Abstract
Introduction: Dihydropyrimidine scaffold represent an important class of pharmacologically active nitrogen containing heterocyclic compounds. A wide range of molecules with dihydropyrimidine moieties have important role in medicinal chemistry on account of their potential biological activities.
Methodology: A series of 3,4-dihydropyrimidine-2(1H)-thione derivatives have been designed and synthesized in a concise way through condensation of variously substituted chalcones with thiourea in alkaline alcoholic solutions. In order to investigate their biological significance, these compounds were tested for their in vitro antimicrobial potential against various bacterial and fungal strains. Moreover, the experimental results were supported by molecular docking studies.
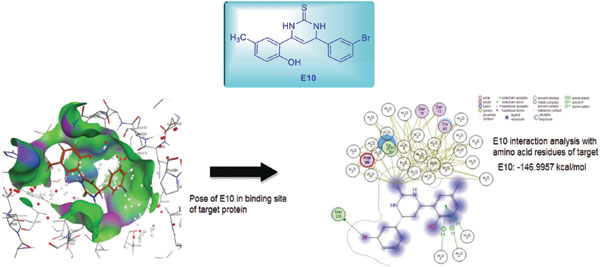
Results and Discussion: The newly synthesized compounds were characterized by the usual spectroscopic techniques In case of antibacterial activity, the compounds 5 (40.3±0.44 mm), 12 and 13 (almost 35 mm) exhibited highest zone of inhibitions against Methicillin-resistant Staphylococcus auerus (MRSA) bacterial strain as compared to the standard drug Cefixime. These compounds displayed moderate to good activities against all attempted fungal strains. In docking analysis, it has been observed that compounds 8 (-6.4017 Kcal/mol) and 10 (-6.1319 Kcal/mol) revealed significant binding affinity against penicillin binding protein (PDB ID: 1VQQ), while compounds 1 (-143.23 Kcal/mol) and 2 (-146.99 Kcal/mol) showed best activity for shikimate dehydrogenase (PDB ID: 3DON).
Conclusion: In conclusion, we have designed, synthesized and characterized an interesting series of biologically active dihydropyrimidine derivatives. Remarkably, most of the synthesized compounds were found more active against all tested bacterial strains in comparison to the standard drug Cefixime as manifested by experimental as well as theoretical results.
Keywords: Chalcones, dihydropyrimidine-2-thione, antibacterial activity, antifungal activity, molecular docking, shikimate dehydrogenase.