Abstract
Background: Vitamin C (VC) is believed to enhance immunity and is regularly integrated as a supplementary agent during several treatments.
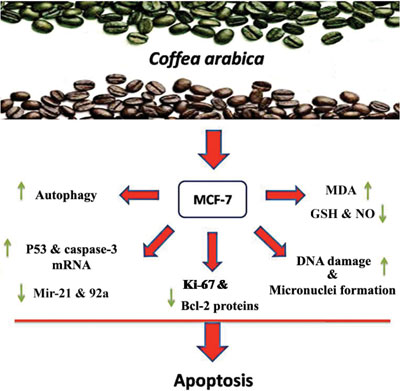
Objective: The green (GC) and roasted (RC) coffee (Coffea arabica) aqueous extracts (0, 125, 250 and 500 μg/ml) combined with VC (50 μg/ml) were examined on the cancerous MCF-7 cell line and normal human lymphocytes. Methods: Neutral red uptake assay, comet assay, immunocytochemical reactivity for protein expression and mRNA expression of apoptosis-related genes were performed. Results: A significant (P< 0.05) concentration-dependent increase of apoptotic features, such as morphological changes, and abundant nuclear condensation, altered the expression of p53 and caspase-3 mRNA, down-regulation of Bcl-2 protein as well as the acidic autophagosomal vacuolization in treated cells. The oxidative stress and DNA single-strand breaks were noticed too. Conclusion: These results suggest that coffee in combination with VC undergoes apoptotic anticancer pathway. This supports the integration of coffee and VC as a valuable candidate for anticancer research and treatments.Keywords: Coffea arabica, vitamin C, MCF-7, anticancer, apoptosis, protein expression.
[1]
Chow, L.W.C.; Loo, W.T.Y. The differential effects of cyclophosphamide, epirubicin and 5-fluorouracil on apoptotic marker (CPP-32), pro-apoptotic protein (p21(WAF-1)) and anti-apoptotic protein (bcl-2) in breast cancer cells. Breast Cancer Res. Treat., 2003, 80(3), 239-244.
[http://dx.doi.org/10.1023/A:1024995202135] [PMID: 14503796]
[http://dx.doi.org/10.1023/A:1024995202135] [PMID: 14503796]
[2]
Joseph, P. Eder & Arthur, T. Skarin, 4th ed; Systemic and Mucocutaneous Reactions to Chemotherapy. Atlas of Diagnostic Oncology, 2010, pp. 721-736.
[3]
Mates, J.; Segura, J.; Alonso, F.; Marquez, J. Natural antioxidants: Therapeutic prospects for cancer and neurological diseases. Mini Rev. Med. Chem., 2009, 9(10), 1202-1214.
[http://dx.doi.org/10.2174/138955709789055180]
[http://dx.doi.org/10.2174/138955709789055180]
[4]
Atta, A.H.; Mouneir, S.M. Antidiarrhoeal activity of some Egyptian medicinal plant extracts. J. Ethnopharmacol., 2004, 92(2-3), 303-309.
[http://dx.doi.org/10.1016/j.jep.2004.03.017] [PMID: 15138016]
[http://dx.doi.org/10.1016/j.jep.2004.03.017] [PMID: 15138016]
[5]
Hassab El Nabi, S.E.; El-Garawani, I.M.; Salman, A.M.; Ouda, R.I. The possible antigenotoxic potential of ginger oil on etoposide-treated albino rats. Saudi J. Med. Pharmaceut. Sci., 2017, 3(7A), 693-703.
[6]
Silvarolla, M.B.; Mazzafera, P.; Fazuoli, L.C. Plant biochemistry: A naturally decaffeinated arabica coffee. Nature, 2004, 429(6994), 826.
[http://dx.doi.org/10.1038/429826a] [PMID: 15215853]
[http://dx.doi.org/10.1038/429826a] [PMID: 15215853]
[7]
Gruenwald, J.; Brendler, T.; Jaenicke, C. PDR for herbal medicines.HerbalGram; Dennis V.C, Awang, Ed.; American Botanical Council: Austin, 2003, Vol. 58, pp. 75-76.
[8]
Orey, C. The healing powers of vinegar: A complete guide to nature’s most remarkable Remedy; Kensington Publishing Corp., 2009.
[10]
Shang, Y-F.; Xu, J-L.; Lee, W-J.; Um, B-H. Antioxidativepolyphenolics obtained from spent coffee grounds by pressurized liquid extraction. S. Afr. J. Bot., 2017, 109, 75-80.
[http://dx.doi.org/10.1016/j.sajb.2016.12.011]
[http://dx.doi.org/10.1016/j.sajb.2016.12.011]
[11]
Nicoli, M.C.; Anese, M.; Manzocco, L.; Lerici, C.R. Antioxidant properties of coffee brews in relation to the roasting degree. LWTFood Sci. and Technol., 1997, 30(3), 292-297.
[http://dx.doi.org/10.1006/fstl.1996.0181]
[http://dx.doi.org/10.1006/fstl.1996.0181]
[12]
El-Garawani, I.; El Nabi, S.H.; Nafie, E.; Almeldin, S. Foeniculum vulgare and Pelargonium graveolens essential oil mixture trigger the cell cycle arrest and apoptosis in MCF-7 cells. Anticancer. Agents Med. Chem., 2019.
[http://dx.doi.org/10.2174/1573399815666190326115116] [PMID: 30914034]
[http://dx.doi.org/10.2174/1573399815666190326115116] [PMID: 30914034]
[13]
Tohamy, A.A.; El-Garawani, I.M.; Ibrahim, S.R.; Moneim, A.E. The apoptotic properties of Salvia aegyptiaca and Trigonella foenum-graecum extracts on Ehrlich ascites carcinoma cells: The effectiveness of combined treatment. Res. J. Pharm. Biol. Chem. Sci., 2016, 7(3), 1872-1883.
[14]
Durak, A.; Gawlik-Dziki, U.; Kowalska, I. Evaluation of interactions between coffee and cardamom, their type, and strength in relation to interactions in a model system. CYTA J. Food, 2017, 15(2), 266-276.
[http://dx.doi.org/10.1080/19476337.2016.1247298]
[http://dx.doi.org/10.1080/19476337.2016.1247298]
[15]
Chou, T.M.; Benowitz, N.L. Caffeine and coffee: Effects on health and cardiovascular disease. Comp. Biochem. Physiol. C Pharmacol. Toxicol. Endocrinol., 1994, 109(2), 173-189.
[http://dx.doi.org/10.1016/0742-8413(94)00048-F] [PMID: 7881818]
[http://dx.doi.org/10.1016/0742-8413(94)00048-F] [PMID: 7881818]
[16]
Higdon, J.V.; Frei, B. Coffee and health: A review of recent human research. Crit. Rev. Food Sci. Nutr., 2006, 46(2), 101-123.
[http://dx.doi.org/10.1080/10408390500400009] [PMID: 16507475]
[http://dx.doi.org/10.1080/10408390500400009] [PMID: 16507475]
[17]
Leung, W.W.; Ho, S.C.; Chan, H.L.Y.; Wong, V.; Yeo, W.; Mok, T.S.K. Moderate coffee consumption reduces the risk of hepatocellular carcinoma in hepatitis B chronic carriers: A case-control study. J. Epidemiol. Community Health, 2011, 65(6), 556-558.
[http://dx.doi.org/10.1136/jech.2009.104125]
[http://dx.doi.org/10.1136/jech.2009.104125]
[18]
Kozuma, K.; Tsuchiya, S.; Kohori, J.; Hase, T.; Tokimitsu, I. Antihypertensive effect of green coffee bean extract on mildly hypertensive subjects. Hypertens. Res., 2005, 28, 711.
[http://dx.doi.org/10.1291/hypres.28.711]
[http://dx.doi.org/10.1291/hypres.28.711]
[19]
Suzuki, A.; Kagawa, D.; Ochiai, R.; Tokimitsu, I.; Saito, I. Green coffee bean extract and its metabolites have a hypotensive effect in spontaneously hypertensive rats. Hypertens. Res., 2002, 25(1), 99-107.
[http://dx.doi.org/10.1291/hypres.25.99] [PMID: 11924733]
[http://dx.doi.org/10.1291/hypres.25.99] [PMID: 11924733]
[20]
Ochiai, R.; Jokura, H.; Suzuki, A.; Tokimitsu, I.; Ohishi, M.; Komai, N. Ogihara, T. Green coffee bean extract improves human vasoreactivity. Hypertens. Res., 2004, 27(10), 731-737.
[http://dx.doi.org/10.1291/hypres.27.731] [PMID: 15785008]
[http://dx.doi.org/10.1291/hypres.27.731] [PMID: 15785008]
[21]
Dellalibera, O.; Lemaire, B.; Lafay, S. Svetol, green coffee extract, induces weight loss and increases the lean to fat mass ratio in volunteers with overweight problem. Phytotherapie, 2006, 4(4), 194-197.
[http://dx.doi.org/10.1007/s10298-006-0181-7]
[http://dx.doi.org/10.1007/s10298-006-0181-7]
[22]
Shimoda, H.; Seki, E.; Aitani, M. Inhibitory effect of green coffee bean extract on fat accumulation and body weight gain in mice. BMC Complement. Altern. Med., 2006, 6, 9.
[http://dx.doi.org/10.1186/1472-6882-6-9] [PMID: 16545124]
[http://dx.doi.org/10.1186/1472-6882-6-9] [PMID: 16545124]
[23]
Blum, J.; Lemaire, B.; Lafay, S. Effect of a green decaffeinated coffee extract on glycaemia. Nutra. Foods Res., 2007, 6, 13-17.
[25]
Pohl, P.; Stelmach, E.; Welna, M.; Szymczycha-Madeja, A. Determination of the elemental composition of coffee using instrumental methods. Food Anal. Methods, 2013, 6(2), 598-613.
[http://dx.doi.org/10.1007/s12161-012-9467-6]
[http://dx.doi.org/10.1007/s12161-012-9467-6]
[26]
Rodrigues, N.P.; Bragagnolo, N. Identification and quantification of bioactive compounds in coffee brews by HPLC-DAD-MSn. J. Food Compos. Anal., 2013, 32(2), 105-115.
[http://dx.doi.org/10.1016/j.jfca.2013.09.002]
[http://dx.doi.org/10.1016/j.jfca.2013.09.002]
[27]
Coimbra, M.A.; Simões, J.; Madureira, P.; Domingues, M.R.; Vilanova, M.; Nunes, F.M. Immunostimulatory properties of coffee mannans. In: 22nd International Conference on Coffee Science, ASIC 2008, Campinas, SP, Brazil, 14-19 September, 2008, Paris Association ScientifiqueInternationale du Café, 2009
[28]
Sun, M.; Estrov, Z.; Ji, Y.; Coombes, K.R.; Harris, D.H.; Kurzrock, R. Curcumin (diferuloylmethane) alters the expression profiles of microRNAs in human pancreatic cancer cells. Mol. Cancer Ther., 2008, 7(3), 464-473.
[http://dx.doi.org/10.1158/1535-7163.MCT-07-2272] [PMID: 18347134]
[http://dx.doi.org/10.1158/1535-7163.MCT-07-2272] [PMID: 18347134]
[29]
Rao, S.; Nadumane, V. K. Evaluation of the anticancer potential of
coffee beans: An in vitro study, 2016
http://nopr.niscair.res.in/handle/123456789/33978
[30]
Poole, R.; Kennedy, O.J.; Roderick, P.; Fallowfield, J.A.; Hayes, P.C.; Parkes, J. Coffee consumption and health: Umbrella review of meta-analyses of multiple health outcomes. BMJ, 2017, 359, j5024.
[http://dx.doi.org/10.1136/bmj.j5024]
[http://dx.doi.org/10.1136/bmj.j5024]
[31]
Stelmach, E.; Pohl, P.; Szymczycha-Madeja, A. The content of Ca, Cu, Fe, Mg and Mn and antioxidant activity of green coffee brews. Food Chem., 2015, 182, 302-308.
[http://dx.doi.org/10.1016/j.foodchem.2015.02.105] [PMID: 25842341]
[http://dx.doi.org/10.1016/j.foodchem.2015.02.105] [PMID: 25842341]
[32]
Farah, A.; Donangelo, C.M. Phenolic compounds in coffee. Braz. J. Plant Physiol., 2006, 18(1)
[http://dx.doi.org/10.1590/S1677-04202006000100003]
[http://dx.doi.org/10.1590/S1677-04202006000100003]
[33]
Wright, G.A.; Baker, D.D.; Palmer, M.J.; Stabler, D.; Mustard, J.A.; Power, E.F.; Borland, A.M.; Stevenson, P.C. Caffeine in floral nectar enhances a pollinator’s memory of reward. Science, 2013, 339(6124), 1202-1204.
[http://dx.doi.org/10.1126/science.1228806] [PMID: 23471406]
[http://dx.doi.org/10.1126/science.1228806] [PMID: 23471406]
[34]
Namba, T.; Matsuse, T. A historical study of coffee in Japanese and Asian countries: Focusing the medicinal uses in Asian traditional medicines. Yakushigaku Zasshi, 2002, 37(1), 65-75.
[PMID: 12412599]
[PMID: 12412599]
[35]
Moeenfard, M.; Rocha, L.; Alves, A. Quantification of caffeoylquinic acids in coffee brews by HPLC-DAD. J. Anal. Methods Chem., 2014, 2014 965353
[http://dx.doi.org/10.1155/2014/965353] [PMID: 25587489]
[http://dx.doi.org/10.1155/2014/965353] [PMID: 25587489]
[36]
El-Nabi, S.H.; Dawoud, G.T.M.; El-Garawani, I.; El-Shafey, S. HPLC analysis of phenolic acids, antioxidant activity and in vitro effectiveness of green and roasted Caffea arabica bean extracts: A comparative study. Anticancer. Agents Med. Chem., 2018, 18(9), 1281-1288.
[http://dx.doi.org/10.2174/1871520618666180124121927] [PMID: 29366428]
[http://dx.doi.org/10.2174/1871520618666180124121927] [PMID: 29366428]
[37]
Filomeni, G.; Graziani, I.; Rotilio, G.; Ciriolo, M.R. trans-Resveratrol induces apoptosis in human breast cancer cells MCF-7 by the activation of MAP kinases pathways. Genes Nutr., 2007, 2(3), 295-305.
[http://dx.doi.org/10.1007/s12263-007-0059-9] [PMID: 18850184]
[http://dx.doi.org/10.1007/s12263-007-0059-9] [PMID: 18850184]
[38]
Yoon, J-A.; Hahm, S-W.; Park, J-E.; Son, Y-S. Total polyphenol and flavonoid of fruit extract of Opuntia humifusa and its inhibitory effect on the growth of MCF-7 human breast cancer cells. J. Korean Soc. Food Sci. Nutr, 2009, 38(12), 1679-1684.
[http://dx.doi.org/10.3746/jkfn.2009.38.12.1679]
[http://dx.doi.org/10.3746/jkfn.2009.38.12.1679]
[39]
Wenzel, U.; Kuntz, S.; Brendel, M.D.; Daniel, H. Dietary flavone is a potent apoptosis inducer in human colon carcinoma cells. Cancer Res., 2000, 60(14), 3823-3831.
[PMID: 10919656]
[PMID: 10919656]
[40]
Subramani, T.; Yeap, S.K.; Ho, W.Y.; Ho, C.L.; Omar, A.R.; Aziz, S.A.; Rahman, N.M.; Alitheen, N.B. Vitamin C suppresses cell death in MCF-7 human breast cancer cells induced by tamoxifen. J. Cell. Mol. Med., 2014, 18(2), 305-313.
[http://dx.doi.org/10.1111/jcmm.12188] [PMID: 24266867]
[http://dx.doi.org/10.1111/jcmm.12188] [PMID: 24266867]
[41]
Ourique, F.; Kviecinski, M.R.; Felipe, K.B.; Correia, J.F.G.; Farias, M.S.; Castro, L.S.; Grinevicius, V.M.; Valderrama, J.; Rios, D.; Benites, J.; Calderon, P.B.; Pedrosa, R.C. DNA damage and inhibition of AKT pathway in MCF-7 cells and ehrlich tumor in mice treated with 1,4-naphthoquinones in combination with ascorbate. Oxid. Med. Cell. Longev., 2015, 2015 495305
[http://dx.doi.org/10.1155/2015/495305] [PMID: 25793019]
[http://dx.doi.org/10.1155/2015/495305] [PMID: 25793019]
[42]
Uetaki, M.; Tabata, S.; Nakasuka, F.; Soga, T.; Tomita, M. Metabolomic alterations in human cancer cells by vitamin C-induced oxidative stress. Sci. Rep., 2015, 5(1), 13896.
[http://dx.doi.org/10.1038/srep13896] [PMID: 26350063]
[http://dx.doi.org/10.1038/srep13896] [PMID: 26350063]
[43]
Fain, O.; Mathieu, E.; Thomas, M. Scurvy in patients with cancer. BMJ, 1998, 316(7145), 1661-1662.
[http://dx.doi.org/10.1136/bmj.316.7145.1661] [PMID: 9603754]
[http://dx.doi.org/10.1136/bmj.316.7145.1661] [PMID: 9603754]
[44]
Kurbacher, C.M.; Wagner, U.; Kolster, B.; Andreotti, P.E.; Krebs, D.; Bruckner, H.W. Ascorbic acid (vitamin C) improves the antineoplastic activity of doxorubicin, cisplatin, and paclitaxel in human breast carcinoma cells in vitro. Cancer Lett., 1996, 103(2), 183-189.
[http://dx.doi.org/10.1016/0304-3835(96)04212-7] [PMID: 8635156]
[http://dx.doi.org/10.1016/0304-3835(96)04212-7] [PMID: 8635156]
[45]
Prasad, K.N.; Hernandez, C.; Edwards‐Prasad, J.; Nelson, J.; Borus, T.; Robinson, W.A. Modification of the effect of tamoxifen, cis‐platin, DTIC, and interferon‐α2b onhuman melanoma cells in culture by a mixture of vitamins. Nutr. Cancer, 1994, 22(3), 233-245.
[http://dx.doi.org/10.1080/01635589409514349] [PMID: 7877893]
[http://dx.doi.org/10.1080/01635589409514349] [PMID: 7877893]
[46]
Taper, H.S.; De Gerlache, J.; Lans, M.; Roberfroid, M. Non‐toxic potentiation of cancer chemotherapy by combined C and K3 vitamin pre‐treatment. Int. J. Cancer, 1987, 40(4), 575-579.
[http://dx.doi.org/10.1002/ijc.2910400424] [PMID: 3666992]
[http://dx.doi.org/10.1002/ijc.2910400424] [PMID: 3666992]
[47]
Najafi, Z.; Sharifi, M.; Javadi, G. Degradation of miR-21 induces apoptosis and inhibits cell proliferation in human hepatocellular carcinoma. Cancer Gene Ther., 2015, 22(11), 530-535.
[http://dx.doi.org/10.1038/cgt.2015.51] [PMID: 26427512]
[http://dx.doi.org/10.1038/cgt.2015.51] [PMID: 26427512]
[48]
Breving, K.; Esquela-Kerscher, A. The complexities of microRNA regulation: Mirandering around the rules. Int. J. Biochem. Cell Biol., 2010, 42(8), 1316-1329.
[http://dx.doi.org/10.1016/j.biocel.2009.09.016] [PMID: 19800023]
[http://dx.doi.org/10.1016/j.biocel.2009.09.016] [PMID: 19800023]
[49]
Rufino-Palomares, E.E.; Reyes-Zurita, F.J.; Lupiáñez, J.A.; Medina, P.P. MicroRNAs as Oncogenes and Tumor Suppressors. MicroRNAs in Medicine; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013, pp. 223-243.
[http://dx.doi.org/10.1002/9781118300312.ch14]
[http://dx.doi.org/10.1002/9781118300312.ch14]
[50]
Rufino‐Palomares, E.E.; Reyes‐Zurita, F.J.; Lupiáñez, J.A.; Medina, P.P. MicroRNAs as Oncogenes and Tumor Suppressors In: MicroRNAs
in Medicine; Charles H. Lawrie, Ed.; John Wiley & Sons,
Inc.: 2003; Chapter. 14.
[51]
Zhang, B.; Pan, X.; Cobb, G.P.; Anderson, T.A. microRNAs as oncogenes and tumor suppressors. Dev. Biol., 2007, 302(1), 1-12.
[http://dx.doi.org/10.1016/j.ydbio.2006.08.028] [PMID: 16989803]
[http://dx.doi.org/10.1016/j.ydbio.2006.08.028] [PMID: 16989803]
[52]
Asangani, I.A.; Rasheed, S.A.K.; Nikolova, D.A.; Leupold, J.H.; Colburn, N.H.; Post, S.; Allgayer, H. Post, S.; Allgayer, H. MicroRNA-21 (miR-21) post-transcriptionally down regulates tumor suppressor Pdcd4 and stimulates invasion, intravasation and metastasis in colorectal cancer. Oncogene, 2008, 27(15), 2128-2136.
[http://dx.doi.org/www.nature.com/articles/1210856] [PMID: 17968323]
[http://dx.doi.org/www.nature.com/articles/1210856] [PMID: 17968323]
[53]
He, L.; Thomson, J.M.; Hemann, M.T.; Hernando-Monge, E.; Mu, D.; Goodson, S.; Powers, S.; Cordon-Cardo, C.; Lowe, S.W.; Hannon, G.J.; Hammond, S.M. A microRNA polycistron as a potential human oncogene. Nature, 2005, 435(7043), 828-833.
[http://dx.doi.org/10.1038/nature03552] [PMID: 15944707]
[http://dx.doi.org/10.1038/nature03552] [PMID: 15944707]
[54]
Frankel, L.B.; Christoffersen, N.R.; Jacobsen, A.; Lindow, M.; Krogh, A.; Lund, A.H. Programmed cell death 4 (PDCD4) is an important functional target of the microRNA miR-21 in breast cancer cells. J. Biol. Chem., 2008, 283(2), 1026-1033.
[http://dx.doi.org/10.1038/nature03552] [PMID: 17991735]
[http://dx.doi.org/10.1038/nature03552] [PMID: 17991735]
[55]
El-Garawani, I.M. Ameliorative effect of Cymbopogon citratus extract on cisplatin-induced genotoxicity in human leukocytes. J. Biosci. Appl. Res., 2015, 1(6), 304-310.
[http://dx.doi.org/10.1038/nature03552] [PMID: 17991735]
[http://dx.doi.org/10.1038/nature03552] [PMID: 17991735]
[56]
Moore, G.E.; Gerner, R.E.; Franklin, H.A. Culture of normal human leukocytes. JAMA, 1967, 199, 519-524.
[57]
Elkhateeb, W.A.; Zaghlol, G.M.; El-Garawani, I.M.; Ahmed, E.F.; Rateb, M.E.; Abdel Moneim, A.E. Ganodermaap planatum secondary metabolites induced apoptosis through different pathways: In vivo and in vitro anticancer studies. Biomed. Pharmacother., 2018, 101, 264-277.
[http://dx.doi.org/10.1016/j.biopha.2018.02.058] [PMID: 29494964]
[http://dx.doi.org/10.1016/j.biopha.2018.02.058] [PMID: 29494964]
[58]
Thippeswamy, G.; Salimath, B.P. Induction of caspase-3 activated DNase mediated apoptosis by hexane fraction of Tinospora cordifolia in EAT cells. Environ. Toxicol. Pharmacol., 2007, 23(2), 212-220.
[http://dx.doi.org/10.1016/j.etap.2006.10.004] [PMID: 21783760]
[http://dx.doi.org/10.1016/j.etap.2006.10.004] [PMID: 21783760]
[59]
Liu, K.; Liu, P.C.; Liu, R.; Wu, X. Dual AO/EB staining to detect apoptosis in osteosarcoma cells compared with flow cytometry. Med. Sci. Monit. Basic Res., 2015, 21, 15-20.
[http://dx.doi.org/10.12659/MSMBR.893327] [PMID: 25664686]
[http://dx.doi.org/10.12659/MSMBR.893327] [PMID: 25664686]
[60]
Kirsch-Volders, M.; Sofuni, T.; Aardema, M.; Albertini, S.; Eastmond, D.; Fenech, M. Report from the in vitro micronucleus assay working group. Mutat. Res./Genet. Toxicol. Environ. Mutagen., 2003, 540(2), 69-77.
[61]
Tolbert, P.E.; Shy, C.M.; Allen, J.W. Micronuclei and other nuclear anomalies in buccal smears: Methods development. Mutat. Res., 1992, 271(1), 69-77.
[http://dx.doi.org/10.1016/0165-1161(92)90033-I]
[http://dx.doi.org/10.1016/0165-1161(92)90033-I]
[62]
Evans, H.J. Cytological methods for detecting chemical mutagens. Chem. Mutagens, 1976, 4, 1-29.
[http://dx.doi.org/link.springer.com/chapter/10.1007/978-1-4684-0892-8_1]
[http://dx.doi.org/link.springer.com/chapter/10.1007/978-1-4684-0892-8_1]
[63]
Singh, N.P.; McCoy, M.T.; Tice, R.R.; Schneider, E.L. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp. Cell Res., 1988, 175(1), 184-191.https://doi.org/https://doi.org/10.1016/0014-4827(88)90265-0
[http://dx.doi.org/10.1016/0014-4827(88)90265-0] [PMID: 3345800]
[http://dx.doi.org/10.1016/0014-4827(88)90265-0] [PMID: 3345800]
[64]
Hsu, S-M.; Raine, L.; Fanger, H.X. Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: A comparison between ABC and unlabeled antibody (PAP) procedures. J. Histochem. Cytochem., 1981, 29(4), 577-580.
[http://dx.doi.org/10.1177/29.4.6166661] [PMID: 6166661]
[http://dx.doi.org/10.1177/29.4.6166661] [PMID: 6166661]
[65]
Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods, 2001, 25(4), 402-408.
[http://dx.doi.org/www.ncbi.nlm.nih.gov/pubmed/11846609] [PMID: 11846609]
[http://dx.doi.org/www.ncbi.nlm.nih.gov/pubmed/11846609] [PMID: 11846609]
[66]
Paglin, S.; Hollister, T.; Delohery, T.; Hackett, N.; McMahill, M.; Sphicas, E. Novel response of cancer cells to radiation involves autophagy and formation of acidic vesicles. Cancer Res., 2001, 61(2), 439-444.
[PMID: 11212227]
[PMID: 11212227]
[67]
Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem., 1951, 193(1), 265-275.
[PMID: 14907713]
[PMID: 14907713]
[68]
Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem., 1979, 95(2), 351-358.
[http://dx.doi.org/10.1016/0003-2697(79)90738-3] [PMID: 36810]
[http://dx.doi.org/10.1016/0003-2697(79)90738-3] [PMID: 36810]
[69]
Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal. Biochem., 1982, 126(1), 131-138.
[http://dx.doi.org/10.1016/0003-2697(82)90118-X] [PMID: 7181105]
[http://dx.doi.org/10.1016/0003-2697(82)90118-X] [PMID: 7181105]
[70]
Ellman, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys., 1959, 82(1), 70-77.
[http://dx.doi.org/10.1016/0003-9861(59)90090-6] [PMID: 13650640]
[http://dx.doi.org/10.1016/0003-9861(59)90090-6] [PMID: 13650640]
[71]
Akbas, H.S.; Timur, M.; Ozben, T. Concurrent use of antioxidants in cancer therapy: An update. Expert Rev. Clin. Immunol., 2006, 2(6), 931-939.
[http://dx.doi.org/10.1586/1744666X.2.6.931] [PMID: 20476980]
[http://dx.doi.org/10.1586/1744666X.2.6.931] [PMID: 20476980]
[72]
El-Garawani, I.M.; Elkhateeb, W.A.; Zaghlol, G.M.; Almeer, R.S.; Ahmed, E.F.; Rateb, M.E.; Abdel Moneim, A.E. Candelariella vitellina extract triggers in vitro and in vivo cell death through induction of apoptosis: A novel anticancer agent. Food Chem. Toxicol., 2019, 127, 110-119.
[http://dx.doi.org/10.1016/j.fct.2019.03.003] [PMID: 30853555]
[http://dx.doi.org/10.1016/j.fct.2019.03.003] [PMID: 30853555]
[73]
Wierzejska, R. The antioxidant vitamin nutritional therapy and the chemotherapy treatment in oncology. Rev. Bras. Cancerol., 2001, 47(3), 303-308.
[74]
Búfalo, M.C.; Ferreira, I.; Costa, G.; Francisco, V.; Liberal, J.; Cruz, M.T. Propolis and its constituent caffeic acid suppress LPS-stimulated pro-inflammatory response by blocking NF-κB and MAPK activation in macrophages. J. Ethnopharmacol., 2013, 149(1), 84-92.
[http://dx.doi.org/10.1016/j.jep.2013.06.004]
[http://dx.doi.org/10.1016/j.jep.2013.06.004]
[75]
Sato, Y.; Itagaki, S.; Kurokawa, T.; Ogura, J.; Kobayashi, M.; Hirano, T.; Sugawara, M.; Iseki, K. In vitro and in vivo antioxidant properties of chlorogenic acid and caffeic acid. Int. J. Pharm., 2011, 403(1-2), 136-138.https://doi.org/https://doi.org/10.1016/j.ijpharm.2010.09.035
[http://dx.doi.org/10.1016/j.ijpharm.2010.09.035] [PMID: 20933071]
[http://dx.doi.org/10.1016/j.ijpharm.2010.09.035] [PMID: 20933071]
[76]
Patay, É.B.; Bencsik, T.; Papp, N. Phytochemical overview and medicinal importance of Coffea species from the past until now. Asian Pac. J. Trop. Med., 2016, 9(12), 1127-1135.
[http://dx.doi.org/10.1016/j.apjtm.2016.11.008]
[http://dx.doi.org/10.1016/j.apjtm.2016.11.008]
[77]
Alao, J.P.; Sunnerhagen, P. The ATM and ATR inhibitors CGK733 and caffeine suppress cyclin D1 levels and inhibit cell proliferation. Radiat. Oncol., 2009, 4(51)
[http://dx.doi.org/10.1186/1748-717X-4-51]
[http://dx.doi.org/10.1186/1748-717X-4-51]
[78]
Negrini, S.; Gorgoulis, V.G.; Halazonetis, T.D. Genomic instability — an evolving hallmark of cancer. Nat. Rev. Mol. Cell Biol., 2010, 11, 220.
[79]
Burgos-Morón, E. 1.; Calderón-Montaño, J.M.; Orta, M.L.; Pastor, N.; Pérez-Guerrero, C.; Austin, C.; Mateos, S.; López-Lázaro, M. The coffee constituent chlorogenic acid induces cellular DNA damage and formation of topoisomerase I- and II-DNA complexes in cells. J. Agric. Food Chem., 2012, 60(30), 7384-7391.
[http://dx.doi.org/10.1021/jf300999e] [PMID: 22793503]
[http://dx.doi.org/10.1021/jf300999e] [PMID: 22793503]
[80]
Wang, Y.; Schattenberg, J.M.; Rigoli, R.M.; Storz, P.; Czaja, M.J. Hepatocyte resistance to oxidative stress is dependent on protein kinase C-mediated down-regulation of c-Jun/AP-1. J. Biologic. Chem., 2004, 279(30), 31089-31097.
[81]
Green, D.R. Means to an End: Apoptosis and Other Cell Death Mechanisms; Cold Spring Harbor Laboratory Press, 2011; Cold Spring Harbor Laboratory Press, 2011.
[82]
Williams, J.L.; Nath, N.; Chen, J.; Hundley, T.R.; Gao, J.; Kopelovich, L. Growth inhibition of human colon cancer cells by nitric oxide (NO)-donating aspirin is associated with cyclooxygenase-2 induction and β-catenin/T-cell factor signaling, nuclear factor-κB, and NO synthase 2 inhibition: Implications for chemoprevention. Cancer Res., 2003, 63(22), 7613-7618.
[PMID: 14633677]
[PMID: 14633677]
[83]
Ignarro, L.J.; Buga, G.M.; Wei, L.H.; Bauer, P.M.; Wu, G.; del Soldato, P. Role of the arginine-nitric oxide pathway in the regulation of vascular smooth muscle cell proliferation. Proc. Natl. Acad. Sci. USA, 2001, 98(7), 4202-4208.
[http://dx.doi.org/10.1073/pnas.071054698] [PMID: 11259671]
[http://dx.doi.org/10.1073/pnas.071054698] [PMID: 11259671]
[84]
Lepoivre, M.; Fieschi, F.; Coves, J.; Thelander, L.; Fontecave, M. Inactivation of ribonucleotide reductase by nitric oxide. Biochem. Biophys. Res. Commun., 1991, 179(1), 442-448.
[http://dx.doi.org/10.1016/0006-291X(91)91390-X] [PMID: 1652957]
[http://dx.doi.org/10.1016/0006-291X(91)91390-X] [PMID: 1652957]
[85]
Drapier, J.C.; Hibbs, J.B. Murine cytotoxic activated macrophages inhibit aconitase in tumor cells. Inhibition involves the iron-sulfur prosthetic group and is reversible. J. Clin. Invest., 1986, 78(3), 790-797.
[http://dx.doi.org/10.1172/JCI112642]
[http://dx.doi.org/10.1172/JCI112642]
[86]
Watts, R.N.; Hawkins, C.; Ponka, P.; Richardson, D.R. Nitrogen Monoxide (NO)-mediated iron release from cells is linked to NO-induced glutathione efflux via multidrug resistance-associated protein 1. Proc. Natl. Acad. Sci. USA, 2006, 7670-7675.
[http://dx.doi.org/10.1073/pnas.0602515103] [PMID: 16679408]
[http://dx.doi.org/10.1073/pnas.0602515103] [PMID: 16679408]
[87]
Takemura, Y.; Satoh, M.; Satoh, K.; Hamada, H.; Sekido, Y.; Kubota, S. High dose of ascorbic acid induces cell death in mesothelioma cells. Biochem. Biophys. Res. Commun., 2010, 394, 249-253.
[http://dx.doi.org/10.1016/j.bbrc.2010.02.012] [PMID: 20171954]
[http://dx.doi.org/10.1016/j.bbrc.2010.02.012] [PMID: 20171954]
[88]
Lv, H.; Wang, C.; Fang, T.; Li, T.; Lv, G.; Han, Q.; Yang, W.; Wang, H. Vitamin C preferentially kills cancer stem cells in hepatocellular carcinoma via SVCT-2. NPJ Precis. Oncol., 2018, 2(1), 1.
[http://dx.doi.org/10.1038/s41698-017-0044-8] [PMID: 29872720]
[http://dx.doi.org/10.1038/s41698-017-0044-8] [PMID: 29872720]
[89]
Banerjee, M.; Singh, P.; Panda, D. Curcumin suppresses the dynamic instability of microtubules, activates the mitotic checkpoint and induces apoptosis in MCF-7 cells. FEBS J., 2010, 277(16), 3437-3448.
[http://dx.doi.org/10.1111/j.1742-4658.2010.07750.x] [PMID: 20646066]
[http://dx.doi.org/10.1111/j.1742-4658.2010.07750.x] [PMID: 20646066]
[90]
Amin, A.R.M.R.; Wang, D.; Zhang, H.; Peng, S.; Shin, H.J.C.; Brandes, J.C.; Tighiouart, M.; Khuri, F.R.; Chen, Z.G.; Shin, D.M. Enhanced anti-tumor activity by the combination of the natural compounds (-)-epigallocatechin-3-gallate and luteolin: Potential role of p53. J. Biol. Chem., 2010, 285(45), 34557-34565.
[http://dx.doi.org/10.1074/jbc.M110.141135] [PMID: 20826787]
[http://dx.doi.org/10.1074/jbc.M110.141135] [PMID: 20826787]
[91]
Joslyn, K. Control of mitochondrial apoptosis by the Bcl-2 family. J. Cell Sci., 2009, 122(4), 437-441.
[http://dx.doi.org/10.1242/jcs.031682]
[http://dx.doi.org/10.1242/jcs.031682]
[92]
Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell, 2018, 144(5), 646-674.
[http://dx.doi.org/10.1016/j.cell.2011.02.013] [PMID: 21376230]
[http://dx.doi.org/10.1016/j.cell.2011.02.013] [PMID: 21376230]
[93]
Motawi, T.K.; Abdelazim, S.A.; Darwish, H.A.; Elbaz, E.M.; Shouman, S.A. Modulation of tamoxifen cytotoxicity by caffeic acid phenethyl ester in MCF-7 breast cancer cells. Oxid. Med. Cell. Longev., 2016, 2016 3017108
[http://dx.doi.org/10.1155/2016/3017108] [PMID: 26697130]
[http://dx.doi.org/10.1155/2016/3017108] [PMID: 26697130]
[94]
Chobotova, K. Aging and cancer: Converging routes to disease prevention. Integr. Cancer Ther., 2009, 8(2), 115-122.
[http://dx.doi.org/10.1177/1534735409335505] [PMID: 19679619]
[http://dx.doi.org/10.1177/1534735409335505] [PMID: 19679619]
[95]
Shashni, B.; Sharma, K.; Singh, R.; Sakharkar, K.R.; Dhillon, S.K.; Nagasaki, Y.; Sakharkar, M.K. Coffee component Hydroxyl Hydroquinone (HHQ) as a putative ligand for PPAR gamma and implications in breast cancer. BMC Genomics, 2013, 14(5)(Suppl. 5), S6.
[http://dx.doi.org/10.1186/1471-2164-14-S5-S6] [PMID: 24564733]
[http://dx.doi.org/10.1186/1471-2164-14-S5-S6] [PMID: 24564733]
[96]
Takaoka, A.; Hayakawa, S.; Yanai, H.; Stoiber, D.; Negishi, H.; Kikuchi, H. Integration of interferon-α/β signalling to p53 responses in tumour suppression and antiviral defence. Nature, 2003, 424, 516.
[97]
Yang, H-L.; Chen, C-S.; Chang, W-H.; Lu, F-J.; Lai, Y-C.; Chen, C-C. Growth inhibition and induction of apoptosis in MCF-7 breast cancer cells by Antrodia camphorata. Cancer Lett., 2006, 231(2), 215-227.
[http://dx.doi.org/10.1016/j.canlet.2005.02.004]
[http://dx.doi.org/10.1016/j.canlet.2005.02.004]
[98]
Wang, S.; He, M.; Li, L.; Liang, Z.; Zou, Z.; Tao, A. Cell-in-cell death is not restricted by caspase-3 deficiency in MCF-7 Cells. J. Breast Cancer, 2016, 19(3), 231-241.
[http://dx.doi.org/10.4048/jbc.2016.19.3.231] [PMID: 27721872]
[http://dx.doi.org/10.4048/jbc.2016.19.3.231] [PMID: 27721872]
[99]
Lui, W-O.; Pourmand, N.; Patterson, B.K.; Fire, A. Patterns of known and novel small RNAs in human cervical cancer. Cancer Res., 2007, 67(13), 6031-6043.
[http://dx.doi.org/10.1158/0008-5472.CAN-06-0561]
[http://dx.doi.org/10.1158/0008-5472.CAN-06-0561]
[100]
Park, J-K.; Lee, E.J.; Esau, C.; Schmittgen, T.D. Antisense inhibition of microRNA-21 or-221 arrests cell cycle, induces apoptosis, and sensitizes the effects of gemcitabine in pancreatic adenocarcinoma. Pancreas, 2009, 38(7), e190-e199.
[http://dx.doi.org/10.1097/MPA.0b013e3181ba82e1]
[http://dx.doi.org/10.1097/MPA.0b013e3181ba82e1]
[101]
Si, M-L.; Zhu, S.; Wu, H.; Lu, Z.; Wu, F.; Mo, Y-Y. miR-21-mediated tumor growth. Oncogene, 2007, 26(19), 2799-2280.
[http://dx.doi.org/10.1038/sj.onc.1210083]
[http://dx.doi.org/10.1038/sj.onc.1210083]
[102]
Northcott, P.A.; Fernandez-L, A.; Hagan, J.P.; Ellison, D.W.; Grajkowska, W.; Gillespie, Y.; Taylor, M.D. The miR-17/92 Polycistron is up-regulated in sonic hedgehog-driven medulloblastomas and induced by N-myc in sonic hedgehog-treated cerebellar neural precursors. Cancer Res., 2009, 69(8), 3249-3255.
[http://dx.doi.org/10.1158/0008-5472.CAN-08-4710]
[http://dx.doi.org/10.1158/0008-5472.CAN-08-4710]
[103]
Fix, L.N.; Shah, M.; Efferth, T.; Farwell, M.A.; Zhang, B. MicroRNA expression profile of MCF-7 human breast cancer cells and the effect of green tea polyphenon-60. Cancer Genomics Proteomics, 2010, 7(5), 261-277.
[http://dx.doi.org/www.ncbi.nlm.nih.gov/pubmed/20952761] [PMID: 20952761]
[http://dx.doi.org/www.ncbi.nlm.nih.gov/pubmed/20952761] [PMID: 20952761]
[104]
Janku, F.; McConkey, D.J.; Hong, D.S.; Kurzrock, R. Autophagy as a target for anticancer therapy. Nat. Rev. Clin. Oncol., 2011, 8(9), 528-539.
[http://dx.doi.org/10.1038/nrclinonc.2011.71] [PMID: 21587219]
[http://dx.doi.org/10.1038/nrclinonc.2011.71] [PMID: 21587219]
[105]
Shen, H-M.; Codogno, P. Autophagic cell death: Loch Ness monster or endangered species? Autophagy, 2011, 7(5), 457-465.
[http://dx.doi.org/10.4161/auto.7.5.14226]
[http://dx.doi.org/10.4161/auto.7.5.14226]
[106]
Maiuri, M.C.; Galluzzi, L.; Morselli, E.; Kepp, O.; Malik, S.A.; Kroemer, G. Autophagy regulation by p53. Curr. Opin. Cell Biol., 2010, 22(2), 181-185.https://doi.org/https://doi.org/10.1016/j.ceb.2009.12.001
[http://dx.doi.org/10.1016/j.ceb.2009.12.001] [PMID: 20044243]
[http://dx.doi.org/10.1016/j.ceb.2009.12.001] [PMID: 20044243]
[107]
Hečimović, I.; Belščak-Cvitanović, A.; Horžić, D.; Komes, D. Comparative study of polyphenols and caffeine in different coffee varieties affected by the degree of roasting. Food Chem., 2011, 129(3), 991-1000.https://doi.org/https://doi.org/10.1016/j.foodchem.2011.05.059
[http://dx.doi.org/10.1016/j.foodchem.2011.05.059] [PMID: 25212328]
[http://dx.doi.org/10.1016/j.foodchem.2011.05.059] [PMID: 25212328]
[108]
Wei, F.; Furihata, K.; Koda, M.; Hu, F.; Miyakawa, T.; Tanokura, M. Roasting process of coffee beans as studied by nuclear magnetic resonance: Time course of changes in composition. J. Agric. Food Chem., 2012, 60(4), 1005-1012.
[http://dx.doi.org/10.1021/jf205315r] [PMID: 22224944]
[http://dx.doi.org/10.1021/jf205315r] [PMID: 22224944]
[109]
Sakr, S.A.; Hassab, S.E.; Nabi, E.; Okdah, Y.A.; El-Garawani, I.M.; El-Shabka, A.M. Cytoprotective effects of aqueous ginger (Zingiber officinale) extract against carbimazole-induced toxicity in albino rats. Eur. J. Pharm. Med. Res., 2016, 3(7), 483-497.
[110]
El-Garawani, I.; Hassab, S.; Nabi, E.; El-Ghandour, E. The protective effect of (Foeniculum vulgare) oil on etoposide-induced genotoxicity on male albino rats. Eur. J. Pharm. Med. Res., 2017, 4(7), 180-194.
[111]
Selvendiran, K.; Ahmed, S.; Dayton, A.; Kuppusamy, M.L.; Tazi, M.; Bratasz, A.; Tong, L.; Rivera, B.K.; Kálai, T.; Hideg, K.; Kuppusamy, P. Safe and targeted anticancer efficacy of a novel class of antioxidant-conjugated difluorodiarylidenylpiperidones: Differential cytotoxicity in healthy and cancer cells. Free Radic. Biol. Med., 2010, 48(9), 1228-1235.
[http://dx.doi.org/10.1016/j.freeradbiomed.2010.02.009] [PMID: 20156552]
[http://dx.doi.org/10.1016/j.freeradbiomed.2010.02.009] [PMID: 20156552]