Abstract
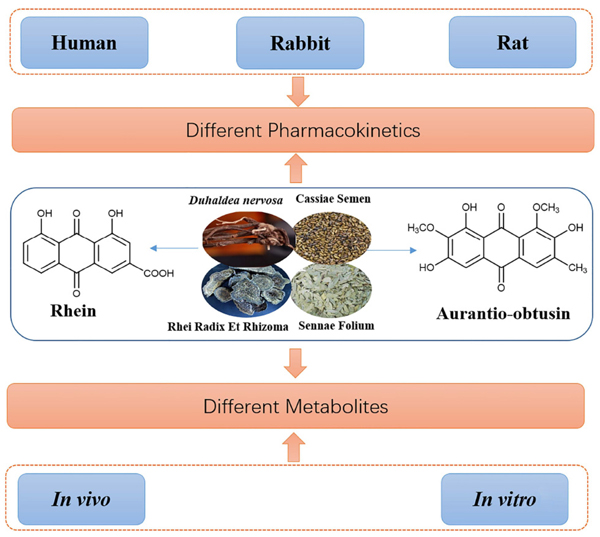
Background: Anthraquinones, rhein and aurantio-obtusin were isolated from the herb Duhaldea nervosa for the first time by our group, which were also found in plants that belong to the plant family Compositae. Anthraquinone compounds have a range of pharmacological activities such as anti-inflammatory, anti-cancer, antioxidation, anti-diabetes, etc. and can be used as a laxative, for liver protection, treatment of chronic renal failure, etc. However, in recent years, anthraquinones have been reported to be cytotoxic to the liver and kidneys. Therefore, it is very important to study the pharmacokinetics and metabolism of rhein and aurantio-obtusin, which are common ingredients in many traditional Chinese medicines (TCM). According to our research, the pharmacokinetics and metabolism of rhein and aurantio-obtusin are comprehensively summarized in the paper for the first time.
Objective: The study provides comprehensive information on pharmacokinetics and metabolism of rhein and aurantio- obtusin in different Species; meanwhile, the aim of this review is also to provide a reference for a reasonable application of TCM enriched with these two ingredients. Methods: The metabolism and pharmacokinetics of rhein and aurantio-obtusin were searched by the Web of Science, PubMed, Google scholar and some Chinese literature databases. Results: Rhein and aurantio-obtusin exist mainly in the form of metabolites in the body. Rhein and aurantio-obtusin and its metabolites might be responsible for pharmacological effects in the body. Therefore, the significance of studying the in vivo metabolites of rhein and aurantio-obtusin is not only essential to clarify their pharmacological mechanism, but also to find new active compound ingredients. The metabolism of rhein is different in different species, so the toxicity effects of rhein may also be different after oral administration in different species; however, the metabolic profiles of aurantio-obtusin in the liver microsomes of different species are similar. Conclusion: This paper not only provides detail regarding the pharmacokinetics of rhein and aurantio-obtusin, but it is anticipated that it will also facilitate further study on the metabolism of rhein and aurantio-obtusin.Keywords: Duhaldea nervosa, rhein, aurantio-obtusin, anthraquinones, pharmacokinetics, metabolism.
[http://dx.doi.org/10.1002/hlca.200900432]
[http://dx.doi.org/ 10.1093/mp/ssq070]
[http://dx.doi.org/10.1016/j.bse.2011.06.001]
[http://dx.doi.org/10.1152/ajpendo.00332.2010] [PMID: 21364120]
[http://dx.doi.org/10.3748/wjg.v19.i26.4137] [PMID: 23864776]
[http://dx.doi.org/10.1016/j.transproceed.2013.03.030] [PMID: 23953579]
[http://dx.doi.org/10.1053/joca.1999.0289] [PMID: 10806046]
[http://dx.doi.org/10.1053/joca.2000.0383] [PMID: 11300749]
[http://dx.doi.org/10.1007/s10753-011-9407-4] [PMID: 22095404]
[http://dx.doi.org/10.1016/j.cbi.2011.03.013] [PMID: 21457705]
[http://dx.doi.org/10.1016/j.oraloncology.2008.07.012] [PMID: 18804415]
[http://dx.doi.org/10.1002/(SICI)1099-1263(199803/04)18:2<117: AID-JAT486>3.0.CO;2-D] [PMID: 9570694]
[http://dx.doi.org/10.1016/j.jbiotec.2008.04.010] [PMID: 18514345]
[http://dx.doi.org/10.7150/ijbs.4575] [PMID: 23139635]
[http://dx.doi.org/10.1097/00005176-200205000-00011] [PMID: 12050580]
[http://dx.doi.org/10.1016/j.phymed.2018.03.004] [PMID: 29631806]
[http://dx.doi.org/10.1016/j.jphs.2015.05.006] [PMID: 26076958]
[http://dx.doi.org/10.3390/molecules23123093] [PMID: 30486383]
[http://dx.doi.org/10.1002/ptr.6082] [PMID: 29675883]
[http://dx.doi.org/10.1055/s-0034-1368445] [PMID: 24841966]
[http://dx.doi.org/10.1016/j.lab.2005.11.011] [PMID: 16581343]
[http://dx.doi.org/10.1016/j.jep.2011.08.028] [PMID: 21884773]
[http://dx.doi.org/10.1142/S0192415X05003508] [PMID: 16355440]
[http://dx.doi.org/10.1016/j.jpba.2011.09.001] [PMID: 21945452]
[http://dx.doi.org/10.1016/j.phymed.2016.10.002] [PMID: 27823631]
[http://dx.doi.org/10.1248/bpb.26.613] [PMID: 12736499]
[http://dx.doi.org/10.1002/bmc.873] [PMID: 17582236]
[http://dx.doi.org/10.1002/ptr.3223] [PMID: 20623608]
[PMID: 23802430]
[http://dx.doi.org/10.1080/10826076.2011.572211]
[http://dx.doi.org/10.4155/bio.12.81] [PMID: 22651564]
[http://dx.doi.org/10.3390/molecules19044058] [PMID: 24699148]
[http://dx.doi.org/10.3923/ijp.2019.19.30]
[http://dx.doi.org/10.1016/j.jep.2014.07.022] [PMID: 25046826]
[http://dx.doi.org/10.1002/bmc.1739] [PMID: 22068685]
[http://dx.doi.org/10.3390/molecules23102716] [PMID: 30360359]
[http://dx.doi.org/10.1016/j.ejps.2012.03.014] [PMID: 22521276]
[http://dx.doi.org/10.1080/03639045.2018.1522326] [PMID: 30196732]
[http://dx.doi.org/10.1080/03639045.2017.1353519] [PMID: 28692315]
[http://dx.doi.org/10.1016/j.jep.2014.02.005] [PMID: 24642020]
[http://dx.doi.org/10.1159/000138436] [PMID: 3368515]
[PMID: 9107544]
[http://dx.doi.org/10.1002/bmc.1467] [PMID: 21321975]
[http://dx.doi.org/10.1016/j.jpba.2015.03.043] [PMID: 25912849]
[http://dx.doi.org/10.1016/j.jpba.2016.03.038] [PMID: 27031575]
[http://dx.doi.org/10.1016/j.cbi.2018.01.001] [PMID: 29331654]
[http://dx.doi.org/10.1016/S0378-8741(02)00222-2] [PMID: 12499069]
[http://dx.doi.org/10.1002/jssc.201200378] [PMID: 22753114]
[http://dx.doi.org/10.1159/000138434] [PMID: 3368513]
[http://dx.doi.org/10.1002/bdd.2510130403] [PMID: 1600110]
[http://dx.doi.org/10.1002/ptr.2572] [PMID: 18814214]
[http://dx.doi.org/10.3109/00498254.2014.984794] [PMID: 25815638]
[http://dx.doi.org/10.1016/j.cbi.2014.05.006] [PMID: 24854283]
[http://dx.doi.org/10.1021/acs.jafc.6b01872] [PMID: 27362917]
[http://dx.doi.org/10.1093/chromsci/bmt159] [PMID: 24149003]
[http://dx.doi.org/10.1016/j.jep.2015.04.008] [PMID: 25907980]
[http://dx.doi.org/10.3390/molecules22111803] [PMID: 29143757]
[http://dx.doi.org/10.1002/jssc.201900007] [PMID: 31037812]
[http://dx.doi.org/10.1016/j.jpba.2019.07.001] [PMID: 31288192]
[http://dx.doi.org/10.3109/00498254.2014.895881] [PMID: 24618000]