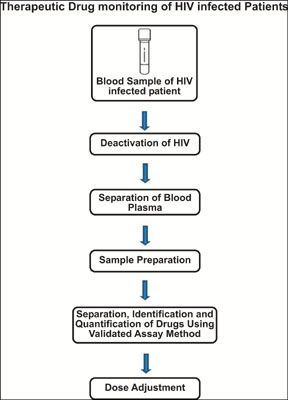
[1]
Kang JS, Lee MH. Overview of therapeutic drug monitoring. Korean J Intern Med 2009; 24: 1-10.
[2]
Mocroft A, Ledergerber B, Katlama C, et al. Decline in the AIDS and death rates in the EuroSIDA study: An observational study. Lancet 2003; 362: 22-9.
[3]
Arts EJ, Hazuda DJ. HIV-1 antiretroviral drug therapy. Cold Spring Harb Perspect Med 2012; 2: 1-23.
[4]
Gallant JE. Initial therapy of HIV infection. J Clin Virol 2002; 25: 317-33.
[5]
Arts EJ, Hazuda DJ. HIV-1 antiretroviral drug therapy. Cold Spring Harb Perspect Med 2012; 2: 1-23.
[6]
Bossi P, Peytavin G, Ait-Mohand H, et al. GENOPHAR: A randomized study of plasma drug measurements in association with genotypic resistance testing and expert advice to optimize therapy in patients failing antiretroviral therapy. HIV Med 2004; 5: 352-9.
[7]
Burger D, Hugen P, Reiss P, et al. Therapeutic drug monitoring of nelfinavir and indinavir in treatment-naive HIV-1-infected individuals. AIDS 2003; 17: 1157-65.
[8]
Clevenbergh P, Garraffo R, Durant J, Dellamonica P. PharmAdapt: A randomized prospective study to evaluate the benefit of therapeutic monitoring of protease inhibitors: 12 week results. AIDS 2002; 16: 2311-5.
[9]
Fletcher CV, Anderson PL, Kakuda TN, et al. Concentration-controlled compared with conventional antiretroviral therapy for HIV infection. AIDS 2002; 16: 551-60.
[10]
Fraaij PL, Rakhmanina N, Burger DM, de Groot R. Therapeutic drug monitoring in children with HIV/AIDS. Ther Drug Monit 2004; 26: 122-6.
[11]
Rakhmanina NY, van den Anker JN, Soldin SJ, van Schaik RH, Mordwinkin N, Neely MN. Can therapeutic drug monitoring improve pharmacotherapy of HIV infection in adolescents? Ther Drug Monit 2010; 32: 273-81.
[12]
Nettles RE, Kieffer TL, Parsons T, et al. Marked intraindividual variability in antiretroviral concentrations may limit the utility of therapeutic drug monitoring. Clin Infect Dis 2006; 42: 1189-96.
[13]
Brereton RG. Coupled Chromatography. In:Applied Chemometrics for Scientists. John Wiley and Sons, Ltd 2007; pp. 221-48.
[14]
Chan CP, Cheung YC, Renneberg R, Seydack M. New trends in immunoassays. Adv Biochem Eng Biotechnol 2007; 109: 123-54.
[15]
Abdissa A, Wiesner L, Mcilleron H, Friis H, Andersen AB, Kaestel P. Evaluation of an immunoassay for determination of plasma efavirenz concentrations in resource-limited settings. J Int AIDS Soc 2014; 17: 18979.
[16]
Findlay JW, Smith WC, Lee JW, et al. Validation of immunoassays for bioanalysis: A pharmaceutical industry perspective. J Pharm Biomed Anal 2000; 21: 1249-73.
[17]
Zheng W, He L. Multiplexed Immunoassays. In: Advances in Immunoassay Technology; Chiu NHL and Christopoulos T K, Ed. InTech: Croatia 2012; 143-64.
[18]
Hwang H, Chon H, Choo J, Park JK. Optoelectrofluidic sandwich immunoassays for detection of human tumor marker using surface-enhanced raman scattering. Anal Chem 2010; 82: 7603-10.
[19]
Dong MW. Modern HPLC for Practicing Scientists. 1st ed. John Wiley & Sons, Inc: New York 2006.
[20]
Eichhorst JC, Etter ML, Rousseaux N, Lehotay DC. Drugs of abuse testing by tandem mass spectrometry: A rapid, simple method to replace immunoassays. Clin Biochem 2009; 42: 1531-42.
[21]
Amad MH, Houk RS. High-resolution mass spectrometry with a multiple pass quadrupole mass analyzer. Anal Chem 1998; 70: 4885-9.
[22]
Douglas DJ, Frank AJ, Mao D. Linear ion traps in mass spectrometry. Mass Spectrom Rev 2005; 24: 1-29.
[23]
Grix R, Kutscher R, Li G, Grüner U, Wollnik H, Matsuda H. A time-of-flight mass analyzer with high resolving power. Rapid Commun Mass Spectrom 1988; 2: 83-5.
[24]
Ter Heine R, Beijnen JH, Huitema AD. Bioanalytical issues in patient-friendly sampling methods for therapeutic drug monitoring: Focus on antiretroviral drugs. Bioanalysis 2009; 1: 1329-38.
[25]
Pedersen OS, Pedersen EB. Non-nucleoside reverse transcriptase inhibitors: The NNRTI boom. Antivir Chem Chemother 1999; 10: 285-314.
[26]
Foldes O, Uherova P, Mayer V. Plasma levels of the anti-HIV drug 3′-azido-2′,3′-dideoxythymidine [AZT]: Deter-mination by RIA and HPLC. Acta Virol 1993; 3: 156-64.
[27]
Nadal T, Ortuño J, Pascual JA. Rapid and sensitive determination of zidovudine and zidovudine glucuronide in human plasma by ion-pair high-performance liquid chromatography. J Chromatogr A 1996; 721: 127-37.
[28]
Bloom J, Ortiz J, Rodríguez J. Azidothymidine triphosphate determination using micellar electrokinetic capillary chromatography. Cell Mol Biol 1997; 43: 1051-5.
[29]
Tan X, Boudinot FD. Simultaneous determination of zidovudine and its monophosphate in mouse plasma and peripheral red blood cells by high-performance liquid chromatography. J Chromatogr B Biomed Sci Appl 2000; 740: 281-7.
[30]
Brown SD, White CA, Chu CK, Bartlett MG. Determination of acyclovir in maternal plasma, amniotic fluid, fetal and placental tissues by high-performance liquid chromatography. J Chromatogr B Analyt Technol Biomed Life Sci 2002; 772: 327-34.
[31]
Dunge A, Sharda N, Singh B, Singh S. Validated specific HPLC method for determination of zidovudine during stability studies. J Pharm Biomed Anal 2005; 37: 1109-14.
[32]
Quevedo MA, Teijeiro SA, Briñón MC. Quantitative plasma determination of a novel antiretroviral derivative of zidovudine by solid-phase extraction and high-performance liquid chromatography. Anal Bioanal Chem 2006; 385: 377-84.
[33]
Granich GG, Eveland MR, Krogstad DJ. Fluorescence polarization immunoassay for zidovudine. Antimicrob Agents Chemother 1989; 33: 1275-9.
[34]
Stefan RI, Bokretsion RG. Diamond paste based immunosensor for the determination of azidothymidine. J Immunoassay Immunochem 2003; 24: 319-24.
[35]
Raviolo MA, Sanchez JM, Briñón MC, Perillo MA. Determination of liposome permeability of ionizable carbamates of zidovudine by steady state fluorescence spectroscopy. Colloids Surf B Biointerfaces 2008; 61: 188-98.
[36]
Barone GC, Halsall HB, Heineman WR. Electrochemistry of azidothymidine. Anal Chim Acta 1991; 248: 399-407.
[37]
Barone GC, Pesce AJ, Halsall HB, Heineman WR. Electrochemical determination of azidothymidine in human whole blood. Anal Biochem 1991; 198: 6-9.
[38]
Trnková L, Kizek R, Vacek J. Square wave and elimination voltammetric analysis of azidothymidine in the presence of oligonucleotides and chromosomal DNA. Bioelectrochemistry 2004; 63: 31-6.
[39]
Castro AA, Cordoves AIP, Farias PAM. Determination of the antiretroviral drug acyclovir in diluted alkaline electrolyte by adsorptive stripping voltammetry at the mercury film electrode. Anal Chem Insights 2013; 8: 21-8.
[40]
Klecker RW, Collins JM, Yarchoan R, et al. Plasma and cerebrospinal fluid pharmacokinetics of 3′-azido-3′-deoxythymidine: A novel pyrimidine analog with potential application for the treatment of patients with AIDS and related diseases. Clin Pharmacol Ther 1987; 41: 407-12.
[41]
Jung BH, Rezk NL, Bridges AS, Corbett AH, Kashuba ADM. Simultaneous determination of 17 antiretroviral drugs in human plasma for quantitative analysis with liquid chromatography-tandem mass spectrometry. Biomed Chromatogr 2007; 21: 1095-104.
[42]
Nandi U, Das A, Roy B, Choudhury H, Gorain B, Pal TK. Development and validation of an HPLC-UV method for simultaneous determination of zidovudine, lamivudine, and nevirapine in human plasma and its application to pharmacokinetic study in human volunteers. Drug Test Anal 2013; 5: 485-91.
[43]
Matta MK, Pilli NR, Kumar IJK, Burugula L. Rao JVLN S. Simultaneous quantitation of lamivudine, zidovudine and nevirapine in human plasma by liquid chromatography-tandem mass spectrometry and application to a pharmacokinetic study. Acta Pharm Sin B 2012; 2: 472-80.
[44]
Veal GJ, Back DJ. Metabolism of zidovudine. Gen Pharmacol 1995; 26: 1469-75.
[45]
Campbell TB, Shulman NS, Johnson SC, et al. Antiviral activity of lamivudine in salvage therapy for multidrug-resistant HIV-1 infection. Clin Infect Dis 2005; 41: 236-42.
[46]
Lau GK, Piratvisuth T, Luo KX, et al. Peginterferon Alfa-2a, lamivudine, and the combination for HBeAg-positive chronic hepatitis B. N Engl J Med 2005; 352: 2682-95.
[47]
Harker AJ, Evans GL, Hawley AE, Morris DM. High-performance liquid chromatographic assay for 2′-deoxy-3′-thiacytidine in human serum. J Chromatogr B Biomed Appl 1994; 657: 227-32.
[48]
Hsyu PH, Lloyd TL. Automated high-performance liquid chromatographic analysis of [-]-2′-deoxy-3′-thiacytidine in biological fluids using the automated sequential trace enrichment of dialysate systems. J Chromatogr B Biomed Appl 1994; 655: 253-9.
[49]
Robbins BL, Tran TT, Pinkerton FH, et al. Development of a new cartridge radioimmunoassay for determination of intracellular levels of lamivudine triphosphate in the peripheral blood mononuclear cells of human immunodeficiency virus-infected patients. Antimicrob Agents Chemother 1998; 42: 2656-60.
[50]
Plumb RS, Gray RDM, Harker aJ, Taylor S. High-performance chromatographic assay for the sulphoxide metabolite 2′-deoxy-3′-thiacytidine in human urine. J Chromatogr B Biomed Appl 1996; 687: 457-61.
[51]
Zhou XJ, Sommadossi JP. Rapid quantitation of [-]-2′-deoxy-3′-thiacytidine in human serum by high-performance liquid chromatography with ultraviolet detection. J Chromatogr B Biomed Sci Appl 1997; 691: 417-24.
[52]
Hoetelmans RM, Van Essenberg M, Meenhorst PL, Mulder JW, Beijnen JH. Determination of saquinavir in human plasma, saliva, and cerebrospinal fluid by ion-pair high-performance liquid chromatography with ultraviolet detection. J Chromatogr B Analyt Technol Biomed Life Sci 1997; 698: 235-41.
[53]
Kenney KB, Wring SA, Carr RM, Wells GN, Dunn JA. Simultaneous determination of zidovudine and lamivudine in human serum using HPLC with tandem mass spectrometry. J Pharm Biomed Anal 2000; 22: 967-83.
[54]
Estrela Rde C, Salvadori MC, Suarez-Kurtz G. A rapid and sensitive method for simultaneous determination of lamivudine and zidovudine in human serum by on-line solid-phase extraction coupled to liquid chromatography/ tandem mass spectrometry detection. Rapid Commun Mass Spectrom 2004; 18: 1147-55.
[55]
Rower JE, Klein B, Bushman LR, Anderson PL. Validation of a sensitive LC/MS/MS method for the determination of zidovudine and lamivudine in human plasma. Biomed Chromatogr 2012; 26: 12-20.
[56]
Pereira AS, Kenney KB, Cohen MS, et al. Simultaneous determination of lamivudine and zidovudine concentrations in human seminal plasma using high-performance liquid chromato-graphy and tandem mass spectrometry. J Chromatogr B Biomed Sci Appl 2000; 742: 173-83.
[57]
Verma S, Mullick P, Bhatt MS, et al. Bioanalytical method development and validation for the simultaneous estimation of lamivudine and stavudine in human plasma by HPLC. Acta Pol Pharm 2010; 67: 429-37.
[58]
Wattananat T, Prasanchaimontri I, Akarawut W. Simultaneous determination of stavudine and lamivudine in human plasma by high performance liquid chromatography and its application to a bioavailability study. Southeast Asian J Trop Med Public Health 2010; 41: 369-77.
[59]
Fan B, Stewart JT. Determination of zidovudine/ lamivudine/nevirapine in human plasma using ion-pair HPLC. J Pharm Biomed Anal 2002; 28: 903-8.
[60]
Aymard G, Legrand M, Trichereau N, Diquet B. Determination of twelve antiretroviral agents in human plasma sample using reversed-phase high-performance liquid chromatography. J Chromatogr B Biomed Sci Appl 2000; 744: 227-40.
[61]
Simon VA, Thiam MD, Lipford LC. Determination of serum levels of thirteen human immunodeficiency virus-suppressing drugs by high-performance liquid chromatography. J Chromatogr A 2001; 913: 447-53.
[62]
Rezk NL, Tidwell RR, Kashuba ADM. Simultaneous determination of six HIV nucleoside analogue reverse transcriptase inhibitors and nevirapine by liquid chromatography with ultraviolet absorbance detection. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 791: 137-47.
[63]
Notari S, Bocedi A, Ippolito G, et al. Simultaneous determination of 16 anti-HIV drugs in human plasma by high-performance liquid chromatography. J Chromatogr B Analyt Technol Biomed Life Sci 2006; 831: 258-66.
[64]
Tarinas A, Tápanes RD, Ferrer G, Pérez J. Validation of high-performance liquid chromatography methods for determination of zidovudine, stavudine, lamivudine and indinavir in human plasma. Farm Hosp 2007; 31: 243-7.
[65]
Pynnönen ST, Tuhkanen TA. Simultaneous detection of three antiviral and four antibiotic compounds in source-separated urine with liquid chromatography. J Sep Sci 2014; 37: 219-27.
[66]
Robbins BL, Poston PA, Neal EF, Slaughter C, Rodman JH. Simultaneous measurement of intracellular triphosphate metabolites of zidovudine, lamivudine and abacavir [carbovir] in human peripheral blood mononuclear cells by combined anion exchange solid phase extraction and LC-MS/MS. J Chromatogr B Analyt Technol Biomed Life Sci 2007; 850: 310-7.
[67]
Solas C, Li YF, Xie MY, Sommadossi JP, Zhou XJ. Intracellular nucleotides of [-]-2′,3′-deoxy-3′-thiacytidine in peripheral blood mononuclear cells of a patient infected with human immunodeficiency virus. Antimicrob Agents Chemother 1998; 42: 2989-95.
[68]
Becher F, Pruvost A, Goujard C, et al. Improved method for the simultaneous determination of d4T, 3TC and ddl intracellular phosphorylated anabolites in human peripheral-blood mononuclear cells using high-performance liquid chromatography/tandem mass spectrometry. Rapid Commun Mass Spectrom 2002; 16: 555-65.
[69]
Nirogi R, Kandikere V, Komarneni P, et al. Exploring dried blood spot sampling technique for simultaneous quantification of antiretrovirals: Lamivudine, stavudine and nevirapine in a rodent pharmacokinetic study. Biomed Chromatogr 2012; 26: 1472-81.
[70]
Chapman T, McGavin J, Noble S. Tenofovir disoproxil fumarate. Drugs 2003; 63: 1597-608.
[71]
Kearney BP, Flaherty JF, Shah J. Tenofovir disoproxil fumarate: Clinical pharmacology and pharmacokinetics. Clin Pharmacokinet 2004; 43: 595-612.
[72]
Gervasoni C, Meraviglia P, Landonio S, et al. Low body weight in females is a risk factor for increased tenofovir exposure and drug-related adverse events. PLoS One 2013; 8: 8-13.
[73]
Delahunty T, Bushman L, Fletcher CV. Sensitive assay for determining plasma tenofovir concentrations by LC/MS/MS. J Chromatogr B Analyt Technol Biomed Life Sci 2006; 830: 6-12.
[74]
Sentenac S, Fernandez C, Thuillier A, Lechat P, Aymard G. Sensitive determination of tenofovir in human plasma samples using reversed-phase liquid chromatography. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 793: 317-24.
[75]
Sparidans RW, Crommentuyn KM, Schellens JH, Beijnen JH. Liquid chromatographic assay for the antiviral nucleotide analogue tenofovir in plasma using derivatization with chloroacetaldehyde. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 791: 227-33.
[76]
Jullien V, Treluyer J, Rey E. Determination of tenofovir in human plasma by high-performance liquid chromatography with spectrofluorimetric detection. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 785: 377-81.
[77]
King T, Bushman L, Kiser J, et al. Liquid chromatography-tandem mass spectrometric determination of tenofovir-diphosphate in human peripheral blood mononuclear cells. J Chromatogr B Analyt Technol Biomed Life Sci 2006; 843: 147-56.
[78]
Barkil ME, Gagnieu MC, Guitton J. Relevance of a combined UV and single mass spectrometry detection for the determination of tenofovir in human plasma by HPLC in therapeutic drug monitoring. J Chromatogr B Analyt Technol Biomed Life Sci 2007; 854: 192-7.
[79]
D’Avolio A, Simiele M, Siccardi M, et al. A HPLC-MS method for the simultaneous quantification of fourteen antiretroviral agents in peripheral blood mononuclear cell of HIV infected patients optimized using medium corpuscular volume evaluation. J Pharm Biomed Anal 2011; 54: 779-88.
[80]
Coulier L, Gerritsen H, van Kampen JJ, et al. Comprehensive analysis of the intracellular metabolism of antiretroviral nucleosides and nucleotides using liquid chromatography-tandem mass spectrometry and method improvement by using ultra performance liquid chromatography. J Chromatogr B Analyt Technol Biomed Life Sci 2011; 879: 2772-82.
[81]
Dando TM, Wagstaff AJ. Emtricitabine/tenofovir disoproxil fumarate. Drugs 2004; 64: 2075-82.
[82]
Avihingsanon A, Lewin SR, Kerr S, et al. Efficacy of tenofovir disoproxil fumarate/emtricitabine compared with emtricitabine alone in antiretroviral-naive HIV-HBV coinfection in Thailand. Antivir Ther 2010; 15: 917-22.
[83]
Moody W. Truvada works to block HIV transmission. Echo Mag 2012; 24: 66-7.
[84]
Rezk NL, Crutchley RD, Kashuba AD. Simultaneous quantification of emtricitabine and tenofovir in human plasma using high-performance liquid chromatography after solid phase extraction. J Chromatogr B Analyt Technol Biomed Life Sci 2005; 822: 201-8.
[85]
Gomes NA, Vaidya VV, Pudage A, Joshi SS, Parekh SA. Liquid chromatography-tandem mass spectrometry [LC-MS/MS] method for simultaneous determination of tenofovir and emtricitabine in human plasma and its application to a bioequivalence study. J Pharm Biomed Anal 2008; 48: 918-26.
[86]
Wang LH, Wiznia AA, Rathore MH, et al. Pharmacokinetics and safety of single oral doses of emtricitabine in human immunodeficiency virus-infected children. Antimicrob Agents Chemother 2004; 48: 183-91.
[87]
Darque A, Valette G, Rousseau F, Wang LH, Sommadossi JP, Zhou XJ. Quantitation of intracellular triphosphate of emtricitabine in peripheral blood mononuclear cells from human immunodeficiency virus-infected patients. Antimicrob Agents Chemother 1999; 43: 2245-50.
[88]
Jansen RS, Rosing H, Kromdijk W, Ter Heine R, Schellens JH, Beijnen JH. Simultaneous quantification of emtricitabine and tenofovir nucleotides in peripheral blood mononuclear cells using weak anion-exchange liquid chromatography coupled with tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2010; 878: 621-7.
[89]
Kromdijk W, Pereira SA, Rosing H, Mulder JW, Beijnen JH, Huitema AD. Development and validation of an assay for the simultaneous determination of zidovudine, abacavir, emtricitabine, lamivudine, tenofovir and ribavirin in human plasma using liquid chromatography-tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2013; 919: 43-51.
[90]
Foster RH, Faulds D. Abacavir. Drugs 1998; 55: 729-38.
[91]
Chittick GE, Gillotin C, McDowell JA, et al. Abacavir: absolute bioavailability, bioequivalence of three oral formulations, and effect of food. Pharmacotherapy 1999; 19: 932-42.
[92]
McDowell JA, Chittick GE, Ravitch JR, Polk RE, Kerkering TM, Stein DS. Pharmacokinetics of [[14]C] abacavir, a human immunodeficiency virus type 1 [HIV-1] reverse transcriptase inhibitor, administered in a single oral dose to HIV-1-infected adults: a mass balance study. Antimicrob Agents Chemother 1999; 43: 2855-61.
[93]
Kumar PN, Sweet DE, McDowell JA, et al. Safety and pharmacokinetics of abacavir [1592U89] following oral administration of escalating single doses in human immunodeficiency virus type 1-infected adults. Antimicrob Agents Chemother 1999; 43: 603-8.
[94]
Weller S, Radomski KM, Lou Y, Stein DS. Population pharmacokinetics and pharmacodynamic modeling of abacavir [1592U89] from a dose-ranging, double-blind, randomized monotherapy trial with human immuno-deficiency virus-infected subjects. Antimicrob Agents Chemother 2000; 44: 2052-60.
[95]
Faletto MB, Miller WH, Garvey EP, St Clair MH, Daluge SM, Good SS. Unique intracellular activation of the potent anti-human immunodeficiency virus agent 1592U89. Antimicrob Agents Chemother 1997; 41: 1099-107.
[96]
Somboonwit C, Kurtyka D, Velez AP. Abacavir and lamivudine combination. Expert Opin Drug Metab Toxicol 2009; 5: 1599-606.
[97]
Ibbotson T, Perry CM. Lamivudine/zidovudine/abacavir: Triple combination tablet. Drugs 2003; 63: 1089-98.
[98]
Mcdowell JA, Chittick GE, Stevens CP, Edwards KD, Stein DS. Pharmacokinetic interaction of abacavir [1592U89] and ethanol in human immunodeficiency virus-infected adults. Antimicrob Agents Chemother 2000; 44: 1686-90.
[99]
Yuen GJ, Weller S, Pakes GE. A review of the pharmacokinetics of abacavir. Clin Pharmacokinet 2008; 47: 351-71.
[100]
Seshachalam U, Haribabu B, Chandrasekhar KB. Development and validation of a reverse-phase liquid chromatographic method for assay and related substances of abacavir sulfate. J Sep Sci 2007; 30: 28-34.
[101]
Uslu B, Özkan SA. Anodic voltammetry of abacavir and its determination in pharmaceuticals and biological fluids. Electrochim Acta 2004; 49: 4321-9.
[102]
Ferrer SM, Modamio P, Lastra CF, Mariño EL. Determination of abacavir in human plasma by high-performance liquid chromatography with ultraviolet detection and the analytical error function. Biomed Chromatogr 2004; 18: 862-5.
[103]
Veldkamp AI, Sparidans RW, Hoetelmans RM, Beijnen JH. Quantitative determination of abacavir [1592U89], a novel nucleoside reverse transcriptase inhibitor, in human plasma using isocratic reversed-phase high-performance liquid chromatography with ultraviolet detection. J Chromatogr B Biomed Sci Appl 1999; 736: 123-8.
[104]
Clark TN, White CA, Bartlett MG. Determination of abacavir in maternal plasma, amniotic fluid, fetal and placental tissues by a polarity switching liquid chromato-graphy/tandem mass spectrometry method. Rapid Commun Mass Spectrom 2004; 18: 405-11.
[105]
Ravitch JR, Moseley CG. High-performance liquid chromatographic assay for abacavir and its two major metabolites in human urine and cerebrospinal fluid. J Chromatogr B Analyt Technol Biomed Life Sci 2015; 762: 165-73.
[106]
Fung EN, Cai Z, Burnette TC, Sinhababu AK. Simultaneous determination of Ziagen and its phosphorylated metabolites by ion-pairing high-performance liquid chromatography-tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2001; 754: 285-95.
[107]
Sparidans RW, Hoetelmans RM, Beijnen JH. Liquid chromatographic assay for simultaneous determination of abacavir and mycophenolic acid in human plasma using dual spectrophotometric detection. J Chromatogr B Biomed Sci Appl 2001; 750: 155-61.
[108]
Lewis SR, White CA, Bartlett MG. Simultaneous determination of abacavir and zidovudine from rat tissues using HPLC with ultraviolet detection. J Chromatogr B Analyt Technol Biomed Life Sci 2007; 850: 45-52.
[109]
Pruvost A, Theodoro F, Agrofoglio L. EN and, BeNech H. Specificity enhancement with LC-positive ESI-MS/MS for the measurement of nucleotides: Application to the quantitative determination of carbovir triphosphate, lamivudine triphosphate and tenofovir diphosphate in human peripheral blood mononuclear cells. J Mass Spectrom 2008; 43: 224-33.
[110]
Verweij-van WCP, Aarnoutse RE, Burger DM. Simultaneous determination of the HIV nucleoside analogue reverse transcriptase inhibitors lamivudine, didanosine, stavudine, zidovudine and abacavir in human plasma by reversed phase high performance liquid chromatography. J Chromatogr B Analyt Technol Biomed Life Sci 2005; 816: 121-9.
[111]
Yadav M, Gupta A, Singhal P, Shrivastav PS. Development and validation of a selective and rapid LC-MS-MS method for the quantification of abacavir in human plasma. J Chromatogr Sci 2010; 48: 654-62.
[112]
Le Saux T, Chhun S, Rey E, et al. Quantification of seven nucleoside/nucleotide reverse transcriptase inhibitors in human plasma by high-performance liquid chromatography with tandem mass-spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2008; 865: 81-90.
[113]
Notari S, Mancone C, Alonzi T, Tripodi M, Narciso P, Ascenzi P. Determination of abacavir, amprenavir, didanosine, efavirenz, nevirapine, and stavudine concentration in human plasma by MALDI-TOF/TOF. J Chromatogr B Anal Technol Biomed LifeSci 2008; 863: 249-57.
[114]
Cheeseman SH, Hattox SE, McLaughlin MM, et al. Pharmacokinetics of nevirapine: Initial single-rising-dose study in humans. Antimicrob Agents Chemother 1993; 37: 178-82.
[115]
Havlir D, Cheeseman SH, McLaughlin M, et al. High-dose nevirapine: Safety, pharmacokinetics, and antiviral effect in patients with human immunodeficiency virus infection. J Infect Dis 1995; 171: 537-45.
[116]
Van Heeswijk RP, Hoetelmans RM, Meenhorst PL, Mulder JW, Beijnen JH. Rapid determination of nevirapine in human plasma by ion-pair reversed-phase high-performance liquid chromatography with ultraviolet detection. J Chromatogr B Biomed Sci Appl 1998; 713: 395-9.
[117]
Pav JW, Rowland LS, Korpalski DJ. HPLC-UV method for the quantitation of nevirapine in biological matrices following solid phase extraction. J Pharm Biomed Anal 1999; 20: 91-8.
[118]
Merry C, Barry MG, Mulcahy F, Halifax KL, Back DJ. Saquinavir pharmacokinetics alone and in combination with nelfinavir in HIV-infected patients. AIDS 1997; 11: 117-20.
[119]
Moyer TP, Temesgen Z, Enger R, et al. Drug monitoring of antiretroviral therapy for HIV-1 infection: method validation and results of a pilot study. Clin Chem 1999; 45: 1465-76.
[120]
Hollanders RM, Kolmer E, Burger DM, Wuis EW, Koopmans PP, Hekster YA. Determination of nevirapine, an HIV-1 non-nucleoside reverse transcriptase inhibitor, in human plasma by reversed-phase high-performance liquid chromatography. J Chromatogr B Biomed Sci Appl 2000; 744: 65-71.
[121]
Silverthorn CF, Parsons TL. A validated new method for nevirapine quantitation in human plasma via high-performance liquid chromatography. Biomed Chromatogr 2006; 20: 23-7.
[122]
Villani P, Feroggio M, Gianelli L, et al. Antiretrovirals: simultaneous determination of five protease inhibitors and three nonnucleoside transcriptase inhibitors in human plasma by a rapid high-performance liquid chromatography--mass spectrometry assay. Ther Drug Monit 2001; 23: 380-8.
[123]
Laurito TL, Santagada V, Caliendo G, Oliveira CH, Barrientos-Astigarraga RE, De Nucci G. Nevirapine quantification in human plasma by high-performance liquid chromatography coupled to electrospray tandem mass spectrometry. Application to bioequivalence study. J Mass Spectrom 2002; 37: 434-41.
[124]
Chi J, Jayewardene AL, Stone JA, Aweeka FT. An LC-MS-MS method for the determination of nevirapine, a non-nucleoside reverse transcriptase inhibitor, in human plasma. J Pharm Biomed Anal 2003; 3: 953-9.
[125]
Liu Z, Fan-Havard P, Xie Z, Ren C, Chan KK. A liquid chromatography/atmospheric pressure ionization tandem mass spectrometry quantitation method for nevirapine and its two oxidative metabolites, 2-hydroxynevirapine and nevirapine 4-carboxylic acid, and pharmacokinetics in baboons. Rapid Commun Mass Spectrom 2007; 21: 2734-42.
[126]
Vogel M, Bertram N, Wasmuth JC, Emmelkamp J, Rockstroh JK, Reichel C. Determination of nevirapine in plasma by GC-MS. J Chromatogr Sci 2010; 48: 91-4.
[127]
Kromdijk W, Mulder JW, Rosing H, Smit PM, Beijnen JH, Huitema ADR. Use of dried blood spots for the determination of plasma concentrations of nevirapine and efavirenz. J Antimicrob Chemother 2012; 67: 1211-6.
[128]
Maggiolo F. Efavirenz: a decade of clinical experience in the treatment of HIV. J Antimicrob Chemother 2009; 64: 910-28.
[129]
McIlleron HM. Effects of rifampin-based antituberculosis therapy on plasma efavirenz concentrations in children vary by CYP2B6 genotype. AIDS 2013; 27: 1933-40.
[130]
Friedland G, Khoo S, Jack C, Lalloo U. Administration of efavirenz [600 mg/day] with rifampicin results in highly variable levels but excellent clinical outcomes in patients treated for tuberculosis and HIV. J Antimicrob Chemother 2006; 58: 1299-302.
[131]
Avery LB, Parsons TL, Meyers DJ, Hubbard WC. A highly sensitive ultra performance liquid chromatography-tandem mass spectrometric [UPLC-MS/MS] technique for quantitation of protein free and bound efavirenz [EFV] in human seminal and blood plasma. J Chromatogr B Analyt Technol Biomed Life Sci 2010; 878: 3217-24.
[132]
Lee LL, Herold ML. Zacchei a G. High-performance liquid chromatographic method for the determination of an HIV-1 non-nucleoside reverse transcriptase inhibitor [L-696,229] in plasma samples from animals. J Chromatogr B Biomed Appl 1996; 685: 323-8.
[133]
Villani P, Pregnolato M, Banfo S, et al. High-performance liquid chromatography method for analyzing the anti-retroviral agent efavirenz in human plasma. Ther Drug Monit 1991; 21: 346-50.
[134]
Veldkamp AI, Van Heeswijk RP, Meenhorst PL, et al. Quantitative determination of efavirenz [DMP 266], a novel non-nucleoside reverse transcriptase inhibitor, in human plasma using isocratic reversed-phase high-performance liquid chromatography with ultraviolet detection. J Chromatogr B Biomed Sci Appl 1999; 734: 55-61.
[135]
Langmann P, Schirmer D, Vath T, Desch S, Zilly M, Klinker H. Rapid determination of nevirapine in human plasma by gas chromatography. J Chromatogr B Analyt Technol Biomed Life Sci 2002; 767: 69-74.
[136]
Saras-Nacenta M, López-Púa Y, Lípez-Cortés LF, Mallolas J, Gatell JM, Carné X. Determination of efavirenz in human plasma by high-performance liquid chromatography with ultraviolet detection. J Chromatogr B Biomed Sci Appl 2001; 763: 53-9.
[137]
Matthews CZ, Woolf EJ, Mazenko RS, et al. Determination of efavirenz, a selective non-nucleoside reverse transcriptase inhibitor, in human plasma using HPLC with post-column photochemical derivatization and fluorescence detection. J Pharm Biomed Anal 2002; 28: 925-34.
[138]
Ramachandran G, Hemanthkumar AK, Kumaraswami V, Swaminathan S. A simple and rapid liquid chromatography method for simultaneous determination of zidovudine and nevirapine in plasma. J Chromatogr B Analyt Technol Biomed Life Sci 2006; 843: 339-44.
[139]
Mogatle S, Kanfer I. Rapid method for the quantitative determination of efavirenz in human plasma. J Pharm Biomed Anal 2009; 49: 1308-12.
[140]
Bienvenu E, Hoffmann KJ, Ashton M, Kayumba PC. A rapid and selective HPLC-UV method for the quantitation of efavirenz in plasma from patients on concurrent HIV/AIDS and tuberculosis treatments. Biomed Chromatogr 2013; 27: 1554-9.
[141]
D’Avolio A, Simiele M, Siccardi M, et al. HPLC-MS method for the quantification of nine anti-HIV drugs from dry plasma spot on glass filter and their long term stability in different conditions. J Pharm Biomed Anal 2010; 52: 774-80.
[142]
Koal T, Burhenne H, Römling R, Svoboda M, Resch K, Kaever V. Quantification of antiretroviral drugs in dried blood spot samples by means of liquid chromatography/ tandem mass spectrometry. Rapid Commun Mass Spectrom 2005; 19: 2995-3001.
[143]
Hoffman JT, Rossi SS, Espina-Quinto R, Letendre S, Capparelli EV. Determination of efavirenz in human dried blood spots by reversed-phase high-performance liquid chromatography with UV detection. Ther Drug Monit 2013; 35: 203-8.
[144]
Rouzes A, Berthoin K, Xuereb F, et al. Simultaneous determination of the antiretroviral agents: Amprenavir, lopinavir, ritonavir, saquinavir and efavirenz in human peripheral blood mononuclear cells by high-performance liquid chromatography-mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2004; 813: 209-16.
[145]
Colombo S, Beguin A, Telenti A, et al. Intracellular measurements of anti-HIV drugs indinavir, amprenavir, saquinavir, ritonavir, nelfinavir, lopinavir, atazanavir, efavirenz and nevirapine in peripheral blood mononuclear cells by liquid chromatography coupled to tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2005; 819: 259-76.
[146]
Chi B, Lee A, Acosta E, Westerman L, Sinkala M, Stringer J. Field Performance of a Thin-Layer Chromatography Assay for Detection of Nevirapine in Umbilical Cord Blood. HIV Clin Trials 2006; 7: 263-9.
[147]
Podany AT, Winchester LC, Robbins BL, Fletcher CV. Quantification of Cell-Associated Atazanavir, Darunavir, Lopinavir, Ritonavir, and Efavirenz Concentrations in Human Mononuclear Cell Extracts. Antimicrob Agents Chemother 2014; 58: 2866-70.
[148]
Amara AB, Else LJ, Tjia J, et al. A validated method for quantification of efavirenz in dried blood spots using high-performance liquid chromatography-mass spectrometry. Ther Drug Monit 2015; 37: 220-8.
[149]
Proust V, Toth K, Hulin A, Taburet AM, Gimenez F, Singlas E. Simultaneous high-performance liquid chromatographic determination of the antiretroviral agents amprenavir, nelfinavir, ritonavir, saquinavir, delavirdine and efavirenz in human plasma. J Chromatogr B Biomed Sci Appl 2000; 742: 453-8.
[150]
Aymard G, Legrand M, Trichereau N, Diquet B. Determination of twelve antiretroviral agents in human plasma sample using reversed-phase high-performance liquid chromatography. J Chromatogr B Biomed Sci Appl 2000; 744: 227-40.
[151]
Marzolini C, Telenti A, Buclin T, Biollaz J, Decosterd LA. Simultaneous determination of the HIV protease inhibitors indinavir, amprenavir, saquinavir, ritonavir, nelfinavir and the non-nucleoside reverse transcriptase inhibitor efavirenz by high-performance liquid chromatography after solid-phase extraction. J Chromatogr B Biomed Sci Appl 2000; 740: 43-58.
[152]
Boffito M, Tija J, Reynolds HE, et al. Simultaneous determination of rifampicin and efavirenz in plasma. Ther Drug Monit 2002; 24: 670-4.
[153]
Rezk NL, Tidwell RR, Kashuba AD. Simple and rapid quantification of the non-nucleoside reverse transcriptase inhibitors nevirapine, delavirdine, and efavirenz in human blood plasma using high-performance liquid chromato-graphy with ultraviolet absorbance detection. J Chromatogr B Analyt Technol Biomed Life Sci 2002; 774: 79-88.
[154]
Tribut O, Arvieux C, Michelet C, Chapplain J, Allain H, Bentué-ferrer D. Simultaneous Quantitative Assay of Six HIV Protease Inhibitors, One Metabolite, And Two Non-Nucleoside Reverse Transcriptase Inhibitors in Human Plasma by Isocratic Reversed-Phase Liquid Chromato-graphy. Ther Drug Monit 2002; 24: 554-62.
[155]
Usami Y, Oki T, Nakai M, Sagisaka M, Kaneda T. A simple HPLC method for simultaneous determination of lopinavir, ritonavir and efavirenz. Chem Pharm Bull 2003; 51: 715-8.
[156]
Turner ML, Reed-Walker K, King JR, Acosta EP. Simultaneous determination of nine antiretroviral compounds in human plasma using liquid chromatography. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 784: 331-41.
[157]
Rezk NL, Tidwell RR, Kashuba AD. High-performance liquid chromatography assay for the quantification of HIV protease inhibitors and non-nucleoside reverse transcriptase inhibitors in human plasma. J Chromatogr B Analyt Technol Biomed Life Sci 2004; 805: 241-7.
[158]
Dailly E, Raffi F, Jolliet P. Determination of atazanavir and other antiretroviral drugs [indinavir, amprenavir, nelfinavir and its active metabolite M8, saquinavir, ritonavir, lopinavir, nevirapine and efavirenz] plasma levels by high performance liquid chromatography with UV detecti. J Chromatogr B Analyt Technol Biomed Life Sci 2004; 813: 353-8.
[159]
Takahashi M, Yoshida M, Oki T, Okumura N, Suzuki T, Kaneda T. Conventional HPLC method used for simultaneous determination of the seven HIV protease inhibitors and nonnucleoside reverse transcription inhibitor efavirenz in human plasma. Biol Pharm Bull 2005; 28: 1286-90.
[160]
Tribut O, Verdier MC, Arvieux C, Allain H, Michelet C, Bentué-Ferrer D. Simultaneous quantitative assay of atazanavir and 6 other HIV protease inhibitors by isocratic reversed-phase liquid chromatography in human plasma. Ther Drug Monit 2005; 27: 265-9.
[161]
Rezk NL, Crutchley RD, Yeh RF, Kashuba AD. Full validation of an analytical method for the HIV-protease inhibitor atazanavir in combination with 8 other antiretroviral agents and its applicability to therapeutic drug monitoring. Ther Drug Monit 2006; 28: 517-25.
[162]
Hirabayashi Y, Tsuchiya K, Kimura S, Oka S. Simultaneous determination of six HIV protease inhibitors [amprenavir, indinavir, lopinavir, nelfinavir, ritonavir and saquinavir], the active metabolite of nelfinavir [M8] and non-nucleoside reverse transcriptase inhibitor [efavirenz] in human plasma by. Biomed Chromatogr 2006; 20: 28-36.
[163]
Weller DR, Brundage RC, Balfour HH, Vezina HE. An isocratic liquid chromatography method for determining HIV non-nucleoside reverse transcriptase inhibitor and protease inhibitor concentrations in human plasma. J Chromatogr B Analyt Technol Biomed Life Sci 2007; 848: 369-73.
[164]
Maganda B, Heudi O, Cortinovis A, Picard F, Kretz O, Minzi O. A fast and reliable reversed phase high performance liquid chromatography method for simultaneous determination of selected anti-retroviral and lumefantrine in human plasma. J Chromatogr B Analyt Technol Biomed Life Sci 2013; 919-920: 52-60.
[165]
Volosov A, Alexander C, Ting L, Soldin SJ. Simple rapid method for quantification of antiretrovirals by liquid chromatography-tandem mass-spectrometry. Clin Biochem 2002; 35: 99-103.
[166]
Rentsch KM. Sensitive and specific determination of eight antiretroviral agents in plasma by high-performance liquid chromatography - mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 788: 339-50.
[167]
Ter Heine R, Alderden-Los CG, Rosing H, et al. Fast and simultaneous determination of darunavir and eleven other antiretroviral drugs for therapeutic drug monitoring: method development and validation for the determination of all currently approved HIV protease inhibitors and non-nucleoside reverse transcriptase inhibitors in human plasma by liquid chromatography coupled with electrospray ionization tandem mass spectrometry. Rapid Commun Mass Spectrom 2007; 21: 2505-14.
[168]
Nirogi R, Bhyrapuneni G, Kandikere V, et al. Simultaneous quantification of a non-nucleoside reverse transcriptase inhibitor efavirenz, a nucleoside reverse transcriptase inhibitor emtricitabine and a nucleotide reverse transcriptase inhibitor tenofovir in plasma by liquid chromatography positive io. Biomed Chromatogr 2009; 23: 371-81.
[169]
Quaranta S, Woloch C, Paccou A, Giocanti M, Solas C, Lacarelle B. Validation of an electrospray ionization LC-MS/MS method for quantitative analysis of raltegravir, etravirine, and 9 other antiretroviral agents in human plasma samples. Ther Drug Monit 2009; 31: 695-702.
[170]
Ter Heine R, Rosing H, Beijnen JH, Huitema ADR. A less sensitive detector does not necessarily result in a less sensitive method: Fast quantification of 13 antiretroviral analytes in plasma with liquid chromatography coupled with tandem mass spectrometry. Clin Chem Lab Med 2010; 48: 1153-5.
[171]
Deeks ED, Keating GM. Etravirine. Drugs 2008; 68: 2357-72.
[172]
Schöller-Gyüre M, Kakuda TN, Raoof A, De Smedt G, Hoetelmans RMW. Clinical pharmacokinetics and pharmacodynamics of etravirine. Clin Pharmacokinet 2009; 48: 561-74.
[173]
Ter Heine R, Rosing H, van Gorp ECM, Mulder JW, Beijnen JH, Huitema ADR. Quantification of etravirine [TMC125] in plasma, dried blood spots and peripheral blood mononuclear cell lysate by liquid chromatography tandem mass spectrometry. J Pharm Biomed Anal 2009; 49: 393-400.
[174]
Ter Heine R, Rosing H, van Gorp ECM, et al. Quantification of protease inhibitors and non-nucleoside reverse transcriptase inhibitors in dried blood spots by liquid chromatography-triple quadrupole mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2008; 867: 205-12.
[175]
Abobo CV, Wu L, John J, Joseph MK, Bates TR, Liang D. LC-MS/MS determination of etravirine in rat plasma and its application in pharmacokinetic studies. J Chromatogr B Analyt Technol Biomed Life Sci 2010; 878: 3181-6.
[176]
Fayet A, Béguin A, Zanolari B, et al. A LC-tandem MS assay for the simultaneous measurement of new antiretroviral agents: Raltegravir, maraviroc, darunavir, and etravirine. J Chromatogr B Analyt Technol Biomed Life Sci 2009; 877: 1057-69.
[177]
Else L, Watson V, Tjia J, et al. Validation of a rapid and sensitive high-performance liquid chromatography-tandem mass spectrometry [HPLC-MS/MS] assay for the simultaneous determination of existing and new antiretroviral compounds. J Chromatogr B Analyt Technol Biomed Life Sci 2010; 878: 1455-65.
[178]
Rezk NL, White NR, Jennings SH, Kashuba ADM. A novel LC-ESI-MS method for the simultaneous determination of etravirine, darunavir and ritonavir in human blood plasma. Talanta 2009; 79: 1372-8.
[179]
D’Avolio A, Siccardi M, Sciandra M, et al. HPLC-MS method for the simultaneous quantification of the new HIV protease inhibitor darunavir, and 11 other antiretroviral agents in plasma of HIV-infected patients. J Chromatogr B Analyt Technol Biomed Life Sci 2007; 859: 234-40.
[180]
Ford N, Lee J, Andrieux-Meyer I, Calmy A. Safety, efficacy, and pharmacokinetics of rilpivirine: systematic review with an emphasis on resource-limited settings. HIV AIDS 2011; 3: 35-44.
[181]
Mills EJ, Nachega JB, Bangsberg DR, et al. Adherence to HAART: A systematic review of developed and developing nation patient-reported barriers and facilitators. PLoS Med 2006; 3: 2039-64.
[182]
Santoscoy M, Cahn P, Gonsalez C, et al. TMC278 [rilpivirine], a next-generation NNRTI, demonstrates long-term efficacy and tolerability in ARV-naive patients: 96-week results of study C204. Proceedings of the XVII International AIDS Conference. 2008 Aug 3-8; Mexico City, MX.
[183]
Crauwels H, Van Heeswijk R, Kestens D, et al. The pharmacokinetic [PK] interaction between rifabutin and TMC278, an investigational non-nucleoside reverse transcriptase inhibitor [NNRTI]. Proceedings of the XVII International AIDS Conference. 2008 Aug 3-8; Mexico City, MX.
[184]
Burugula L, Pilli NR, Makula A, Lodagala DS, Kandhagatla R. Liquid chromatography-tandem mass spectrometric assay for the non-nucleoside reverse transcriptase inhibitor rilpivirine in human plasma. Biomed Chromatogr 2013; 27: 172-8.
[185]
Van Heeswijk R, Hoetelmans RMW, Kestens D. The effects of CYP3A4 modulation on the pharmacokinetics of TMC278, an investigational non-nucleoside reverse transcriptase inhibitor [NNRTI]. Proceedings of the 7th International Workshop of Clinical Pharmacology. 2006 April 20-22; Lisbon, Portugal.
[186]
Shibata M, Takahashi M, Yoshino M, et al. Development and application of a simple LC-MS method for the determination of plasma rilpivirine [TMC-278] concentrations. J Med Invest 2013; 60: 35-40.
[187]
Aouri M, Calmy A, Hirschel B, et al. A validated assay by liquid chromatography-tandem mass spectrometry for the simultaneous quantification of elvitegravir and rilpivirine in HIV positive patients. J Mass Spectrom 2013; 48: 616-25.
[188]
Gupta A, Guttikar S, Patel Y, Shrivastav PS, Sanyal M. Reliable LC-MS/MS assay for the estimation of rilpivirine in human plasma: application to a bioequivalence study and incurred sample reanalysis. Drug Test Anal 2014; 7: 290-9.
[189]
Grégoire M, Deslandes G, Renaud C, et al. A liquid chromatography-tandem mass spectrometry assay for quantification of rilpivirine and dolutegravir in human plasma. J Chromatogr B Analyt Technol Biomed Life Sci 2014; 971: 1-9.
[190]
Cozzi V, Charbe N, Baldelli S, et al. Development And Validation Of A Chromatographic Uv Method For The Simultaneous Quantification Of Dolutegravir And Rilpivirine In Human Plasma. Ther Drug Monit 2016; 38: 407-13.
[191]
Charbe N, Baldelli S, Cozzi V, Castoldi S, Cattaneo D, Clementi E. Development of HPLC-UV assay methods for the simultaneous quantification of nine antiretroviral agents in the plasma of HIV-infected patients. J Pharm Anal 2016; 6: 396-403.
[192]
Shehu-Xhilaga M, Crowe SM, Mak J. Maintenance of the Gag/Gag-Pol ratio is important for human immuno-deficiency virus type 1 RNA dimerization and viral infectivity. J Virol 2001; 75: 1834-41.
[193]
Hurst M, Faulds D. Lopinavir. Drugs 2000; 60: 1371-81.
[194]
Murphy RL, Brun S, Hicks C, et al. ABT-378/ritonavir plus stavudine and lamivudine for the treatment of antiretroviral-naive adults with HIV-1 infection: 48-week results. AIDS 2001; 15: F1-9.
[195]
La Porte CJ, Colbers EP, Bertz R, et al. Pharmacokinetics of Adjusted-Dose Lopinavir-Ritonavir Combined with Rifampin in Healthy Volunteers. Antimicrob Agents Chemother 2004; 48: 1553-60.
[196]
Leibenguth P, Le Guellec C, Besnier JM, et al. Therapeutic drug monitoring of HIV protease inhibitors using high-performance liquid chromatography with ultraviolet or photodiode array detection. Ther Drug Monit 2001; 23: 679-88.
[197]
Ray J, Pang E, Carey D. Simultaneous determination of indinavir, ritonavir and lopinavir [ABT 378] in human plasma by high-performance liquid chromatography. J Chromatogr B Analyt Technol Biomed Life Sci 2002; 775: 225-30.
[198]
Marzolini C, Béguin A, Telenti A, et al. Determination of lopinavir and nevirapine by high-performance liquid chromatography after solid-phase extraction: Application for the assessment of their transplacental passage at delivery. J Chromatogr B Analyt Technol Biomed Life Sci 2002; 774: 127-40.
[199]
Justesen US, Pedersen C, Klitgaard NA. Simultaneous quantitative determination of the HIV protease inhibitors indinavir, amprenavir, ritonavir, lopinavir, saquinavir, nelfinavir and the nelfinavir active metabolite M8 in plasma by liquid chromatography. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 783: 491-500.
[200]
Verbesselt R, Van Wijngaerden E, de Hoon J. Simultaneous determination of 8 HIV protease inhibitors in human plasma by isocratic high-performance liquid chromato-graphy with combined use of UV and fluorescence detection: Amprenavir, indinavir, atazanavir, ritonavir, lopinavir, saquinavir, nelfinavir. J Chromatogr B Analyt Technol Biomed Life Sci 2007; 845: 51-60.
[201]
Choi SO, Rezk NL, Kashuba ADM. High-performance liquid chromatography assay for the determination of the HIV-protease inhibitor tipranavir in human plasma in combination with nine other antiretroviral medications. J Pharm Biomed Anal 2007; 43: 1562-7.
[202]
Hirano A, Takahashi M, Kinoshita E, et al. High performance liquid chromatography using UV detection for the simultaneous quantification of the new non-nucleoside reverse transcriptase inhibitor etravirine [TMC-125], and 4 protease inhibitors in human plasma. Biol Pharm Bull 2010; 33: 1426-9.
[203]
D’Avolio A, Baietto L, Siccardi M, et al. An HPLC-PDA method for the simultaneous quantification of the HIV integrase inhibitor raltegravir, the new nonnucleoside reverse transcriptase inhibitor etravirine, and 11 other antiretroviral agents in the plasma of HIV-infected patients. Ther Drug Monit 2008; 30: 662-9.
[204]
Elens L, Veriter S, Di Fazio V, et al. Quantification of 8 HIV-protease inhibitors and 2 nonnucleoside reverse transcriptase inhibitors by ultra-performance liquid chromatography with diode array detection. Clin Chem 2009; 55: 170-4.
[205]
Crommentuyn KM, Rosing H, Nan-Offeringa LG, Hillebrand MJ, Huitema AD, Beijnen JH. Rapid quantification of HIV protease inhibitors in human plasma by high-performance liquid chromatography coupled with electrospray ionization tandem mass spectrometry. J Mass Spectrom 2003; 38: 157-66.
[206]
Wang PG, Wei JS, Kim G, Chang M, El-Shourbagy T. Validation and application of a high-performance liquid chromatography-tandem mass spectrometric method for simultaneous quantification of lopinavir and ritonavir in human plasma using semi-automated 96-well liquid-liquid extraction. J Chromatogr A 2006; 1130: 302-7.
[207]
DiFrancesco R, DiCenzo R, Vicente G, et al. Determination of lopinavir cerebral spinal fluid and plasma ultrafiltrate concentrations by liquid chromatography coupled to tandem mass spectrometry. J Pharm Biomed Anal 2007; 44: 1139-46.
[208]
Zhang J, Wu H, Kim E, El-Shourbagy TA. Salting-out assisted liquid/liquid extraction with acetonitrile: A new high throughput sample preparation technique for good laboratory practice bioanalysis using liquid chromatography-mass spectrometry. Biomed Chromatogr 2009; 23: 419-25.
[209]
Estrela RC, Ribeiro FS, Seixas BV, Suarez-Kurtz G. Determination of lopinavir and ritonavir in blood plasma, seminal plasma, saliva and plasma ultra-filtrate by liquid chromatography/tandem mass spectrometry detection. Rapid Commun Mass Spectrom 2008; 22: 657-64.
[210]
Myasein F, Kim E, Zhang J, Wu H, El-Shourbagy TA. Rapid, simultaneous determination of lopinavir and ritonavir in human plasma by stacking protein precipitations and salting-out assisted liquid/liquid extraction, and ultrafast LC-MS/MS. Anal Chim Acta 2009; 651: 112-6.
[211]
Notari S, Sergi M, Montesano C, et al. Simultaneous determination of lamivudine, lopinavir, ritonavir, and zidovudine concentration in plasma of HIV-infected patients by HPLC-MS/MS. IUBMB Life 2012; 64: 443-9.
[212]
Yadav M, Rao R, Kurani H, Singhal P, Goswami S, Shrivastav PS. Application of a rapid and selective method for the simultaneous determination of protease inhibitors, lopinavir and ritonavir in human plasma by UPLC-ESI-MS/MS for bioequivalence study in Indian subjects. J Pharm Biomed Anal 2009; 49: 1115-22.
[213]
Meesters RJ, Van Kampen JJ, Reedijk ML, et al. Ultrafast and high-throughput mass spectrometric assay for therapeutic drug monitoring of antiretroviral drugs in pediatric HIV-1 infection applying dried blood spots. Anal Bioanal Chem 2010; 398: 319-28.
[214]
Das Mishra T, Kurani H, Singhal P, Shrivastav PS. Simultaneous quantitation of HIV-protease inhibitors ritonavir, lopinavir and indinavir in human plasma by UPLC-ESI-MS-MS. J Chromatogr Sci 2012; 50: 625-35.
[215]
Djerada Z, Feliu C, Tournois C, et al. Validation of a fast method for quantitative analysis of elvitegravir, raltegravir, maraviroc, etravirine, tenofovir, boceprevir and 10 other antiretroviral agents in human plasma samples with a new UPLC-MS/MS technology. J Pharm Biomed Anal 2013; 86: 100-11.
[216]
Watanabe K, Varesio E, Hopfgartner G. Parallel ultra high pressure liquid chromatography-mass spectrometry for the quantification of HIV protease inhibitors using dried spot sample collection format. J Chromatogr B Analyt Technol Biomed Life Sci 2014; 965: 244-53.
[217]
Azoulay S, Nevers MC, Créminon C, et al. An enzyme immunoassay for the quantification of plasma and intracellular lopinavir in HIV-infected patients. J Immunol Methods 2004; 295: 37-48.
[218]
Burger DM, Hoetelmans RM, Koopmans PP, et al. Clinically relevant drug interactions with antiretroviral agents. Antivir Ther 1997; 2: 149-65.
[219]
Piscitelli SC, Flexner C, Minor JR, Polis MA, Masur H. Drug interactions in patients infected with human immunodeficiency virus. Clin Infect Dis 1996; 23: 685-93.
[220]
Svensson JO, Sönnerborg A, Ståhle L. Rapid and simple determination of indinavir in serum, urine, and cerebrospinal fluid using high-performance liquid chromatography. Ther Drug Monit 2000; 22: 626-9.
[221]
Dailly E, Thomas L, Kergueris MF, Jolliet P, Bourin M. High-performance liquid chromatographic assay to determine the plasma levels of HIV-protease inhibitors [amprenavir, indinavir, nelfinavir, ritonavir and saquinavir] and the non-nucleoside reverse transcriptase inhibitor [nevirapine] after liquid-liquid extraction. J Chromatogr B Analyt Technol Biomed Life Sci 2001; 758: 129-35.
[222]
Sarasa-Nacenta M, López-Púa Y, Mallolas J, Blanco JL, Gatell JM, Carné X. Simultaneous determination of the HIV-protease inhibitors indinavir, amprenavir, ritonavir, saquinavir and nelfinavir in human plasma by reversed-phase high-performance liquid chromatography. J Chromatogr B Biomed Sci Appl 2001; 757: 325-32.
[223]
Fizzano MR, Valvo L, Dupuis ML, Mennella V, Cianfriglia M. LC determination of Indinavir in biological matrices with electrochemical detection. J Pharm Biomed Anal 2000; 22: 307-14.
[224]
Jayewardene AL, Kearney B, Stone JA, Gambertoglio JG, Aweeka FT. An LC-MS-MS method for the determination of indinavir, an HIV-1 protease inhibitor, in human plasma. J Pharm Biomed Anal 2001; 25: 309-17.
[225]
Justesen US, Pedersen C, Klitgaard NA. Simultaneous quantitative determination of the HIV protease inhibitors indinavir, amprenavir, ritonavir, lopinavir, saquinavir, nelfinavir and the nelfinavir active metabolite M8 in plasma by liquid chromatography. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 783: 491-500.
[226]
Langmann P, Klinker H, Schirmer D, Zilly M, Bienert A, Richter E. High-performance liquid chromatographic method for the simultaneous determination of HIV-1 protease inhibitors indinavir, saquinavir and ritonavir in plasma of patients during highly active antiretroviral therapy. J Chromatogr B Biomed Sci Appl 1999; 735: 41-50.
[227]
Hurwitz SJ, Schinazi RF. Development of a pharmacodynamic model for HIV treatment with nucleoside reverse transcriptase and protease inhibitors. Antiviral Res 2002; 56: 115-27.
[228]
Woolf E, Haddix HM, Matuszewski B. Determination of an in vivo metabolite of a human immunodeficiency virus protease-inhibitor in human plasma by high-performance liquid chromatography with tandem mass spectrometry. J Chromatogr A 1997; 762: 311-9.
[229]
Armbruster C, Vorbach H, Steindl F, El Menyawi I. Intracellular concentration of the HIV protease inhibitors indinavir and saquinavir in human endothelial cells. J Antimicrob Chemother 2001; 47: 487-90.
[230]
Perry CM, Benfield P. Nelfinavir. Drugs 1997; 54: 81-7.
[231]
Lillibridge JH, Liang BH, Kerr BM, et al. Characterization of the selectivity and mechanism of human cytochrome P450 inhibition by the human immunodeficiency virus-protease inhibitor nelfinavir mesylate. Drug Metab Dispos 1998; 26: 609-16.
[232]
Wu EY, Wilkinson JM, Naret DG, et al. High-performance liquid chromatographic method for the determination of nelfinavir, a novel HIV-1 protease inhibitor, in human plasma. J Chromatogr B Biomed Sci Appl 1997; 695: 373-80.
[233]
Lamotte C, Peytavin G, Farinotti R. Determination of nelfinavir, a potent HIV protease inhibitor, and its active metabolite M8 in human plasma by high-performance liquid chromatography with photodiode-array detection. J Chromatogr B Biomed Sci Appl 1999; 735: 159-70.
[234]
Janoly A, Bleyzac N, Favetta P, et al. Simple and rapid high-performance liquid chromatographic method for nelfinavir, M8 nelfinavir metabolite, ritonavir and saquinavir assay in plasma. J Chromatogr B Analyt Technol Biomed Life Sci 2002; 780: 155-60.
[235]
Lawrence CC, Thomas A, Shastri S, et al. An Automated Microparticle Agglutination Immunoassay For The Therapeutic Drug Monitoring Of Nelfinavir. Ther Drug Monit 2005; 27: 232.
[236]
Uglietti A, Genco F, Donadel E, et al. Evaluation of Elisa test for therapeutic monitoring of Nelfinavir in HIV-positive patients. New Microbiol 2007; 30: 318-20.
[237]
Sadler BM, Stein DS. Clinical pharmacology and pharmacokinetics of amprenavir. Ann Pharmacother 2002; 36: 102-18.
[238]
Polk RE, Crouch MA, Israel DS, et al. Pharmacokinetic interaction between ketoconazole and amprenavir after single doses in healthy men. Pharmacotherapy 1999; 19: 1378-84.
[239]
Murphy RL, Gulick RM, DeGruttola V, et al. Treatment with amprenavir alone or amprenavir with zidovudine and lamivudine in adults with human immunodeficiency virus infection. AIDS Clinical Trials Group 347 Study Team. J Infect Dis 1999; 179: 808-16.
[240]
Eron JJ, Smeaton LM, Fiscus SA, et al. The effects of protease inhibitor therapy on human immunodeficiency virus type 1 levels in semen. [AIDS clinical trials group protocol 850]. J Infect Dis 2000; 181: 1622-8.
[241]
Polli JW, Jarrett JL, Studenberg SD, et al. Role of P-glycoprotein on the CNS disposition of amprenavir [141W94], an HIV protease inhibitor. Pharm Res 1999; 16: 1206-12.
[242]
Pereira AS, Kenney KB, Cohen MS, Eron JJ, Tidwell RR, Dunn JA. Determination of amprenavir, a HIV-1 protease inhibitor, in human seminal plasma using high-performance liquid chromatography-tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2002; 766: 307-17.
[243]
Sparidans RW, Hoetelmans RM, Beijnen JH. Sensitive liquid chromatographic assay for amprenavir, a human immunodeficiency virus protease inhibitor, in human plasma, cerebrospinal fluid and semen. J Chromatogr B Biomed Sci Appl 2000; 742: 185-92.
[244]
Bastiani E, Benedetti F, Berti F, et al. Development and evaluation of an immunoassay for the monitoring of the anti-HIV drug amprenavir. J Immunol Methods 2007; 325: 35-41.
[245]
Chi J, Jayewardene AL, Stone JA, Motoya T, Aweeka FT. Simultaneous determination of five HIV protease inhibitors nelfinavir, indinavir, ritonavir, saquinavir and amprenavir in human plasma by LC/MS/MS. J Pharm Biomed Anal 2002; 30: 675-84.
[246]
Dickinson L, Robinson L, Tjia J, Khoo S, Back D. Simultaneous determination of HIV protease inhibitors amprenavir, atazanavir, indinavir, lopinavir, nelfinavir, ritonavir and saquinavir in human plasma by high-performance liquid chromatography-tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2005; 829: 82-90.
[247]
Koal T, Sibum M, Koster E, Resch K, Kaever V. Direct and fast determination of antiretroviral drugs by automated online solid-phase extraction-liquid chromatography-tandem mass spectrometry in human plasma. Clin Chem Lab Med 2006; 44: 299-305.
[248]
Naxing Xu R, Fan L, Kim GE, El-Shourbagy TA. A monolithic-phase based on-line extraction approach for determination of pharmaceutical components in human plasma by HPLC-MS/MS and a comparison with liquid-liquid extraction. J Pharm Biomed Anal 2006; 40: 728-36.
[249]
Martin J, Deslandes G, Dailly E, et al. A liquid chromatography-tandem mass spectrometry assay for quantification of nevirapine, indinavir, atazanavir, amprenavir, saquinavir, ritonavir, lopinavir, efavirenz, tipranavir, darunavir and maraviroc in the plasma of patients infected with HIV. J Chromatogr B Analyt Technol Biomed Life Sci 2009; 877: 3072-82.
[250]
Fitzsimmons ME, Collins JM. Selective biotransformation of the human immunodeficiency virus protease inhibitor saquinavir by human small-intestinal cytochrome P4503A4: Potential contribution to high first-pass metabolism. Drug Metab Dispos 1997; 25: 256-66.
[251]
Profit L, Eagling VA, Back DJ. Modulation of P-glycoprotein function in human lymphocytes and Caco-2 cell monolayers by HIV-1 protease inhibitors. AIDS 1999; 13: 1623-7.
[252]
Alsenz J, Steffen H, Alex R. Active apical secretory efflux of the HIV protease inhibitors saquinavir and ritonavir in Caco-2 cell monolayers. Pharm Res 1998; 15: 423-8.
[253]
Hsu A, Granneman GR, Cao G, et al. Pharmacokinetic interactions between two human immunodeficiency virus protease inhibitors, ritonavir and saquinavir. Clin Pharmacol Ther 1998; 63: 453-64.
[254]
Kilby JM, Sfakianos G, Gizzi N, et al. Safety and pharmacokinetics of once-daily regimens of soft-gel capsule saquinavir plus minidose ritonavir in human immunodeficiency virus-negative adults. Antimicrob Agents Chemother 2000; 44: 2672-8.
[255]
Ribera E, Azuaje C, Lopez RM, et al. Pharmacokinetic interaction between rifampicin and the once-daily combination of saquinavir and low-dose ritonavir in HIV-infected patients with tuberculosis. J Antimicrob Chemother 2007; 59: 690-7.
[256]
Perry CM, Noble S. Saquinavir soft-gel capsule formulation. A review of its use in patients with HIV infection. Drugs 1998; 55: 461-86.
[257]
Kupferschmidt HH, Fattinger KE, Ha HR, Follath F, Krähenbühl S. Grapefruit juice enhances the bioavailability of the HIV protease inhibitor saquinavir in man. Br J Clin Pharmacol 1998; 45: 355-9.
[258]
Knebel NG, Sharp SR, Madigan MJ. Rapid quantitative determination of a collagenase inhibitor and its major metabolite by on-line liquid chromatography with ionspray tandem mass spectrometric detection. J Chromatogr B Biomed Appl 1995; 673: 213-22.
[259]
Ha HR, Follath F, Bloemhard Y, Krähenbühl S. Determination of saquinavir in human plasma by high-performance liquid chromatography. J Chromatogr B Analyt Technol Biomed Life Sci 1997; 694: 427-33.
[260]
Frappier S, Breilh D, Diarte E, et al. Simultaneous determination of ritonavir and saquinavir, two human immunodeficiency virus protease inhibitors, in human serum by high-performance liquid chromatography. J Chromatogr B Analyt Technol Biomed Life Sci 1998; 714: 384-9.
[261]
Campanero M, Escolar M, Arangoa M, Sdaba B, Azanza JR. Development of a chromatographic method for the determination of saquinavir in plasma samples of HIV patients. Biomed Chromatogr 2002; 16: 7-12.
[262]
Kim AE, Dintaman JM, Waddell DS, Silverman JA. Saquinavir, an HIV protease inhibitor, is transported by P-glycoprotein. J Pharmacol Exp Ther 1998; 286: 1439-45.
[263]
Ucpinar SD, Stavchansky S. Quantitative determination of saquinavir from Caco-2 cell monolayers by HPLC-UV. Biomed Chromatogr 2003; 17: 21-5.
[264]
Van Heeswijk RP, Hoetelmans RM, Harms R, et al. Simultaneous quantitative determination of the HIV protease inhibitors amprenavir, indinavir, nelfinavir, ritonavir and saquinavir in human plasma by ion-pair high-performance liquid chromatography with ultraviolet detection. J Chromatogr B Biomed Sci Appl 1998; 719: 159-68.
[265]
Remmel RP, Kawle SP, Weller D, Fletcher CV. Simultaneous HPLC assay for quantification of indinavir, nelfinavir, ritonavir, and saquinavir in human plasma. Clin Chem 2000; 46: 73-81.
[266]
Wiltshire HR, Wiltshire BG, Clarke AF, Worth E, Prior KJ, Tjia JF. Chromatographic and immunochemical approaches to the analysis of the HIV protease inhibitor saquinavir in plasma. Anal Biochem 2000; 281: 105-14.
[267]
Knebel NG, Sharp SR, Madigan MJ. Quantification of the anti-HIV drug saquinavir by high-speed on-line high-performance liquid chromatography/tandem mass spectrometry. J Mass Spectrom 1995; 30: 1149-56.
[268]
Burhenne J, Riedel KD, Martin-Facklam M, Mikus G, Haefeli WE. Highly sensitive determination of saquinavir in biological samples using liquid chromatography-tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 784: 233-42.
[269]
Lea AP, Faulds D. Ritonavir. Drugs 1996; 52: 541-8.
[270]
Eagling VA, Back DJ, Barry MG. Differential inhibition of cytochrome P450 isoforms by the protease inhibitors, ritonavir, saquinavir and indinavir. Br J Clin Pharmacol 1997; 44: 190-4.
[271]
Hsu A, Granneman GR, Witt G, et al. Multiple-dose pharmacokinetics of ritonavir in human immunodeficiency virus-infected subjects. Antimicrob Agents Chemother 1997; 41: 898-905.
[272]
Marsh KC, Eiden E, McDonald E. Determination of ritonavir, a new HIV protease inhibitor, in biological samples using reversed-phase high-performance liquid chromatography. J Chromatogr B Biomed Sci Appl 1997; 704: 307-13.
[273]
Hoetelmans RM, van Essenberg M, Profijt M, Meenhorst PL, Mulder JW, Beijnen JH. High-performance liquid chromatographic determination of ritonavir in human plasma, cerebrospinal fluid and saliva. J Chromatogr B Analyt Technol Biomed Life Sci 1998; 705: 119-26.
[274]
Granda BW, Giancarlo GM, Von Moltke LL, Greenblatt DJ. Analysis of ritonavir in plasma/serum and tissues by high-performance liquid chromatography. J Pharmacol Toxicol Methods 1998; 40: 235-9.
[275]
Penzak SR, Lawhorn WD, Gubbins PO. Rapid and sensitive high-performance liquid chromatographic method for the determination of ritonavir in human plasma. Int J Clin Pharmacol Ther 2001; 39: 400-5.
[276]
Elens L, Veriter S, Yombi JC, et al. Validation and clinical application of a high performance liquid chromatography tandem mass spectrometry [LC-MS/MS] method for the quantitative determination of 10 anti-retrovirals in human peripheral blood mononuclear cells. J Chromatogr B Analyt Technol Biomed Life Sci 2009; 877: 1805-14.
[277]
Akeb F, Ferrua B, Creminon C, et al. Quantification of plasma and intracellular levels of the HIV protease inhibitor ritonavir by competitive ELISA. J Immunol Methods 2002; 263: 1-9.
[278]
Titier K, Lagrange F, Péhourcq F, Edno-Mcheik L, Moore N, Molimard M. High-performance liquid chromatographic method for the simultaneous determination of the six HIV-protease inhibitors and two non-nucleoside reverse transcriptase inhibitors in human plasma. Ther Drug Monit 2002; 24: 417-24.
[279]
Wood R. Atazanavir: its role in HIV treatment. Expert Rev Anti Infect Ther 2008; 6: 785-96.
[280]
Busti AJ, Hall RG, Margolis DM. Atazanavir for the treatment of human immunodeficiency virus infection. Pharmacotherapy 2004; 24: 1732-47.
[281]
Schuster A, Burzawa S, Jemal M, Loizillon E, Couerbe P, Whigan D. Quantitative determination of the HIV protease inhibitor atazanavir [ BMS-232632 ] in human plasma by liquid chromatography - tandem mass spectrometry following automated solid-phase extraction. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 788: 377-86.
[282]
Jemal M, Rao S, Gatz M, Whigan D. Liquid chromatography-tandem mass spectrometric quantitative determination of the HIV protease inhibitor atazanavir [ BMS-232632 ] in human peripheral blood mononuclear cells [ PBMC ]: practical approaches to PBMC preparation and PBMC assay design for hig. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 795: 273-89.
[283]
Colombo S, Guignard N, Marzolini C, Telenti A, Biollaz J, Decosterd LA. Determination of the new HIV-protease inhibitor atazanavir by liquid chromatography after solid-phase extraction. J Chromatogr B Analyt Technol Biomed Life Sci 2004; 810: 25-34.
[284]
Sparidans RW, Dost F, Crommentuyn KM, Huitema AD, Schellens JH, Beijnen JH. Liquid chromatographic assay for the protease inhibitor atazanavir in plasma. Biomed Chromatogr 2006; 20: 72-6.
[285]
Cateau E, Tournier N, Dupuis A, Venisse N. Determination of atazanavir in human plasma using solid-phase extraction and high-performance liquid chromatography. J Pharm Biomed Anal 2005; 39: 791-5.
[286]
Loregian A, Pagni S, Ballarin E, Sinigalia E, Giuseppe S, Pal G. Simple determination of the HIV protease inhibitor atazanavir in human plasma by high-performance liquid chromatography with UV detection. J Pharm Biomed Anal 2006; 42: 500-5.
[287]
Cattaneo D, Maggiolo F, Ripamonti D, Perico N. Determination of atazanavir in human plasma by high-performance liquid chromatography with UV detection. J Chromatogr Sci 2008; 46: 485-9.
[288]
Müller AC, Kanfer I. An efficient HPLC method for the quantitative determination of atazanavir in human plasma suitable for bioequivalence and pharmacokinetic studies in healthy human subjects. J Pharm Biomed Anal 2010; 53: 113-8.
[289]
Van Heeswijk RP, Veldkamp AI, Mulder JW, et al. Saliva as an alternative body fluid for therapeutic drug monitoring of the nonnucleoside reverse transcription inhibitor nevirapine. Ther Drug Monit 2001; 23: 255-8.
[290]
Fayet A, Béguin A, De Tejada BM, et al. Determination of unbound antiretroviral drug concentrations by a modified ultrafiltration method reveals high variability in the free fraction. Ther Drug Monit 2008; 30: 511-22.
[291]
Leschziner GD, Andrew T, Pirmohamed M, Johnson MR. ABCB1 genotype and PGP expression, function and therapeutic drug response: a critical review and recommendations for future research. Pharmacogenomics J 2007; 7: 154-79.
[292]
Schinkel AH, Jonker JW. Mammalian drug efflux transporters of the ATP binding cassette [ABC] family: An overview. Adv Drug Deliv Rev 2012; 551: 138-53.
[293]
Elens L, Veriter S, Di Fazio V, et al. Quantification of 8 HIV-Protease Inhibitors and 2 Nonnucleoside Reverse Transcriptase Inhibitors by Ultra-Performance Liquid Chromatography with Diode Array Detection. Clin Chem 2009; 55: 170-4.
[294]
Crommentuyn KM, Rosing H, Hillebrand MJ, Huitema AD, Beijnen JH. Simultaneous quantification of the new HIV protease inhibitors atazanavir and tipranavir in human plasma by high-performance liquid chromatography coupled with electrospray ionization tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2004; 804: 359-67.
[295]
De Meyer S, Azijn H, Surleraux D, et al. TMC114, a novel human immunodeficiency virus type 1 protease inhibitor active against protease inhibitor-resistant viruses, including a broad range of clinical isolates. Antimicrob Agents Chemother 2005; 49: 2314-21.
[296]
Koh Y, Nakata H, Maeda K, et al. Novel bis-tetrahydrofuranylurethane-containing nonpeptidic protease inhibitor [PI] UIC-94017 [TMC114] with potent activity against multi-PI-resistant human immunodeficiency virus in vitro. Antimicrob Agents Chemother 2003; 47: 3123-9.
[297]
Rotty J, Hoy J. New therapeutic agents in the management of HIV: An overview of darunavir for clinicians. Sex Health 2008; 5: 235-41.
[298]
Molina JM, Hill A. Darunavir [TMC114]: a new HIV-1 protease inhibitor. Expert Opin Pharmacother 2007; 8: 1951-64.
[299]
Vermeir M, Lachau-Durand S, Mannens G, Cuyckens F, Van Hoof B, Raoof A. Absorption, metabolism, and excretion of darunavir, a new protease inhibitor, administered alone and with low-dose ritonavir in healthy subjects. Drug Metab Dispos 2009; 37: 809-20.
[300]
Yilmaz A, Izadkhashti A, Price RW, et al. Darunavir concentrations in cerebrospinal fluid and blood in HIV-1-infected individuals. AIDS Res Hum Retroviruses 2009; 25: 457-61.
[301]
Goldwirt L, Chhun S, Rey E, et al. Quantification of darunavir [TMC114] in human plasma by high-performance liquid chromatography with ultra-violet detection. J Chromatogr B Analyt Technol Biomed Life Sci 2007; 857: 327-31.
[302]
Sekar VJ, Lefebvre E, De Paepe E, et al. Pharmacokinetic interaction between darunavir boosted with ritonavir and omeprazole or ranitidine in human immunodeficiency virus-negative healthy volunteers. Antimicrob Agents Chemother 2007; 51: 958-61.
[303]
Gupta A, Singhal P, Shrivastav PS, Sanyal M. Application of a validated ultra performance liquid chromatography-tandem mass spectrometry method for the quantification of darunavir in human plasma for a bioequivalence study in Indian subjects. J Chromatogr B Analyt Technol Biomed Life Sci 2011; 879: 2443-53.
[304]
Flexner C, Bate G, Kirkpatrick P. Tipranavir. Nat Rev Drug Discov 2005; 4: 955-6.
[305]
King JR, Acosta EP. Tipranavir: A novel nonpeptidic protease inhibitor of HIV. Clin Pharmacokinet 2006; 45: 665-82.
[306]
Vourvahis M, Kashuba AD. Mechanisms of pharmacokinetic and pharmacodynamic drug interactions associated with ritonavir-enhanced tipranavir. Pharmacotherapy 2007; 27: 888-909.
[307]
Keil K, Difrancesco R, Morse GD. Determination of tipranavir in human plasma by reverse phase liquid chromatography with UV detection using photodiode array. Ther Drug Monit 2006; 28: 512-6.
[308]
Giraud E, Rey E, Treluyer JM, Pons G, Jullien V. Quantification of tipranavir in human plasma by high-performance liquid chromatography with UV detection. J Chromatogr B Analyt Technol Biomed Life Sci 2006; 830: 86-90.
[309]
Dailly E, Reliquet V, Victorri-Vigneau C, Raffi F, Jolliet P. A simple high performance liquid chromatography assay for monitoring plasma concentrations of tipranavir in HIV infected patients. J Chromatogr B Analyt Technol Biomed Life Sci 2006; 832: 317-20.
[310]
Colombo S, Béguin A, Marzolini C, Telenti A, Biollaz J, Decosterd LA. Determination of the novel non-peptidic HIV-protease inhibitor tipranavir by HPLC-UV after solid-phase extraction. J Chromatogr B Analyt Technol Biomed Life Sci 2006; 832: 138-43.
[311]
D’Avolio A, Sciandra M, Siccardi M, et al. A simple and sensitive assay for determining plasma tipranavir concentration in the clinical setting by new HPLC method. J Chromatogr B Analyt Technol Biomed Life Sci 2007; 848: 374-8.
[312]
Langmann P, Heinz W, Klinker H, et al. High performance liquid chromatographic method for the determination of HIV-1 protease inhibitor tipranavir in plasma of patients during highly active antiretroviral therapy. Eur J Med Res 2008; 13: 52-8.
[313]
Tilton JC, Doms RW. Entry inhibitors in the treatment of HIV-1 infection. Antiviral Res 2010; 85: 91-100.
[314]
Haqqani AA, Tilton JC. Entry inhibitors and their use in the treatment of HIV-1 infection. Antiviral Res 2013; 98: 158-70.
[315]
Stoiber H, Ebenbichler C, Schneider R, Janatova J, Dierich MP. Interaction of several complement proteins with gp120 and gp41, the two envelope glycoproteins of HIV-1. AIDS 1995; 9: 19-26.
[316]
Ostrowski MA, Justement SJ, Catanzaro A, et al. Expression of chemokine receptors CXCR4 and CCR5 in HIV-1-infected and uninfected individuals. J Immunol 1998; 161: 3195-201.
[317]
Chen RY, Kilby JM, Saag MS. Enfuvirtide. Expert Opin Investig Drugs 2002; 11: 1837-43.
[318]
Lawless MK, Hopkins S, Anwer MK. Quantitation of a 36-amino-acid peptide inhibitor of HIV-1 membrane fusion in animal and human plasma using high- performance liquid chromatography and fluorescence detection. J Chromatogr B Biomed Sci Appl 1998; 707: 213-7.
[319]
Chang D, Kolis SJ, Linderholm KH, et al. Bioanalytical method development and validation for a large peptide HIV fusion inhibitor [ Enfuvirtide, T-20 ] and its metabolite in human plasma using LC - MS / MS. J Pharm Biomed Anal 2005; 38: 487-96.
[320]
Dorr P, Westby M, Dobbs S, et al. Maraviroc [UK-427,857], a potent, orally bioavailable, and selective small-molecule inhibitor of chemokine receptor CCR5 with broad-spectrum anti-human immunodeficiency virus type 1 activity. Antimicrob Agents Chemother 2005; 49: 4721-32.
[321]
Walker DK, Abel S, Comby P, Muirhead GJ, Nedderman ANR, Smith DA. Species differences in the disposition of the CCR5 antagonist, UK-427,857, a new potential treatment for HIV. Drug Metab Dispos 2005; 33: 587-95.
[322]
Hyland R, Dickins M, Collins C, Jones H, Jones B. Maraviroc: In vitro assessment of drug-drug interaction potential. Br J Clin Pharmacol 2008; 66: 498-507.
[323]
Notari S, Tommasi C, Nicastri E, et al. Simultaneous Determination of Maraviroc and Raltegravir in Human Plasma by HPLC-UV. IUBMB Life 2009; 61: 470-5.
[324]
Avolio AD, Simiele M, Baietto L, et al. A Validated High-Performance Liquid Chromatography-Ultraviolet Method for Quantification of the CCR5 Inhibitor Maraviroc in Plasma of HIV-Infected Patients. Ther Drug Monit 2010; 32: 86-92.
[325]
Takahashi M, Hirano A, Okubo N, Kinoshita E, Nomura T, Kaneda T. Development and application of a simple LC-MS method for the determination of plasma maraviroc concentrations. J Med Invest 2010; 57: 245-50.
[326]
Brewer E, Felix T, Clarke P, Muirhead D. An LC-MS-MS method for quantitative determination of maraviroc [ UK-427, 857 ] in human plasma, urine and cerebrospinal fluid. Biomed Chromatogr 2010; 24: 1316-23.
[327]
Simiele M, Baietto L, Audino A, et al. A validated HPLC-MS method for quantification of the CCR5 inhibitor maraviroc in HIV+ human plasma. J Pharm Biomed Anal 2014; 94: 65-70.
[328]
Pommier Y, Johnson AA, Marchand C. Integrase inhibitors to treat HIV/AIDS. Nat Rev Drug Discov 2005; 4: 236-48.
[329]
Williams DI, Churchill DD, Anderson PJ, et al. British HIV Association guidelines for the treatment of HIV-1-positive adults with antiretroviral therapy 2012. HIV Med 2012; 13: 1-85.
[330]
Clumeck N, Pozniak A, Raffi F. European AIDS Clinical Society [EACS] guidelines for the clinical management and treatment of HIV-infected adults. HIV Med 2008; 9: 65-71.
[331]
Kassahun K, McIntosh I, Cui D, et al. Metabolism and disposition in humans of raltegravir [MK-0518], an anti-AIDS drug targeting the human immunodeficiency virus 1 integrase enzyme. Drug Metab Dispos 2007; 35: 1657-63.
[332]
Moss DM, Kwan WS, Liptrott NJ, et al. Raltegravir is a substrate for SLC22A6: A putative mechanism for the interaction between raltegravir and tenofovir. Antimicrob Agents Chemother 2011; 55: 879-87.
[333]
Merschman SA, Vallano PT, Wenning LA, Matuszewski BK, Woolf EJ. Determination of the HIV integrase inhibitor, MK-0518 [ raltegravir ], in human plasma using 96-well liquid-liquid extraction and HPLC-MS / MS. J Chromatogr B Analyt Technol Biomed Life Sci 2007; 857: 15-24.
[334]
Poirier J, Robidou P, Jaillon P. Quantification of the HIV-integrase inhibitor raltegravir [MK-0518] in human plasma by high-performance liquid chromatography with fluorescence detection. J Chromatogr B Analyt Technol Biomed Life Sci 2008; 867: 277-81.
[335]
Goldwirt L, Barrail-Tran A, Da Cruz M, Taburet AM, Furlan V. Quantification of raltegravir [ MK0518 ] in human plasma by high-performance liquid chromatography with photodiode array detection. J Chromatogr B Analyt Technol Biomed Life Sci 2010; 878: 456-60.
[336]
Long MC, Bennetto-hood C, Acosta EP. A sensitive HPLC - MS - MS method for the determination of raltegravir in human plasma. J Chromatogr B Analyt Technol Biomed Life Sci 2008; 867: 165-71.
[337]
Rezk NL, White N, Kashuba AD. An accurate and precise high-performance liquid chromatography method for the rapid quantification of the novel HIV integrase inhibitor raltegravir in human blood plasma after solid phase extraction. Anal Chim Acta 2008; 628: 204-13.
[338]
Ter Heine R, Hillebrand MJ, Rosing H, et al. Quantification of the HIV-integrase inhibitor raltegravir and detection of its main metabolite in human plasma, dried blood spots and peripheral blood mononuclear cell lysate by means of high-performance liquid chromatography tandem mass spectrometry. J Pharm Biomed Anal 2009; 49: 451-8.
[339]
Talameh JA, Rezk NL, Kashuba AD. Quantifying the HIV-1 integrase inhibitor raltegravir in female genital tract secretions using high-performance liquid chromatography with ultraviolet detection. J Chromatogr B Analyt Technol Biomed Life Sci 2010; 878: 92-6.
[340]
Robbins BL, Nelson SR, Fletcher CV. A novel ultrasensitive LC-MS/MS assay for quantification of intracellular raltegravir in human cell extracts. J Pharm Biomed Anal 2012; 70: 378-87.
[341]
Jourdil JF, Tonini J, Stanke-Labesque F. Simultaneous quantitation of azole antifungals, antibiotics, imatinib, and raltegravir in human plasma by two-dimensional high-performance liquid chromatography - tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2013; 920: 1-9.
[342]
Peris-vicente J, Villareal-traver M, Casas-breva I. A micellar liquid chromatography method for the quantification of abacavir, lamivudine and raltegravir in plasma. J Pharm Biomed Anal 2014; 98: 351-5.
[343]
Manzardo C, Gatell JM. Stribild® [Elvitegravir/Cobicistat/ Emtricitabine/Tenofovir Disoproxil Fumarate]: A new paradigm for HIV-1 treatment. AIDS Rev 2014; 16: 35-42.
[344]
Temesgen Z. Cobicistat, a pharmacoenhancer for HIV treatments. Drugs Today 2013; 49: 233-7.
[345]
Klibanov OM. Elvitegravir, an oral HIV integrase inhibitor, for the potential treatment of HIV infection. Curr Opin Investig Drugs 2009; 10: 190-200.
[346]
Shimura K, Kodama E, Sakagami Y, et al. Broad antiretroviral activity and resistance profile of the novel human immunodeficiency virus integrase inhibitor elvitegravir. [JTK-303/GS-9137]. J Virol 2008; 82: 764-74.
[347]
DeJesus E, Berger D, Markowitz M, et al. Antiviral activity, pharmacokinetics, and dose response of the HIV-1 integrase inhibitor GS-9137 [JTK-303] in treatment-naive and treatment-experienced patients. J Acquir Immune Defic Syndr 2006; 43: 1-5.
[348]
Ramanathan S, Kakuda TN, Mack R, West S, Kearney BP. Pharmacokinetics of elvitegravir and etravirine following coadministration of ritonavir-boosted elvitegravir and etravirine. Antivir Ther 2008; 13: 1011-7.
[349]
Mathias AA, Hinkle J, Shen G, et al. Effect of ritonavir-boosted tipranavir or darunavir on the steady-state pharmacokinetics of elvitegravir. J Acquir Immune Defic Syndr 2008; 49: 156-62.
[350]
Cohen C, Elion R, Ruane P, et al. Randomized, phase 2 evaluation of two single-tablet regimens elvitegravir/ cobicistat/emtricitabine/tenofovir disoproxil fumarate versus efavirenz/emtricitabine/tenofovir disoproxil fumarate for the initial treatment of HIV infection. AIDS 2011; 25: F7-F12.
[351]
Ishikawa T, Nishino N, Kimura M, Irie S. Effects of a protein-rich drink or a standard meal on the pharmacokinetics of elvitegravir, cobicistat, emtricitabine and tenofovir in healthy japanese male subjects : a randomized, three-way crossover study. J Clin Pharmacol 2014; 54: 640-8.
[352]
Custodio JM, Wang H, Hao J, et al. Pharmacokinetics of cobicistat boosted ‐ elvitegravir administered in combination with rosuvastatin. J Clin Pharmacol 2014; 54: 649-56.
[353]
Ramanathan S, Shen G, Cheng A, Kearney BP. Pharmacokinetics of emtricitabine, tenofovir, and GS-9137 following coadministration of emtricitabine/tenofovir disoproxil fumarate and ritonavir-boosted GS-9137. J Acquir Immune Defic Syndr 2007; 45: 274-9.
[354]
Ballantyne AD, Perry CM. Dolutegravir: First Global Approval. Drugs 2013; 73: 1627-37.
[355]
Shah BM, Schafer JJ, Desimone JA. Dolutegravir: A new integrase strand transfer inhibitor for the treatment of HIV. Pharmacotherapy 2014; 34: 506-20.
[356]
Koteff J, Borland J, Chen S, et al. A phase 1 study to evaluate the effect of dolutegravir on renal function via measurement of iohexol and para-aminohippurate clearance in healthy subjects. Br J Clin Pharmacol 2013; 75: 990-6.
[357]
Letendre S, Mills A, Tashima K, et al. Distribution and antiviral activity in cerebrospinal
fluid [CSF] of the integrase inhibitor, dolutegravir [DTG]:
ING116070 Week 16 Results. Proceedings of the 20th
Conference on Retroviruses and Opportunistic Infections;
2013 March 3-6; Atlanta, GA. NATAP 2013.
[358]
Castellino S, Moss L, Wagner D, et al. Metabolism, excretion, and mass balance of the hiv-1 integrase inhibitor dolutegravir in humans. Antimicrob Agents Chemother 2013; 57: 3536-46.
[359]
Walmsley SL, Antela A, Clumeck N, et al. Dolutegravir plus abacavir-lamivudine for the treatment of HIV-1 infection. N Engl J Med 2013; 369: 1807-18.
[360]
Reese MJ, Savina PM, Generaux GT, Tracey H, Humphreys JE, Kanaoka E, et al. In vitro investigations into the roles of drug transporters and metabolizing enzymes in the disposition and drug interactions of dolutegravir, a hiv integrase inhibitor. Drug Metab Dispos 2013; 41: 353-61.
[361]
Cottrell ML, Hadzic T, Kashuba AD. Clinical pharmacokinetic, pharmacodynamic and drug-interaction profile of the integrase inhibitor dolutegravir. Clin Pharmacokinet 2013; 52: 981-94.
[362]
Kakuda TN, Schöller-Gyüre M, Hoetelmans RM. Clinical perspective on antiretroviral drug-drug interactions with the non-nucleoside reverse transcriptase inhibitor etravirine. Antivir Ther 2010; 15: 817-29.
[363]
Song I, Borland J, Chen S, et al. Effect of atazanavir and atazanavir/ritonavir on the pharmacokinetics of the next-generation HIV integrase inhibitor, S/GSK1349572. Br J Clin Pharmacol 2011; 72: 103-8.
[364]
Song I, Min SS, Borland J, et al. The effect of lopinavir/ritonavir and darunavir/ritonavir on the HIV integrase inhibitor S/GSK1349572 in healthy participants. J Clin Pharmacol 2011; 51: 237-42.
[365]
Bennetto-hood C, Tabolt G, Savina P, Acosta EP. A sensitive HPLC - MS / MS method for the determination of dolutegravir in human plasma. J Chromatogr B Analyt Technol Biomed Life Sci 2014; 946: 225-32.
[366]
Cozzi V, Charbe N, Baldelli S, et al. Development and Validation of a chromatographic ultraviolet method for the simultaneous quantification of dolutegravir and rilpivirine in human plasma. Ther Drug Monit 2016; 38: 407-13.
[367]
Poirier JM, Robidou P, Jaillon P. Simple and simultaneous determination of the hiv-protease inhibitors amprenavir, atazanavir, indinavir, lopinavir, nelfinavir, ritonavir and saquinavir plus m8 nelfinavir metabolite and the nonnucleoside reverse transcriptase inhibitors efavirenz and nevi. Ther Drug Monit 2005; 27: 186-92.
[368]
Kabra V, Agrahari V, Karthikeyan C, Trivedi P. Simultaneous quantitative determination of zidovudine and nevirapine in human plasma using isocratic, reverse phase high performance liquid chromatography. Trop J Pharm Res 2009; 8: 79-86.
[369]
Marzinke MA, Breaud A, Parsons TL, et al. The development and validation of a method using high-resolution mass spectrometry [HRMS] for the qualitative detection of antiretroviral agents in human blood. Clin Chim Acta 2014; 433: 157-68.