Abstract
Background: Although conventional chemotherapy is the most common method for cancer treatment, it has several side effects such as neuropathy, alopecia and cardiotoxicity. Since the drugs are given to body systemically, normal cells are also affected, just like cancer cells. However, in recent years, targeted drug delivery has been developed to overcome these drawbacks.
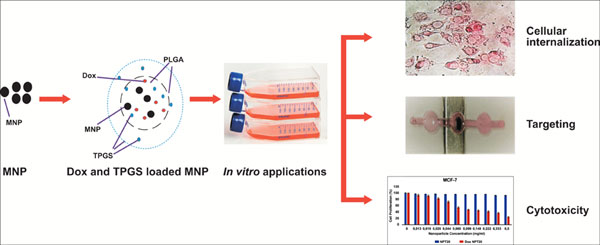
Objective: The aim of this study was targeted co-delivery of doxorubicin (Dox) which is an anticancer agent and D-α-Tocopherol polyethylene glycol 1000 succinate (vitamin E TPGS or simply TPGS) to breast cancer cells. For this purpose, Magnetic Nanoparticles (MNPs) were synthesized and coated with Oleic Acid (OA). Coated nanoparticles were encapsulated in Poly Lactic-co-Glycolic Acid (PLGA) and TPGS polymers and loaded with Dox. The Nanoparticles (NPs) were characterized by Fourier Transform Infrared (FTIR) spectroscopy, zetapotential analysis, Dynamic Light Scattering (DLS) analysis, Thermal Gravimetric Analysis (TGA) and Scanning Electron Microscope (SEM) analysis.
Results: The results showed that NPs were spherical, superparamagnetic and in the desired range for use in drug targeting. The targetability of NPs was confirmed. Moreover, TPGS and Dox loading was shown by TGA and FTIR analyses. NPs were internalized by cells and the cytotoxic effect of drug loaded NPs on sensitive (MCF-7) and drug-resistant (MCF-7/Dox) cells were examined. It was seen that the presence of TPGS increased cytotoxicity significantly. TPGS also enhanced drug loading efficiency, release rate, cellular internalization. In MCF- 7/Dox cells, the drug resistance seems to be decreased when Dox is loaded onto TPGS containing NPs.
Conclusion: This magnetic PLGA nanoparticle system is important for new generation targeted chemotherapy and could be used for breast cancer treatment after in vivo tests.
Keywords: Breast cancer, doxorubicin, MNP, targeted co-delivery, vitamin E TPGS, PLGA.