]
[http://dx.doi.org/10.1016/j.fct.2013.12.015] [PMID: 24355171]
[http://dx.doi.org/10.1016/j.bbrc.2013.07.110] [PMID: 23911786]