Abstract
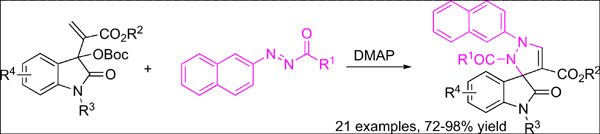
Background: A cheap and commercially available organocatalyst, 4-dimethylaminopyridine was successfully employed in the regioselective [3+2] cycloaddition of isatin-derived Morita-Baylis- Hillman carbonates with azonaphthalenes for the construction of 3-spiropyrazole-2-oxindoles in excellent yields under mild conditions.
Methods: In the presence of 4-dimethylaminopyridine with a loading of 10 mol%, a series of isatinderived Morita-Baylis-Hillman carbonates reacted smoothly with azonaphthalenes in dichloromethane at room temperature to furnish 3-spiropyrazole-2-oxindoles in 72-98% yield.
Results and Conclusion: In summary, we have developed reasonably cheap and commercially available 4-dimethylaminopyridine-mediated regioselective [3+2] annulations between isatin-derived Morita- Baylis-Hillman carbonates and azonaphthalenes for the construction of 3-spiropyrazole-2-oxindoles under mild conditions.
Keywords: Annulation, azonaphthalenes, MBH carbonates, 4-dimethylaminopyridine, regioselective, spirooxindole.
[http://dx.doi.org/10.1002/anie.200701342] [PMID: 17943924]
[http://dx.doi.org/10.1016/j.ejmech.2015.03.020] [PMID: 25791677]
[http://dx.doi.org/10.1016/j.ejmech.2014.06.056] [PMID: 24994707]
[http://dx.doi.org/10.1002/adsc.201000161]
[http://dx.doi.org/10.1021/cr300135y] [PMID: 22950860]
[http://dx.doi.org/10.1039/c2cs35100e] [PMID: 22899437]
[http://dx.doi.org/10.1039/c2ob25184a] [PMID: 22581310]
[http://dx.doi.org/10.1002/ajoc.201200180]
[http://dx.doi.org/10.1002/adsc.201200808]
[http://dx.doi.org/10.1016/j.tet.2014.08.005]
[http://dx.doi.org/10.1021/cs401172r]
[http://dx.doi.org/10.1055/s-0034-1378837]
[http://dx.doi.org/10.1039/C7QO00163K]
[http://dx.doi.org/10.1039/b924066g] [PMID: 20369196]
[http://dx.doi.org/10.1039/C5RA02794B]
[http://dx.doi.org/10.1021/ol201776h] [PMID: 21815615]
[http://dx.doi.org/10.1021/acs.orglett.5b00456] [PMID: 25781216]
[http://dx.doi.org/10.1021/acs.joc.6b01315] [PMID: 27649297]
[http://dx.doi.org/10.1002/adsc.201600954]
[http://dx.doi.org/10.1002/adsc.201800552]
[http://dx.doi.org/10.1038/s41929-019-0247-1]
[http://dx.doi.org/10.1039/C9CC06360A] [PMID: 31588462]
[http://dx.doi.org/10.1038/nchem.2866] [PMID: 29256504]
[http://dx.doi.org/10.1039/C9SC00810A] [PMID: 31391898]
[http://dx.doi.org/10.1021/acs.orglett.9b02507] [PMID: 31353910]
[http://dx.doi.org/10.1021/acs.orglett.7b02144] [PMID: 28846432]
[http://dx.doi.org/10.1039/C8QO00487K]
[http://dx.doi.org/10.1039/C8RA09453E]
[http://dx.doi.org/10.1021/acs.orglett.8b03612] [PMID: 30574783]
[http://dx.doi.org/10.1039/C5OB00865D] [PMID: 26133693]