Abstract
Background: Recent human and animal studies have demonstrated the oncostatic properties of N-acetyl-5-methoxytryptamine (melatonin) in different types of cancer. However, in few cancer cell lines including colorectal cancer cell line (HT-29), acute T cell leukemia cell line (JURKAT) and cervical cancer cell line (HeLa), precise oncostatic mechanism induced by melatonin is yet to be described.
Objectives: The aim of this study is to investigate the effects of melatonin in HT-29, JURKAT and HeLa cells and to determine the underlying molecular mechanism.
Methods: Cell viability was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay while cell cycle, apoptosis and membrane potential were analysed by flow cytometry. Reactive oxygen species (ROS) was detected by 2',7'-dichlorofluorescein diacetate (DCFH-DA) staining. Protein expressions were determined by Western blot.
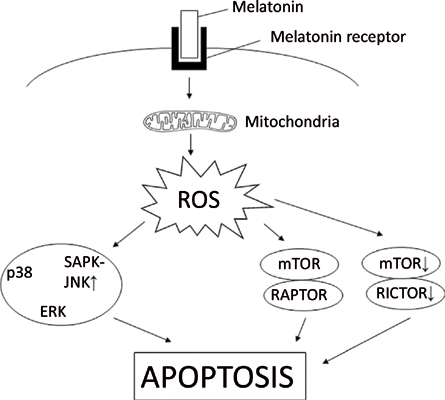
Results: Our results showed that melatonin suppressed cell proliferation, increased the number of sub G1 hypodiploid cells and cell cycle arrest in HT-29, JURKAT and HeLa cells. Besides, melatonin also induced early and late apoptosis, although there were marked variations in responses between different cell lines (sensitivity; HeLa > HT-29 >JURKAT). Apart from that, staining with DCHF-DA demonstrated ROS production that was induced in a dose-dependent manner in HeLa, HT-29 and JURKAT cells. Moreover, the apoptotic process and oncostatic effect of melatonin were seen to be associated with extracellular-signal-regulated kinase (ERK) and stress-activated protein kinase/c-Jun NH (2)-terminal kinase (SAPK-JNK) signalling cascades in HeLa cells. In HT-29 and JURKAT cells, melatonin induced apoptosis via activation of p38 mitogen-activated protein kinases (p38), ERK and SAPK-JNK signalling pathways. In all three cell lines, the apoptotic event was triggered by the mammalian target of rapamycin (mTOR)-mediated activation of the downstream target rapamycininsensitive companion of mTOR (RICTOR) and/or regulatory-associated protein of mTOR (RAPTOR) proteins.
Conclusion: Our findings confirm that melatonin induces apoptosis through reactive oxygen speciesmediated dysregulated mitogen-activated protein kinase (MAPK) and mTOR signalling pathways in these cancer cell lines.
Keywords: Melatonin, apoptosis, cancer, reactive oxygen species, MAPK, mTOR.
[http://dx.doi.org/10.1001/jamaoncol.2015.0735]
[http://dx.doi.org/10.1016/S0140-6736(16)31679-8] [PMID: 27733284]
[http://dx.doi.org/10.1186/1477-7800-4-10] [PMID: 17470282]
[http://dx.doi.org/10.1080/10409230903044914] [PMID: 19635037]
[http://dx.doi.org/10.1111/j.1600-079X.2006.00342.x] [PMID: 16879318]
[http://dx.doi.org/10.1111/j.1600-079X.2011.00946.x] [PMID: 22225575]
[http://dx.doi.org/10.1371/journal.pone.0144517] [PMID: 26656265]
[http://dx.doi.org/10.1002/ar.21361] [PMID: 21416626]
[http://dx.doi.org/10.1038/s41419-018-0383-6] [PMID: 29500364]
[http://dx.doi.org/10.1111/jpi.12369] [PMID: 27696512]
[http://dx.doi.org/10.7150/jca.10196] [PMID: 25368672]
[PMID: 15073572]
[http://dx.doi.org/10.1111/j.1600-079X.2006.00315.x] [PMID: 16635015]
[http://dx.doi.org/10.1111/j.1600-079X.2012.00974.x] [PMID: 22288984]
[PMID: 11335879]
[http://dx.doi.org/10.1111/jpi.12007] [PMID: 23013414]
[http://dx.doi.org/10.1111/jpi.12270] [PMID: 26292662]
[http://dx.doi.org/10.2174/1871520614666140812110246] [PMID: 25323035]
[PMID: 9218571]
[http://dx.doi.org/10.1111/jpi.12119] [PMID: 24484372]
[http://dx.doi.org/10.3892/mmr.2015.3599] [PMID: 25873273]
[http://dx.doi.org/10.1016/j.micron.2013.12.003] [PMID: 24530360]
[http://dx.doi.org/10.1111/jpi.12131] [PMID: 24628039]
[http://dx.doi.org/10.1111/j.1600-079X.2007.00463.x] [PMID: 17645698]
[http://dx.doi.org/10.1111/j.1600-079X.2010.00850.x] [PMID: 21392090]
[http://dx.doi.org/10.1016/j.ejphar.2012.02.011] [PMID: 22532966]
[http://dx.doi.org/10.18632/oncotarget.20949] [PMID: 29156677]
[http://dx.doi.org/10.1007/s10495-006-0623-6] [PMID: 17191113]
[http://dx.doi.org/10.1111/j.1582-4934.2008.00318.x] [PMID: 18373739]
[http://dx.doi.org/10.1159/000438587] [PMID: 26645893]
[http://dx.doi.org/10.1371/journal.pone.0101132] [PMID: 24992189]
[http://dx.doi.org/10.18632/oncotarget.6600] [PMID: 26678033]
[http://dx.doi.org/10.1093/jnci/djw182] [PMID: 27576731]
[http://dx.doi.org/10.1158/1078-0432.CCR-04-0361] [PMID: 15501983]
[PMID: 24600487]
[http://dx.doi.org/10.1002/ijc.20837] [PMID: 15800915]
[http://dx.doi.org/10.1111/j.1600-079X.2012.01006.x] [PMID: 22582944]
[http://dx.doi.org/10.1111/j.1600-079X.1998.tb00390.x] [PMID: 9885990]
[http://dx.doi.org/10.1002/(SICI)1099-1263(200001/02)20:1<21::AID-JAT623>3.0.CO;2-M] [PMID: 10641013]
[PMID: 30003730]
[http://dx.doi.org/10.1016/j.bbrc.2018.06.162] [PMID: 29966656]
[PMID: 12183413]
[http://dx.doi.org/10.1111/j.1600-079X.2010.00765.x] [PMID: 20459460]
[PMID: 3167858]
[http://dx.doi.org/10.1111/j.1600-079X.1999.tb00574.x] [PMID: 10231725]
[http://dx.doi.org/10.1158/1078-0432.CCR-07-4790] [PMID: 18223206]
[http://dx.doi.org/10.3892/ijo.2018.4319] [PMID: 29568920]
[http://dx.doi.org/10.3892/ijo.2015.2831] [PMID: 25586525]
[http://dx.doi.org/10.1681/ASN.2008111186] [PMID: 19875810]
[http://dx.doi.org/10.1016/S0092-8674(02)00808-5] [PMID: 12150925]
[http://dx.doi.org/10.4161/cc.7.21.6965] [PMID: 18971624]
[http://dx.doi.org/10.1038/s41419-017-0132-2] [PMID: 29374144]
[http://dx.doi.org/10.1586/14789450.2014.939635] [PMID: 25059473]