Abstract
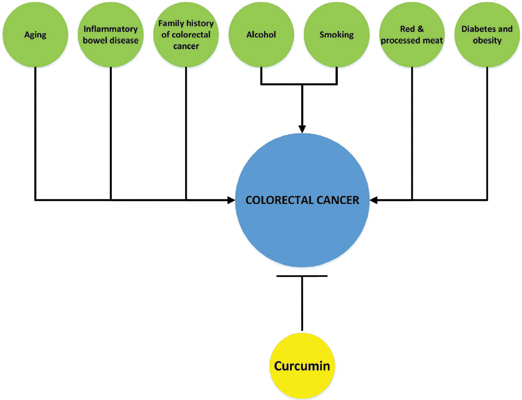
Background: Colorectal cancer is the third important cause of cancer-associated deaths across the world. Hence, there is an urgent need for understanding the complete mechanism associated with colorectal cancer, which in turn can be utilized toward early detection as well as the treatment of colorectal cancer in humans. Though colorectal cancer is a complex process and chemotherapy is the first step toward the treatment of colorectal cancer, recently several studies suggested that dietary phytochemicals may also aid significantly in reducing colorectal cancer risk in human. However, only few phytochemicals, specifically curcumin derived from the rhizomes of Curcuma longa, have better chemotherapeutic property, which might be because of its ability to regulate the activity of key factors associated with the initiation, promotion, as well as progression of tumors.
Objectives: In the present review, the authors made an attempt to summarize the physiochemical properties of curcumin, which in turn prevent colorectal cancer via regulating numerous cell signaling as well as genetic pathways.
Conclusion: Accumulated evidence suggested that curcumin suppresses tumour/colon cancer in various ways, (a) restricting cell cycle progression, or stimulating apoptosis, (b) restricting angiogenesis, anti-apoptotic proteins expression, cell survival signaling pathways & their cross-communication and (c) regulating immune responses. The information discussed in the present review will be useful in the drug discovery process as well as the treatment and prevention of colorectal cancer in humans.
Keywords: Colorectal cancer, curcumin, anti-cancer, apoptosis, angiogenesis, Wnt signaling pathway.
[http://dx.doi.org/10.1615/CritRevOncog.2019031335]
[http://dx.doi.org/10.3322/caac.21551] [PMID: 30620402]
[http://dx.doi.org/10.1136/gutjnl-2015-310912] [PMID: 26818619]
[http://dx.doi.org/10.1016/j.gene.2016.10.032] [PMID: 27777109]
[PMID: 28748833]
[http://dx.doi.org/10.1016/j.compbiolchem.2019.01.010] [PMID: 30708140]
[http://dx.doi.org/10.1016/j.jmgm.2019.01.018] [PMID: 30743158]
[http://dx.doi.org/10.1038/nrgastro.2011.149] [PMID: 21894197]
[http://dx.doi.org/10.1016/S0140-6736(13)61649-9] [PMID: 24225001]
[http://dx.doi.org/10.1371/journal.pone.0074616] [PMID: 24069323]
[http://dx.doi.org/10.1016/S0016-5085(03)00376-7] [PMID: 12761718]
[http://dx.doi.org/10.1016/j.soncn.2018.12.011] [PMID: 30665732]
[http://dx.doi.org/10.1038/ng.2503] [PMID: 23263490]
[http://dx.doi.org/10.1038/nrdp.2015.65] [PMID: 27189416]
[http://dx.doi.org/10.1309/87BD0C6UCGUG236J] [PMID: 16483003]
[http://dx.doi.org/10.1111/his.12055] [PMID: 23339363]
[http://dx.doi.org/10.1016/0092-8674(92)90526-I] [PMID: 1377983]
[http://dx.doi.org/10.1038/onc.2009.449] [PMID: 19966864]
[http://dx.doi.org/10.1177/0192623313505155] [PMID: 24178577]
[http://dx.doi.org/10.1007/s10620-014-3444-4] [PMID: 25492499]
[http://dx.doi.org/10.18632/oncotarget.23607] [PMID: 30279970]
[http://dx.doi.org/10.5772/63382]
[http://dx.doi.org/10.3748/wjg.v21.i17.5158] [PMID: 25954089]
[PMID: 10387965]
[http://dx.doi.org/10.1200/JCO.2008.20.6235]
[http://dx.doi.org/10.3748/wjg.v21.i31.9262] [PMID: 26309353]
[http://dx.doi.org/10.1111/j.1541-4337.2008.00044.x]
[http://dx.doi.org/10.1007/s11010-009-0269-0] [PMID: 19826768]
[http://dx.doi.org/10.1080/07391102.2019.1567391] [PMID: 30633652]
[http://dx.doi.org/10.1002/jcb.28316] [PMID: 30565717]
[http://dx.doi.org/10.1016/j.bcp.2007.08.016] [PMID: 17900536]
[http://dx.doi.org/10.1007/978-0-387-46401-5]
[http://dx.doi.org/10.1177/107424849700200409] [PMID: 10684472]
[http://dx.doi.org/10.2741/s95] [PMID: 20036978]
[http://dx.doi.org/10.2147/JEP.S70568] [PMID: 28435333]
[http://dx.doi.org/10.1517/13543784.13.10.1327] [PMID: 15461561]
[http://dx.doi.org/10.1208/s12248-012-9432-8] [PMID: 23143785]
[http://dx.doi.org/10.1146/annurev-food-032818-121738] [PMID: 30633561]
[http://dx.doi.org/10.1093/advances/nmx011] [PMID: 29438458]
[http://dx.doi.org/10.1080/19490976.2017.1361093] [PMID: 28767339]
[http://dx.doi.org/10.1080/03670244.2017.1337570] [PMID: 28891681]
[http://dx.doi.org/10.1080/19490976.2018.1445955] [PMID: 29509057]
[http://dx.doi.org/10.1080/19490976.2017.1421885] [PMID: 29437527]
[http://dx.doi.org/10.1080/19490976.2018.1448354] [PMID: 29533126]
[http://dx.doi.org/10.1096/fasebj.31.1_supplement.646.12.]
[http://dx.doi.org/10.3109/09637486.2015.1095865] [PMID: 26471074]
[http://dx.doi.org/10.1021/acs.jafc.7b00943] [PMID: 28401758]
[http://dx.doi.org/10.1186/1747-1028-3-14] [PMID: 18834508]
[http://dx.doi.org/10.1074/jbc.M410670200] [PMID: 15738001]
[http://dx.doi.org/10.1179/102453308X343437] [PMID: 19055861]
[http://dx.doi.org/10.2174/1381612820666140826154601] [PMID: 25341940]
[PMID: 17201156]
[http://dx.doi.org/10.1021/jf001129v] [PMID: 11312881]
[http://dx.doi.org/10.1016/j.exphem.2005.12.015] [PMID: 16569593]
[PMID: 18570726]
[http://dx.doi.org/10.1080/01635580802416703] [PMID: 19003584]
[http://dx.doi.org/10.1016/S0014-5793(02)02292-5] [PMID: 11852106]
[http://dx.doi.org/10.1016/j.canlet.2008.06.031] [PMID: 18701210]
[PMID: 19181006]
[PMID: 12050094]
[PMID: 10625938]
[http://dx.doi.org/10.1093/carcin/bgm123] [PMID: 17522064]
[http://dx.doi.org/10.1093/carcin/bgh239] [PMID: 15271854]
[http://dx.doi.org/10.1111/jfbc.12802] [PMID: 31353575]
[http://dx.doi.org/10.7150/jca.15690] [PMID: 27390600]
[http://dx.doi.org/10.1016/j.ijrobp.2009.06.034] [PMID: 19735878]
[http://dx.doi.org/10.1159/000098655] [PMID: 17228149]
[http://dx.doi.org/10.3109/03639041003695139] [PMID: 20545506]
[http://dx.doi.org/10.3109/03639045.2015.1064941] [PMID: 26165247]
[http://dx.doi.org/10.1158/1940-6207.CAPR-10-0098] [PMID: 21372035]
[PMID: 15668484]
[PMID: 20332435]
[http://dx.doi.org/10.1590/S0100-879X2005001200007] [PMID: 16302093]
[http://dx.doi.org/10.2147/OTT.S199601] [PMID: 31190888]
[http://dx.doi.org/10.1016/j.canlet.2015.05.005] [PMID: 25979230]
[http://dx.doi.org/10.1097/MIB.0000000000000522] [PMID: 26218141]
[PMID: 11712783]
[PMID: 11448902]
[http://dx.doi.org/10.21037/dmr.2018.09.01] [PMID: 30381803]
[http://dx.doi.org/10.2174/1568009053202081] [PMID: 15810876]
[http://dx.doi.org/10.3892/etm.2015.2749] [PMID: 26640527]
[http://dx.doi.org/10.1038/sj.onc.1204997] [PMID: 11753638]
[http://dx.doi.org/10.1006/excr.2001.5381] [PMID: 11716543]
[http://dx.doi.org/10.1556/IMAS.6.2014.4.1] [PMID: 25598986]
[http://dx.doi.org/10.1093/ecam/nem043] [PMID: 17549234]
[http://dx.doi.org/10.15430/JCP.2013.18.2.186] [PMID: 25337545]
[PMID: 25667441]
[http://dx.doi.org/10.1146/annurev.pharmtox.48.113006.094731] [PMID: 17883328]
[http://dx.doi.org/10.1074/jbc.M409024200] [PMID: 15383533]
[http://dx.doi.org/10.1007/s12263-011-0222-1] [PMID: 21516481]
[http://dx.doi.org/10.1371/journal.pone.0059003] [PMID: 23520547]
[http://dx.doi.org/10.1371/journal.pone.0055934] [PMID: 23457487]
[http://dx.doi.org/10.4251/wjgo.v2.i4.169] [PMID: 21160593]
[http://dx.doi.org/10.1007/s12307-011-0070-y] [PMID: 21710272]
[http://dx.doi.org/10.1016/j.cgh.2006.03.020] [PMID: 16757216]
[http://dx.doi.org/10.1016/j.jtcme.2016.08.002] [PMID: 28725630]