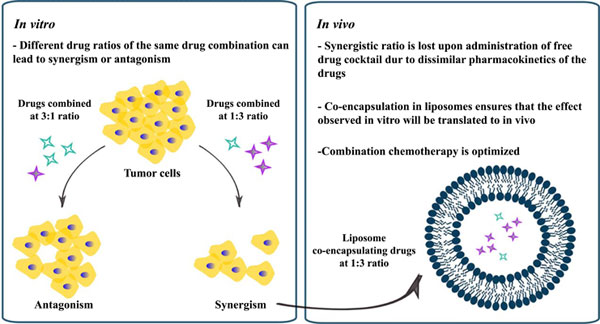
[1]
Chabner, B.A.; Roberts, T.G. Chemotherapy and the war on cancer. Nat. Rev. Cancer, 2005, 5, 65-72.
[2]
DeVita, V.T.; Chu, E. A history of cancer chemotherapy. Cancer Res., 2008, 68(21), 8643-8653.
[3]
Al-Lazikani, B.; Banerji, U.; Workman, P. Combinatorial drug therapy for cancer in the post-genomic era. Nat. Biotechnol., 2012, 30(7), 679-692.
[4]
Mayer, L.D.; Janoff, A.S. Optimizing combination chemotherapy by controlling drug ratios. Mol. Interv., 2007, 7(4), 216-223.
[5]
Mayer, L.D.; Harasym, T.O.; Tardi, P.G.; Harasym, N.L.; Shew, C.R.; Johnstone, S.A.; Ramsay, E.C.; Bally, M.B.; Janoff, A.S. Ratiometric dosing of anticancer drug combinations: Controlling drug ratios after systemic administration regulates therapeutic activity in tumor-bearing mice. Mol. Cancer Ther., 2006, 5(7), 1854-1863.
[6]
Zucker, D.; Barenholz, Y. Optimization of vincristine-topotecan combination - Paving the way for improved chemotherapy regimens by nanoliposomes. J. Control. Release, 2010, 146(3), 326-333.
[7]
Wong, M.Y.; Chiu, G.N.C. Liposome formulation of co-encapsulated vincristine and quercetin enhanced antitumor activity in a trastuzumab-insensitive breast tumor xenograft model. Nanomed-Nanotechnol, 2011, 7, 834-840.
[8]
Ciofani, T.; Harasym, T.; Juan, M.C.Y.; Mayer, L.; Cabral-Lilly, D.; Xie, S. Determination of total and encapsulated drug pharmacokinetics
for CPX-351, a nanoscale liposomal fixed molar ratio of
cytarabine-daunorubicin (Cyt:Daun). In:. Proceedings of the 102nd
Annual Meeting of the American Association for Cancer Research,
Orlando, US, Apr 2-6. 2011, 71, (8), 5464.
[9]
Shaikh, I.M.; Tan, K.B.; Chaudhury, A.; Liu, Y.; Tan, B.J.; Tan, B.M.J.; Chiu, G.N.C. Liposome co-encapsulation of synergistic combination of irinotecan anddoxorubicin for the treatment of intraperitoneally grown ovarian tumor xenograft. J. Control. Release, 2013, 172(3), 852-861.
[10]
Liu, Y.; Fang, J.; Kim, Y.J.; Wong, M.K.; Wang, P. Codelivery of doxorubicin and paclitaxel by cross-linked multilamellar liposome enables synergistic antitumor activity. Mol. Pharm., 2014, 11(5), 1651-1661.
[11]
Batist, G.; Gelmon, K.A.; Chi, K.N.; Miller, W.H., Jr; Chia, S.K.; Mayer, L.D.; Swenson, C.E.; Janoff, A.S.; Louie, A.C. Safety, pharmacokinetics, and efficacy of CPX-1 liposome injection in patients with advanced solid tumors. Clin. Cancer Res., 2009, 15(2), 692-700.
[12]
Feldman, E.J.; Kolitz, J.E.; Trang, J.M.; Liboiron, B.D.; Swenson, C.E.; Chiarella, M.T.; Mayer, L.D.; Louie, A.C.; Lancet, J.E. Pharmacokinetics of CPX-351; A nano-scale liposomal fixed molar ratio formulation of cytarabine: Daunorubicin, in patients with advanced leukemia. Leuk. Res., 2012, 36(10), 1283-1289.
[13]
Chou, T.C.; Talalay, P. Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv. Enzyme Regul., 1984, 22, 27-55.
[14]
Chou, J.H.; Chou, T.C. Dose-effect analysis with microcomputers: Quantitation of ED50, ID50, synergism, antagonism, low-dose risk, receptor ligand binding and enzyme kinetics: computer software for the IBM PC series; Elsevier-Biosoft: Cambridge, 1989.
[15]
Chou, T.C. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol. Rev., 2006, 58(3), 621-681.
[16]
Chou, T.C.; Martin, N. CompuSyn for drug combinations: PC software and user’s guide: A computer program for quantitation of synergism and antagonism in drug combinations, and the determination of IC50 and ED50 and LD50 values. ComboSyn Inc; Paramus, 2005.
[17]
Chou, T.C. Drug combination studies and their synergy quantification. Cancer Res., 2010, 70(2), 440-446.
[18]
Tardi, P.G.; Gallagher, R.C.; Johnstone, S.; Harasym, N.; Webb, M.; Bally, M.B.; Mayer, L.D. Coencapsulation of irinotecan and floxuridine into low cholesterol-containing liposomes that coordinate drug release in vivo. Biochim. Biophys. Acta, 2007, 1768(3), 678-687.
[19]
New, R.R.C. Introduction.In Liposomes: A pratical approach; New, R.R.C., Ed.; Oxford University Press: New York, 1990, pp. 1-32.
[20]
Lopes, S.C.A.; Giuberti, C.S.; Rocha, T.G.R.; Ferreira, D.S.; Leite, E.A.; Oliveira, M.C. Liposomes as Carriers of Anticancer Drugs.In Cancer Treatment - Conventional and Innovative Approaches; Rangel, L., Ed.; InTech: Vienna, 2013.
[21]
Klibanov, A.L.; Maruyama, K.; Torchilin, V.P.; Huang, L. Amphipathic polyethyleneglycols effectively prolong the circulation time of liposomes. FEBS Lett., 1990, 268(1), 235-237.
[22]
Narang, A.S.; Varia, S. Role of tumor vascular architecture in drug delivery. Adv. Drug Deliv. Rev., 2011, 63(8), 640-658.
[23]
Danhier, F.; Feron, O.; Préat, V. To exploit the tumor microenvironment: Passive and active tumor targeting of nanocarriers for anti-cancer drug delivery. J. Control. Release, 2010, 148(2), 135-146.
[24]
Yuan, F.; Leunig, M.; Huang, S.K.; Berk, D.A.; Papahadjopoulos, D.; Jain, R.K. Microvascular permeability and interstitial penetration of sterically stabilized (Stealth) liposomes in a human tumor xenograft. Cancer Res., 1994, 54(13), 3352-3356.
[25]
Torchilin, V.P. Passive and active drug targeting: Drug delivery to
tumors as an example. In:. andbook of Experimental Pharmacology;, Schafer-Korting, M., Ed.; Springer-Verlag, Berlin, 2010, 4-36/
[26]
Ferreira, D.S.; Lopes, S.C.; Franco, M.S.; Oliveira, M.C. ph-sensitive liposomes for drug delivery in cancer treatment. Therap. Del., 2013, 4(9), 1099-1123.
[27]
Harasym, T.O.; Tardi, P.G.; Harasym, N.L.; Harvie, P.; Johnstone, S.A.; Mayer, L.D. Increased preclinical efficacy of irinotecan and floxuridine coencapsulated inside liposomes is associated with tumor delivery of synergistic drug ratios. Oncol. Res., 2007, 16(8), 361-374.
[28]
Riviere, K.; Kieler-Ferguson, H.M.; Jerger, K.; Szoka, F.C., Jr Anti-tumor activity of liposome encapsulated fluoroorotic acid as a single agent and in combination with liposome irinotecan. J. Control. Release, 2011, 153(3), 288-296.
[29]
Agrawal, V.; Paul, M.K.; Mukhopadhyay, A.K. 6-Mercaptopurine and daunorubicin double drug liposomes-preparation, drug-drug interaction and characterization. J. Liposome Res., 2005, 15(3-4), 141-155.
[30]
Tardi, P.; Johnstone, S.; Harasym, N.; Xie, S.; Harasym, T.; Zisman, N.; Harvie, P.; Bermudes, D.; Mayer, L. In vivo maintenance of synergistic cytarabine: Daunorubicin ratios greatly enhances therapeutic efficacy. Leuk. Res., 2009, 33(1), 129-139.
[31]
Zhao, X.; Wu, J.; Muthusamy, N.; Byrd, J.C.; Lee, R.J. Liposomal coencapsulated fludarabine and mitoxantrone for lymphoproliferative disorder treatment. J. Pharm. Sci., 2008, 97(4), 1508-1518.
[32]
Wong, M.Y.; Chiu, G.N.C. Simultaneous liposomal delivery of quercetin and vincristine for enhanced estrogen-receptor-negative breast cancer treatment. Anticancer Drugs, 2010, 21(4), 401-410.
[33]
Abraham, S.A.; McKenzie, C.; Masin, D.; Ng, R.; Harasym, T.O.; Mayer, L.D.; Bally, M.B. In vitro and in vivo characterization of doxorubicin and vincristine coencapsulated within liposomes through use of transition metal ion complexation and pH gradient loading. Clin. Cancer Res., 2004, 10(2), 728-738.
[34]
Zucker, D.; Andriyanov, A.V.; Steiner, A.; Raviv, U.; Barenholz, Y. Characterization of PEGylatednanoliposomes co-remotely loaded with topotecan and vincristine: Relating structure and pharmacokinetics to therapeutic efficacy. J. Control. Release, 2012, 160(2), 281-289.
[35]
Batist, G.; Sawyer, M.; Gabrail, N.; Christiansen, N.; Marshall, J.L.; Spigel, D.R.; Louie, A. A multicenter, phase II study of CPX-1 liposome injection in patients (pts) with advanced colorectal cancer (CRC). J. Clin. Oncol., 2008, 26(15S), 4108.
[36]
Lim, W.S.; Tardi, P.G.; Xie, X.; Fan, M.; Huang, R.; Ciofani, T.; Harasym, T.O.; Mayer, L.D. Schedule- and dose-dependency of CPX-351, a synergistic fixed ratio cytarabine: Daunorubicin formulation, in consolidation treatment against human leukemia xenografts. Leuk. Lymphoma, 2010, 51(8), 1536-1542.
[37]
Lim, W.S.; Tardi, P.G.; Dos Santos, N.; Xie, X.; Fan, M.; Liboiron, B.D.; Huang, X.; Harasym, T.O.; Bermudes, D.; Mayer, L.D. Leukemia-selective uptake and cytotoxicity of CPX-351, a synergistic fixed-ratio cytarabine: Daunorubicin formulation, in bone marrow xenografts. Leuk. Res., 2010, 34(9), 1214-1223.
[38]
Kim, H.P.; Gerhard, B.; Harasym, T.O.; Mayer, L.D.; Hogge, D.E. Liposomal encapsulation of a synergistic molar ratio of cytarabine and daunorubicin enhances selective toxicity for acute myeloid leukemia progenitors as compared to analogous normal hematopoietic cells. Exp. Hematol., 2011, 39(7), 741-750.
[39]
Feldman, E.J.; Lancet, J.E.; Kolitz, J.E. Ritchie; E.K.; Roboz, G.J.; List, A.F.; Allen, S.L.; Asatiani, E.; Mayer, L.D.; Swenson, C.; Louie, A.C. First-in-man study of CPX-351: A liposomal carrier containing cytarabine and daunorubicin in a fixed 5:1 molar ratio for the treatment of relapsed and refractory acute Myeloid Leukemia. J. Clin. Oncol., 2011, 29(8), 979-985.
[41]
Lancet. J.E.; Cortes, J.E.; Hogge, D.E.; Tallman, M.S.; Kovacsovics, T.J.; Damon, L.E.; Komrokji, R.; Solomon, S.R.; Kolitz, J.E.; Cooper, M.; Yeager, A.M.; Louie, A.C.; Feldman, E.J. Phase 2 trial of CPX-351, a fixed 5:1 molar ratio of cytarabine/daunorubicin, vs cytarabine/daunorubicin in older adults with untreated AML. Blood, 2014, 123(21), 3239-3246.
[42]
Cortes, J.E.; Goldberg, S.L.; Feldman, E.J.; Rizzeri, D.A.; Hogge, D.E.; Larson, M.; Pigneux, A.; Recher, C.; Schiller, G.; Warzocha, K.; Kantarjian, H.; Louie, A.C.; Kolitz, J.E. Phase II, multicenter, randomized trial of CPX-351 (cytarabine: Daunorubicin) liposome injection versus intensive salvage therapy in adults with first relapse AML. Cancer, 2015, 121(2), 234-242.
[44]
Lancet. J.E.; Uy, G.L.; Cortes, J.E.; Newell, L.F.; Lin, T.L.; Ritchie, E.K.; Stuart, R.K.; Strickland, S.A.; Hogge, D.; Solomon, S.R.; Stone, R.M.; Bixby, D.L.; Kolitz, J.E.; Schiller, G.J.; Wieduwilt, M.J.; Ryan, D.H.; Hoering, A.; Chiarella, M.; Louie, A.C.; Medeiros, B.C. Final results of a phase III randomized trial of CPX-351 versus 7+3 in older patients with newly diagnosed high risk (secondary) AML. J. Clin. Oncol., 2016, 34(15), 7000.
[45]
Lancet. J.E.; Rizzieri, D.; Schiller, G.J.; Stuart, R.K.; Kolitz, J.E.; Solomon, S.R.; Newell, L.F.; Erba, H.P.; Uy, G.L.; Ryan, R.; Chiarella, M.; Louie, A.C.; Cortes, J.E. Overall Survival (OS) with CPX-351 versus 7+3 in older adults with newly diagnosed, therapy-related acute myeloid leukemia (tAML): Subgroup analysis of a phase III study. J. Clin. Oncol., 2017, 35(15), 7035.
[48]
Franco, M.S.; Oliveira, M.C. Ratiometric drug delivery using non-liposomal nanocarriers as an approach to increase efficacy and safety of combination chemotherapy. Biomed. Pharmacother., 2017, 96, 584-595.