Abstract
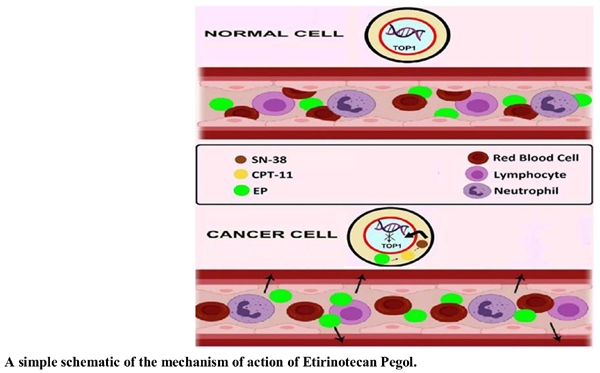
Background: Due to the increasing prevalence of cancer and the inadequacy of current therapies, the development of novel antitumor pharmaceutics with higher efficacies and lower adverse effects is considered a fundamental tenet of contemporary cancer management. Poly-Ethylene-Glycol (PEG) attachment is a novel pharmaceutical technology to improve the efficacy and safety of chemotherapies. Etirinotecan Pegol (EP), also known as NKTR-102, is the PEGylated form of Irinotecan (CPT-11), which causes cancer cell apoptosis by inhibiting the topoisomerase I enzyme.
Objective: The present study reviews and evaluates various reports of the EP’s anti-tumor activity in various cancers.
Data Source: Studies were identified using the Scopus database, with no exclusions. The search terms included Etirinotecan Pegol and NKTR-102, which yielded 125 articles (66 and 59 articles, respectively). In addition, the clinicaltrials.gov website was used to find ongoing studies, which resulted in the addition of two studies.
Study Eligibility Criteria: Subsequently, we excluded studies that were published in languages other than English, duplicate articles, and studies with no data.
Results: This systematic review clarifies that EP possesses numerous advantages over many other medications, such as safety, efficacy, increased half-life, increased health-related quality of life, increased overall survival, increased progression-free survival, and decreasing the adverse events in the treatment of various cancers.
Conclusion: Therefore, Etirinotecan Pegol may represent a major contribution to the treatment of various cancers in the future.
Keywords: Cancer, chemotherapy, drug safety, toxicology, clinical trials, topoisomerase inhibitors.
[http://dx.doi.org/10.3322/caac.21387] [PMID: 28055103]
[http://dx.doi.org/10.18632/oncotarget.27274] [PMID: 31695842]
[http://dx.doi.org/10.2174/1566524020666200903120344] [PMID: 32881669]
[http://dx.doi.org/10.1111/bju.13157] [PMID: 25908534]
[http://dx.doi.org/10.1007/978-3-319-75693-6_13]
[http://dx.doi.org/10.1038/nrclinonc.2016.25] [PMID: 26977780]
[http://dx.doi.org/10.1016/S0140-6736(16)31594-X] [PMID: 28017406]
[http://dx.doi.org/10.1038/527S108a] [PMID: 26580158]
[http://dx.doi.org/10.1016/j.colsurfb.2014.12.041] [PMID: 25591851]
[http://dx.doi.org/10.1186/s13058-018-1008-9] [PMID: 30005601]
[http://dx.doi.org/10.1038/s41389-020-0204-5] [PMID: 32054828]
[http://dx.doi.org/10.3390/cancers12051270] [PMID: 32429591]
[http://dx.doi.org/10.1111/cas.13455] [PMID: 29168596]
[http://dx.doi.org/10.1016/j.reprotox.2020.07.015] [PMID: 32810592]
[http://dx.doi.org/10.1016/j.canlet.2017.09.045] [PMID: 29024812]
[http://dx.doi.org/10.1158/0008-5472.CAN-18-2636] [PMID: 30733195]
[http://dx.doi.org/10.1158/1541-7786.MCR-18-0151] [PMID: 29934326]
[http://dx.doi.org/10.1038/s41467-018-05406-y] [PMID: 30082834]
[http://dx.doi.org/10.1038/nrm.2016.111] [PMID: 27649880]
[http://dx.doi.org/10.1016/j.jconrel.2016.11.015] [PMID: 27871992]
[http://dx.doi.org/10.1007/s00280-016-2998-6] [PMID: 27169793]
[http://dx.doi.org/10.1021/np030498t] [PMID: 14987046]
[http://dx.doi.org/10.1016/j.jconrel.2011.03.007] [PMID: 21406204]
[http://dx.doi.org/10.1200/JCO.1992.10.4.647] [PMID: 1312588]
[http://dx.doi.org/10.1200/JCO.1996.14.12.3056] [PMID: 8955650]
[http://dx.doi.org/10.1200/JCO.2001.19.14.3312] [PMID: 11454878]
[http://dx.doi.org/10.1007/s11060-011-0629-y] [PMID: 21706359]
[http://dx.doi.org/10.1093/oxfordjournals.annonc.a059329] [PMID: 8589028]
[http://dx.doi.org/10.1200/JCO.1996.14.9.2540] [PMID: 8823333]
[http://dx.doi.org/10.1097/00000421-199706000-00019] [PMID: 9167758]
[http://dx.doi.org/10.1200/JCO.2006.06.5821] [PMID: 17135646]
[http://dx.doi.org/10.1200/JCO.2001.19.8.2114] [PMID: 11304763]
[http://dx.doi.org/10.1006/gyno.2000.5807] [PMID: 10831357]
[http://dx.doi.org/10.1182/blood.V88.7.2473.bloodjournal8872473] [PMID: 8839838]
[http://dx.doi.org/10.1002/cncr.11180] [PMID: 12599230]
[http://dx.doi.org/10.1002/cncr.21700] [PMID: 16456814]
[http://dx.doi.org/10.1016/j.bmcl.2016.12.085] [PMID: 28073672]
[http://dx.doi.org/10.1002/cam4.1083] [PMID: 28544814]
[http://dx.doi.org/10.1038/bjc.2015.257] [PMID: 26291057]
[http://dx.doi.org/10.1634/theoncologist.10-9-686] [PMID: 16249347]
[http://dx.doi.org/10.1177/0091270009343699] [PMID: 19808951]
[http://dx.doi.org/10.1200/JCO.2006.08.3998] [PMID: 17513814]
[http://dx.doi.org/10.2165/00003088-199733040-00001] [PMID: 9342501]
[http://dx.doi.org/10.1016/S0140-6736(00)02034-1] [PMID: 10744089]
[http://dx.doi.org/10.1056/NEJMoa032691] [PMID: 15175435]
[http://dx.doi.org/10.1038/s41419-018-0700-0] [PMID: 29855512]
[http://dx.doi.org/10.1016/S0140-6736(16)30354-3] [PMID: 27156933]
[http://dx.doi.org/10.1200/JCO.2004.10.047] [PMID: 15254052]
[http://dx.doi.org/10.1200/JCO.2004.09.053] [PMID: 14691125]
[http://dx.doi.org/10.1056/NEJMoa003034] [PMID: 11784874]
[http://dx.doi.org/10.1215/S1152851703000292] [PMID: 14769140]
[http://dx.doi.org/10.1016/S0140-6736(00)03167-6] [PMID: 11089838]
[http://dx.doi.org/10.1016/j.pathophys.2017.07.002] [PMID: 28801072]
[http://dx.doi.org/10.1155/2017/7350251] [PMID: 28167974]
[http://dx.doi.org/10.1016/j.tiv.2005.06.045] [PMID: 16271446]
[http://dx.doi.org/10.1007/s00216-014-8237-2] [PMID: 25311193]
[http://dx.doi.org/10.1002/ijc.30082] [PMID: 26950035]
[PMID: 16907]
[http://dx.doi.org/10.1007/s00280-014-2577-7] [PMID: 25228368]
[http://dx.doi.org/10.1186/s12885-015-1672-4] [PMID: 26463521]
[http://dx.doi.org/10.1007/s00280-016-3192-6] [PMID: 27904955]
[http://dx.doi.org/10.1158/1078-0432.CCR-12-1201] [PMID: 23136196]
[http://dx.doi.org/10.1016/S1470-2045(13)70429-7] [PMID: 24095299]
[http://dx.doi.org/10.1200/JCO.2012.45.1278] [PMID: 24081946]
[http://dx.doi.org/10.1016/j.ygyno.2017.08.026] [PMID: 28935273]
[http://dx.doi.org/10.1007/s11060-015-1795-0] [PMID: 25935109]
[http://dx.doi.org/10.1016/j.cllc.2017.10.007] [PMID: 29129435]
[http://dx.doi.org/10.1007/s00280-017-3438-y] [PMID: 29043412]
[http://dx.doi.org/10.1016/S1470-2045(15)00332-0] [PMID: 26482278]
[http://dx.doi.org/10.1016/j.ejca.2017.02.011] [PMID: 28360015]
[http://dx.doi.org/10.1007/s10549-017-4304-7] [PMID: 28612225]