Abstract
Triazole ring is a cyclic scaffold containing three heteroatoms of nitrogen. They display a broad variety of biological activities. The uncatalyzed/catalyzed 1,3-dipolar cycloadditions are a chemical reaction between a 1,3-dipole and a dipolarophile to achieve 1,2,3-triazoles.
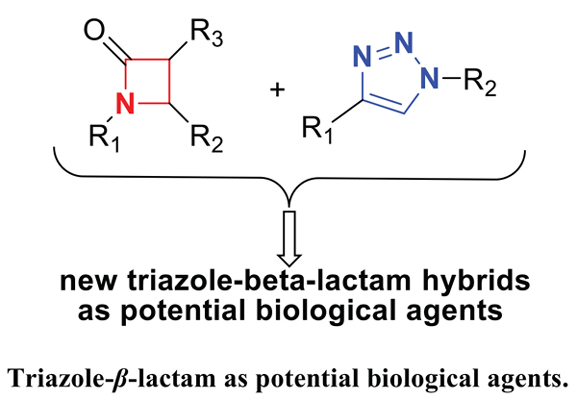
The hybrid approach is an innovative and powerful synthetic tool for the synthesis of two or more distinct entities in one molecule with novel biological activities. Owing to the high potential of β-lactams to display noticeable biological properties, these compounds have been one of the important ingredients in hybrid molecules. The four-membered lactams have been recognized as a part of penicillin. There are various synthetic protocols for the synthesis of β-lactams. Staudinger reaction of the Schiff bases with diphenylketenes is a successful and famous strategy for the synthesis of these products.
Even though, the number of heterocyclic compounds is limited, plenty of hybrids based on heterocyclic compounds can be designed and prepared. The synthesis of hybrid products of triazole-β-lactam has proved to be highly challenging. The current review article outlines the diversity and creativity in the elegant synthesis of triazole-β-lactam hybrids as potential biological agents. Molecules including isatin, ferrocene, bile acid, chalcone, and etc were attached to β-lactam with triazole linker, as well.
Keywords: Triazole, β-Lactam, molecule hybrids, biological agents, Triazole-β-lactam hybrids, triazole linked β-lactamanother molecule.
[http://dx.doi.org/10.1016/j.ejmech.2016.08.039] [PMID: 27598238]
[http://dx.doi.org/10.1002/9781119998372]
[http://dx.doi.org/10.1002/aoc.4974]
[http://dx.doi.org/10.3762/bjoc.14.253] [PMID: 30498525]
[http://dx.doi.org/10.1055/s-0034-1378206]
[http://dx.doi.org/10.3762/bjoc.13.193] [PMID: 29062417]
[http://dx.doi.org/10.1016/j.ejmech.2019.07.043] [PMID: 31330449]
[http://dx.doi.org/10.1016/j.ejmech.2019.06.036] [PMID: 31260892]
[http://dx.doi.org/10.1021/acs.orglett.8b03648] [PMID: 30638388]
[http://dx.doi.org/10.1016/j.ejmech.2019.111700] [PMID: 31546197]
[http://dx.doi.org/10.1016/j.ejmech.2019.01.047] [PMID: 30711831]
[http://dx.doi.org/10.1016/j.bmcl.2010.06.141] [PMID: 20655212]
[http://dx.doi.org/10.1016/j.ejmech.2019.04.043] [PMID: 31009913]
[http://dx.doi.org/10.1081/NCN-100108325] [PMID: 11794800]
[http://dx.doi.org/10.1039/C9GC03769A]
[http://dx.doi.org/10.1039/D0GC00865F]
[http://dx.doi.org/10.1021/acssuschemeng.9b05978]
[http://dx.doi.org/10.1021/acssuschemeng.9b03118]
[http://dx.doi.org/10.1039/C9RA04841C]
[http://dx.doi.org/10.1021/jo400164y] [PMID: 23535067]
[http://dx.doi.org/10.1021/jo5010422] [PMID: 25105212]
[http://dx.doi.org/10.1002/ardp.201900293] [PMID: 31917485]
[http://dx.doi.org/10.1016/j.bioorg.2019.103515] [PMID: 31884134]
[http://dx.doi.org/10.1016/j.tet.2017.02.042]
[http://dx.doi.org/10.1039/C9NJ02759A]
[http://dx.doi.org/10.1021/acs.orglett.9b01192] [PMID: 31059273]
[http://dx.doi.org/10.1128/AAC.00128-19] [PMID: 30782985]
[http://dx.doi.org/10.1002/slct.201803785]
[http://dx.doi.org/10.1016/j.tet.2017.01.009]
[http://dx.doi.org/10.1002/1521-3773(20011203)40:23<4377:AID-ANIE4377>3.0.CO;2-J] [PMID: 12404420]
[http://dx.doi.org/10.1016/j.ejmech.2019.111744] [PMID: 31605865]
[http://dx.doi.org/10.2174/092986707781058805] [PMID: 17627520]
[http://dx.doi.org/10.1016/j.ejmech.2019.111587] [PMID: 31404864]
[http://dx.doi.org/10.1016/j.ejmech.2019.06.069] [PMID: 31301563]
[http://dx.doi.org/10.1039/C6NJ03117J]
[http://dx.doi.org/10.1007/s00044-015-1474-x]
[http://dx.doi.org/10.1002/open.201600052] [PMID: 27777842]
[http://dx.doi.org/10.1002/cmdc.201600258] [PMID: 27465595]
[http://dx.doi.org/10.1021/jo301113g] [PMID: 22812653]
[http://dx.doi.org/10.1007/s00044-014-0956-6] [PMID: 32214766]
[http://dx.doi.org/10.1016/j.ejmech.2013.03.019] [PMID: 23631874]
[http://dx.doi.org/10.1016/j.ejmech.2011.10.033] [PMID: 22071256]
[http://dx.doi.org/10.1039/c2dt30514c] [PMID: 22473422]
[http://dx.doi.org/10.1039/C2DT32148C] [PMID: 23108229]
[http://dx.doi.org/10.1039/b809221d] [PMID: 18843413]
[http://dx.doi.org/10.1016/j.bmcl.2008.01.102] [PMID: 18267360]
[http://dx.doi.org/10.1007/s13738-018-1330-2]
[http://dx.doi.org/10.1002/cbdv.201900462] [PMID: 31788939]
[http://dx.doi.org/10.1002/jhet.3136]