Abstract
Background: Cerium oxide nanoparticles (nanoceria) are efficient free-radical scavengers due to their dual valence state and thus exhibit optical and catalytic properties. Therefore, the main purpose of this work was to understand the peroxidase mimic activity of polymer-stabilized nanoceria for enzyme-less H2O2 sensing by fluorescence spectrometer.
Objective: This research revealed the development of fluorescence hydrogen peroxide nanosensor based on the peroxidase-like activity of polyacrylic acid stabilized nanoceria (PAA-CeO2 Nps).
Methods: PAA-CeO2 Nps were synthesized by simple cross-linking reaction at a low temperature and characterized by XRD, SEM, Zeta potential, TGA, FT-IR and UV-VIS spectroscopic analysis. H2O2 sensing was performed by a fluorescence spectrometer.
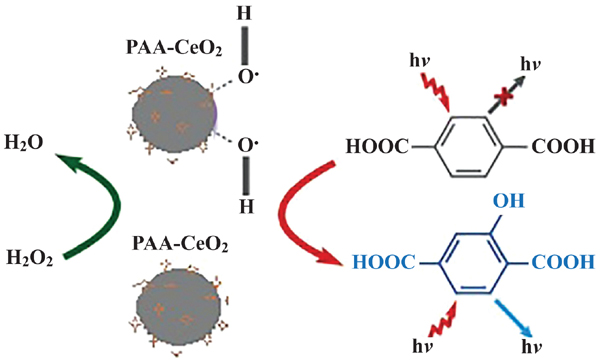
Results: The synthesized polymer nanocomposite was characterized by XRD, SEM, TGA, FT-IR and UV-VIS spectroscopic analysis. The XRD diffraction patterns confirmed the polycrystalline nature and SEM micrograph showed nanoparticles having hexagonal symmetry and crystallite size of 32 nm. The broad peak of Ce–O bond appeared at 508 cm-1. UV-VIS measurements revealed a welldefined absorbance peak around 315 nm and an optical band-gap of 3.17 eV. As synthesized PAACeO2 Nps effectively catalysed the decomposition of hydrogen peroxide (H2O2) into hydroxyl radicals. Then terephthalic acid was oxidized by hydroxyl radical to form a highly fluorescent product. Under optimized conditions, the linear range for determination of hydrogen peroxide was 0.01 - 0.2 mM with a limit of detection (LOD) of 1.2 μM.
Conclusion: The proposed method is ideally suited for the sensing of H2O2 at a low cost and this detection system enabled the sensing of analytes (sugars), which can enzymatically generate hydrogen peroxide.
Keywords: Cerium, biosensor, PAA-CeO2, hydrogen peroxide, fluorescence, sensor.
[http://dx.doi.org/10.1007/s00262-011-1082-7] [PMID: 21811786]
[http://dx.doi.org/10.1016/S0891-5849(00)00510-4] [PMID: 11295356]
[PMID: 1846317]
[http://dx.doi.org/10.1021/nn1010057] [PMID: 20565121]
[http://dx.doi.org/10.1016/j.snb.2015.01.064]
[http://dx.doi.org/10.1016/j.electacta.2015.06.067]
[http://dx.doi.org/10.1016/j.electacta.2014.11.170]
[http://dx.doi.org/10.1016/j.bios.2014.05.048] [PMID: 24912038]
[http://dx.doi.org/10.1016/j.jelechem.2016.08.008]
[http://dx.doi.org/10.3390/ma7042945] [PMID: 28788600]
[http://dx.doi.org/10.1006/jmbi.1999.3458] [PMID: 10656833]
[http://dx.doi.org/10.1016/j.enzmictec.2003.08.005]
[http://dx.doi.org/10.1016/j.elecom.2003.12.004]
[http://dx.doi.org/10.1351/PAC-CON-09-11-40]
[http://dx.doi.org/10.1002/adma.200903783] [PMID: 20564257]
[http://dx.doi.org/10.1002/adfm.201001302]
[http://dx.doi.org/10.1002/cphc.201100906] [PMID: 22383315]
[http://dx.doi.org/10.1039/c1cc14300j] [PMID: 21892508]
[http://dx.doi.org/10.1002/chem.201100478] [PMID: 21769953]
[http://dx.doi.org/10.1016/j.bios.2014.01.034] [PMID: 24534581]
[http://dx.doi.org/10.1016/j.carbon.2013.06.025]
[http://dx.doi.org/10.1039/C4EN00079J]
[http://dx.doi.org/10.1016/j.msec.2003.09.103]
[http://dx.doi.org/10.1360/N032013-00060]
[http://dx.doi.org/10.1021/la900730b] [PMID: 19422256]
[http://dx.doi.org/10.1039/C0CE00467G]
[http://dx.doi.org/10.1039/c2cc32833j] [PMID: 22760735]
[http://dx.doi.org/10.1002/adfm.200500602]
[http://dx.doi.org/10.1007/s00604-003-0090-5]
[http://dx.doi.org/10.1016/j.talanta.2008.10.032] [PMID: 19159810]
[http://dx.doi.org/10.1016/j.jphotochem.2011.05.003]
[http://dx.doi.org/10.3923/jas.2012.1734.1737]
[http://dx.doi.org/10.1021/jp036815m]
[http://dx.doi.org/10.1021/cm0507967]
[http://dx.doi.org/10.1016/j.jbiotec.2009.04.005] [PMID: 19393698]
[http://dx.doi.org/10.1039/a907510k]
[http://dx.doi.org/10.2147/IJN.S113508] [PMID: 27785011]
[http://dx.doi.org/10.1590/1980-5373-MR-2015-0698]
[http://dx.doi.org/10.1021/cg900809b]
[http://dx.doi.org/10.1002/adfm.201707178]
[http://dx.doi.org/10.1186/s40199-015-0124-7] [PMID: 26381740]
[http://dx.doi.org/10.1021/jp061134n] [PMID: 16771388]
[http://dx.doi.org/10.17159/0379-4350/2015/v68a15]
[http://dx.doi.org/10.1002/adfm.201807672]
[http://dx.doi.org/10.1021/nl4042356] [PMID: 24397241]
[http://dx.doi.org/10.1016/j.snb.2017.05.069]
[http://dx.doi.org/10.1021/acsami.5b03591] [PMID: 26076372]
[http://dx.doi.org/10.1016/j.aca.2015.02.024] [PMID: 25818144]
[http://dx.doi.org/10.1039/C4AY02801E]
[http://dx.doi.org/10.1039/C4TB01702A]
[http://dx.doi.org/10.1016/j.snb.2015.04.091]
[http://dx.doi.org/10.1002/elan.201800656]
[http://dx.doi.org/10.1149/2.0161705jes]
[http://dx.doi.org/10.1166/jnn.2016.12963]
[http://dx.doi.org/10.1016/j.bios.2010.07.071] [PMID: 20729064]
[http://dx.doi.org/10.1016/j.elecom.2010.03.031]
[http://dx.doi.org/10.1021/jp902830z]
[http://dx.doi.org/10.1002/adfm.201706006]
[http://dx.doi.org/10.1007/s10876-012-0497-4]
[http://dx.doi.org/10.1016/j.snb.2010.06.019]
[http://dx.doi.org/10.1016/j.electacta.2012.01.030]
[http://dx.doi.org/10.3390/s8085141] [PMID: 27873806]
[http://dx.doi.org/10.1007/s00604-011-0731-z]
[http://dx.doi.org/10.1016/j.electacta.2011.11.045]
[http://dx.doi.org/10.1016/j.bios.2006.08.013] [PMID: 16996257]
[http://dx.doi.org/10.1016/j.bios.2011.12.037] [PMID: 22236778]
[http://dx.doi.org/10.1016/j.talanta.2008.09.042] [PMID: 19084672]
[http://dx.doi.org/10.1016/j.electacta.2010.11.053]
[http://dx.doi.org/10.1021/bm070329d] [PMID: 17824641]
[http://dx.doi.org/10.1016/j.electacta.2010.01.035]
[http://dx.doi.org/10.1021/ac011052p] [PMID: 11924592]
[http://dx.doi.org/10.1016/j.snb.2016.08.143]