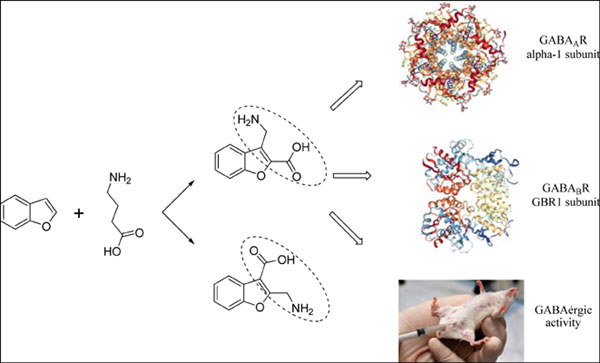
[1]
Curia, G.; Lucchi, C.; Vinet, J.; Gualtieri, F.; Marinelli, C.; Torsello, A.; Constantino, L.; Biagini, G. Pathophysiogenesis of mesial temporal lobe epilepsy: Is prevention of damage antiepileptogenic? Curr. Med. Chem., 2014, 21(6), 663-688.
[2]
Avoli, M.; D’Antuono, M.; Louvel, J.; Köhling, R.; Biagini, G.; Pumain, R.; D’Arcangelo, G.; Tancredi, V. Network and pharmacological mechanisms leading to epileptiform synchronization in the limbic system in vitro. Prog. Neurobiol., 2002, 68(3), 167-207.
[3]
Brito, V.B.; Rocha, J.B.T.; Folmer, V.; Erthal, F. Diphenyl diselenide and diphenyl ditelluride increase the latency for 4-aminopyridine-induced chemical seizure and prevent death in mice. Acta Biochim. Pol., 2009, 56(1), 125-134.
[4]
Lippi, G.; Fernandes, C.C.; Ewell, L.A.; John, D.; Romoli, B.; Curia, G.; Taylor, S.R.; Frady, E.P.; Jensen, A.B.; Liu, J.C.; Chaabane, M.M.; Belal, C.; Nathanson, J.L.; Zoli, M.; Leutgeb, J.K.; Biagini, G.; Yeo, G.W.; Chaabane, M.M. MicroRNA-101 regulates multiple developmental programs to constrain excitation in adult neural networks. Neuron, 2016, 92(6), 1337-1351.
[5]
Ye, H.; Kaszuba, S. Inhibitory or excitatory? Optogenetic interrogation of the functional roles of GABAergic interneurons in epileptogenesis. J. Biomed. Sci., 2017, 24(93), 2-9.
[6]
Dauvermann, M.R.; Lee, G.; Dawson, N. Glutamatergic regulation of cognition and functional brain connectivity: insights from pharmacological, genetic and translational schizophrenia research. Br. J. Pharmacol., 2017, 174(19), 3136-3160.
[7]
Semyanov, A.V. GABA-ergic Inhibition in the CNS: Types of GABA receptors and mechanisms of tonic GABA-mediated inhibitory action. Neurophysiology, 2002, 34(1), 71-80.
[8]
Bozzi, Y.; Provenzano, G.; Casarosa, S. Neurobiological bases of autism-epilepsy comorbidity: A focus on excitation/inhibition im-balance. Eur. J. Neurosci., 2018, 47(6), 534-548.
[9]
Meldrum, B.S. Update on the mechanism of action of antiepileptic drugs. Epilepsia, 1996, 37(S6), S4-S11.
[10]
Petersen, J.G.; Bergmann, R.; Krogsgaard-Larsen, P.; Balle, T.; Frølund, B. Probing the orthosteric binding site of GABAA receptors with heterocyclic GABA carboxylic acid bioisosteres. Neurochem. Res., 2014, 39, 1005-1015.
[11]
Saavedra-Vélez, M.V.; Correa-Basurto, J.; Matus, M.H.; Gasca-Pérez, E.; Bello, M.; Cuevas-Hernández, R.; García-Rodríguez, R.V.; Trujillo-Ferrara, J.; Ramos-Morales, F.R. Seeking potential anticonvulsant agents that target GABAA receptors using experimental and theoretical procedures. J. Comput. Aided Mol. Des., 2014, 28(12), 1217-1232.
[12]
Bergmann, R.; Kongsbak, K.; Soerensen, P.L.; Sander, T.; Balle, T. A unified model of the GABAA receptor comprising agonist and benzodiazepine binding sites. PLoS One, 2013, 8(1), 1-13.
[13]
Geng, Y.; Bush, M.; Mosyak, L.; Wang, F.; Fan, Q.R. Structural mechanism of ligand activation in human GABAB receptor. Nature, 2013, 504, 254-261.
[14]
Khanam, H. Shamsuzzaman. Bioactive benzofuran derivatives: A review. Eur. J. Med. Chem., 2015, 97, 483-504.
[15]
Radadiya, A.; Shah, A. Bioactive benzofuran derivatives: An insight on lead developments, radioligands and advances of the last decade. Eur. J. Med. Chem., 2015, 97, 356-376.
[16]
Carrër, A.; Brinet, D.; Florent, J-C.; Rousselle, P. Palladium-catalyzed direct arylation of polysubstituted benzofurans. J. Org. Chem., 2012, 77, 1316-1327.
[17]
Ohkawa, S.; Fukatsu, K.; Miki, S.; Hashimoto, T.; Sakamoto, J.; Doi, T.; Nagai, Y.; Aono, T. 5-Aminocoumarans: Dual inhibitors of lipid peroxidation and dopamine release with protective effects against central nervous system trauma and ischemia. J. Med. Chem., 1997, 40, 559-573.
[18]
Park, N-S.; Jung, Y-S.; Park, Ch.H. Seong, Ch-M.; Lim, H-J. Synthesis of Novel 2-Aryl-2-methyl-2,3-dihydrobenzofurans. Bull. Korean Chem. Soc., 2001, 22(2), 139-140.
[19]
Kraus, G.A.; Zhang, N.; Verkade, J.G.; Nagarajan, M.; Kisanga, P.B. Deprotonation of benzylic ethers using a hindered phosphazene base. A synthesis of benzofurans from Ortho-substituted benzaldehydes. Org. Lett., 2000, 2(16), 2409-2410.
[20]
Tripathi, L.; Kumar, P.; Singh, R.; Stables, J.P. Design, synthesis and anticonvulsant evaluation of novel N-(4-substituted phenyl)-2-[4-(substituted) benzylidene]-hydrazinecarbothio amides. Eur. J. Med. Chem., 2012, 47, 153-166.
[21]
Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev., 2001, 46, 3-26.
[22]
Giordano, C.; Marchiò, M.; Timofeeva, E.; Biagini, G. Neuroactive peptides as putative mediators of antiepileptic ketogenic diets. Front. Neurol., 2014, 5(63), 1-14.
[24]
Lorke, D. A new approach to practical acute toxicity testing. Arch. Toxicol., 1983, 54, 275-287.
[25]
Ramos-Morales, F.R.; Correa-Basurto, J.; Saavedra-Vélez, M.; Acosta-Hernández, M.E.; Gasca-Pérez, E.; Pérez-Palacios, A.; Trujillo-Ferrara, J. Modelo PTZ: un screening primario para el desarrollo de nuevas moléculas con actividad anticonvulsivante. Arch. Neurocien., 2012, 17(1), 45-48.
[26]
Choleris, E.; Thomas, A.W.; Kavaliers, M.; Prato, F.S. A detailed ethological analysis of the mouse open field test: effects of diazepam, chlordiazepoxide and an extremely low frequency pulsed magnetic field. Neurosci. Biobehav. Rev., 2001, 25, 235-260.
[27]
Prut, L.; Beltzung, C. The open field as a paradigm to measure the effects of drugs on anxiety-like behaviors: a review. Eur. J. Pharm., 2003, 463, 3-33.
[28]
Suzuki, T.; Tanemura, K.; Horaguchi, T.; Shimizu, T.; Sakakibara, T. Benzofuran derivatives. Part 4, synthesis of benzofurans and 2,3,4,5-tetrahydro-1-benzoxepin-3,5-diones. J. Heteroc. Chem., 1992, 29(2), 423-429.
[29]
Tseng, P.W.; Yeh, S.W.; Chou, C.H. Syntheses and pyrolyses of benzofuran analogues of α-Oxo-o-quinodimethane. A study on vinylcarbene-cyclopropene rearrangement. J. Org. Chem., 2008, 73(9), 3481-3485.
[30]
Prevysh-Kvinto, A. Synthesis and conversions of 2-carbethoxy-3-bromomethyl-7-chlorobenzofuran. Chem. Heteroc. Compd., 1994, 30(6), 661-664.
[31]
Pearson, J.R.; Porter, Q.N. Vinylindenes and some heteroanalogues in the diels-alder reaction. IX 3-Vinylbenzofuran and 1,4-naphthoquinone. Aust. J. Chem., 1991, 44, 907-917.
[32]
Cugnon de Sévricourt, M.; Robba, M. Benzofuro [2,3-d] pyridazines II. Etude des benzofuropyridazones. J. Heterocycl. Chem., 1977, 14, 777-780.
[33]
Charrier, C.; Bertrand, P. Resolution of 2,3-dihydro-benzofuran-3-ols. J. Chem. Sci., 2011, 123(4), 459-466.
[34]
Crimmins, M.T.; Martin, T.J.; Martinot, T.A. Synthesis of the bis-tetrahydropyran core of amphidinol 3. Org. Lett., 2010, 12(17), 3890-3893.
[35]
Shi, G.Q.; Dropinski, J.F.; Zhang, Y.; Santini, C.; Sahoo, S.P.; Berger, J.P.; MacNaul, K.L.; Zhou, G.; Agrawal, A.; Alvaro, R.; Cai, T.; Hernández, M.; Waright, S.D.; Moller, D.E.; Heck, J.V.; Meinke, P.T. Novel 2,3-dihydrobenzofuran-2-carboxylic acids: Highly potent and subtype-selective PPARα agonists with potent hypolipidemic activity. J. Med. Chem., 2005, 48(17), 5589-5599.
[36]
Rupprecht, K.M.; Boger, J.; Hoogsteen, K.; Nachbar, R.B.; Springer, J.P. Controlling the stereochemistry of the ring junction in hexahydrodibenzofurans. J. Org. Chem., 1991, 56, 6180-6188.