Abstract
Background: Among a wide range of pyridines, 3-cyanopyridines acquired a special attention due to their wide range of pharmacological activities especially the therapeutic activities. Many pharmacological drugs containing the pyridine nucleus were known in the market.
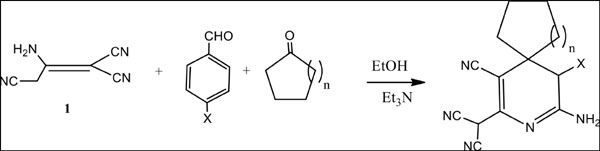
Objective: The aim of this work was to synthesize target molecules not only possess anti-tumor activities but also kinase inhibitors. To achieve this goal, our strategy was to synthesize a series of 3-cyanopyridine derivatives using 2-aminoprop-1-ene-1,1,3-tricarbonitrile (1) as the key starting material for many heterocyclization reactions.
Method: Muticoponent reactions were adopted using compound 1 to get different pyridine derivatives that were capable for different heterocyclization reactions.
Results: Antiproliferative evaluations and c-Met kinase, Pim-1 kinse inhibitions were perform where some compounds gave high activities.
Conclusion: Compounds that showed high antiprolifeative activity were tested gor c-Met-independent and the results showed that compounds 5c, 5e, 5f, 7c, 7f and 16d were more active than foretinib. The Pim-1 kinase inhibition activity of some selected compounds showed that compounds 5e and 16c were high potent to inhibit Pim-1 activity.
Keywords: 2-aminoprop-1-ene-1, 1, 3-tricarbonitrile, pyran, pyridine, kinase inhibitors, antiproliferative activity.
Anti-Cancer Agents in Medicinal Chemistry
Title:Synthesis of Pyridine and Spiropyridine Derivatives Derived from 2-aminoprop- 1-ene-1,1,3-tricarbonitrile Together with their c-Met Kinase and Antiproliferative Evaluations
Volume: 17 Issue: 14
Author(s): Rafat M. Mohareb*, Amr S. Abouzied and Nermeen S. Abbas
Affiliation:
- Department of Chemistry, Faculty of Science, Cairo University, Giza,Egypt
Keywords: 2-aminoprop-1-ene-1, 1, 3-tricarbonitrile, pyran, pyridine, kinase inhibitors, antiproliferative activity.
Abstract: Background: Among a wide range of pyridines, 3-cyanopyridines acquired a special attention due to their wide range of pharmacological activities especially the therapeutic activities. Many pharmacological drugs containing the pyridine nucleus were known in the market.
Objective: The aim of this work was to synthesize target molecules not only possess anti-tumor activities but also kinase inhibitors. To achieve this goal, our strategy was to synthesize a series of 3-cyanopyridine derivatives using 2-aminoprop-1-ene-1,1,3-tricarbonitrile (1) as the key starting material for many heterocyclization reactions.
Method: Muticoponent reactions were adopted using compound 1 to get different pyridine derivatives that were capable for different heterocyclization reactions.
Results: Antiproliferative evaluations and c-Met kinase, Pim-1 kinse inhibitions were perform where some compounds gave high activities.
Conclusion: Compounds that showed high antiprolifeative activity were tested gor c-Met-independent and the results showed that compounds 5c, 5e, 5f, 7c, 7f and 16d were more active than foretinib. The Pim-1 kinase inhibition activity of some selected compounds showed that compounds 5e and 16c were high potent to inhibit Pim-1 activity.
Export Options
About this article
Cite this article as:
Mohareb M. Rafat *, Abouzied S. Amr and Abbas S. Nermeen, Synthesis of Pyridine and Spiropyridine Derivatives Derived from 2-aminoprop- 1-ene-1,1,3-tricarbonitrile Together with their c-Met Kinase and Antiproliferative Evaluations, Anti-Cancer Agents in Medicinal Chemistry 2017; 17 (14) . https://dx.doi.org/10.2174/1871520617666170725153523
| DOI https://dx.doi.org/10.2174/1871520617666170725153523 |
Print ISSN 1871-5206 |
| Publisher Name Bentham Science Publisher |
Online ISSN 1875-5992 |
Call for Papers in Thematic Issues
Induction of cell death in cancer cells by modulating telomerase activity using small molecule drugs
Telomeres are distinctive but short stretches present at the corners of chromosomes and aid in stabilizing chromosomal makeup. Resynthesis of telomeres supported by the activity of reverse transcriptase ribonucleoprotein complex telomerase. There is no any telomerase activity in human somatic cells, but the stem cells and germ cells undergone telomerase ...read more
Role of natural compounds as anti anti-cancer agents
Cancer is considered the leading cause of worldwide mortality, accounting for nearly 10 million deaths in 2022. Cancer outcome can be improved through an appropriate screening and early detection and through an efficient clinical treatment. Chemotherapy remains an important approach in treatment o f several types of cancers, even though ...read more
Signaling and enzymatic modulators in cancer treatment
Cancer accounts for nearly 10 million deaths in 2022 and is considered the leading cause of worldwide mortality. Cancer outcome can be improved through an appropriate screening and early detection and through an efficient clinical treatment. Chemotherapy, radiotherapy and surgery are the most important approach for the treatment of several ...read more
 30
30 2
2 1
1 1
1
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
Related Articles
-
Recent Advances in Characterizing Natural Products that Regulate Autophagy
Anti-Cancer Agents in Medicinal Chemistry Inflammation-Mediating Proteases: Structure, Function in (Patho) Physiology and Inhibition
Protein & Peptide Letters Prognostic Implications of Immunohistochemical Biomarkers in Non-muscle-invasive Blad Cancer and Muscle-invasive Bladder Cancer
Mini-Reviews in Medicinal Chemistry Leucine-zipper and Sterile-α Motif Kinase (ZAK): A Potential Target for Drug Discovery
Current Medicinal Chemistry PET Tracers for Serotonin Receptors and Their Applications
Central Nervous System Agents in Medicinal Chemistry Bone Morphogenetic Protein-Smad Pathway as Drug Targets for Osteoporosis and Cancer Therapy
Endocrine, Metabolic & Immune Disorders - Drug Targets Isolation of a New Trypsin Inhibitor from the Faba Bean (Vicia faba cv. Giza 843) with Potential Medicinal Applications
Protein & Peptide Letters Improvements in Algorithms for Phenotype Inference: The NAT2 Example
Current Drug Metabolism Telocytes as a Source of Progenitor Cells in Regeneration and Repair Through Granulation Tissue
Current Stem Cell Research & Therapy GABA System as a Target for New Drugs
Current Medicinal Chemistry Telomerase Inhibitors: Potential Anticancer Agents
Mini-Reviews in Organic Chemistry Preclinical and Clinical Studies of Novel Breast Cancer Drugs Targeting Molecules Involved in Protein Kinase C Signaling, the Putative Metastasis-Suppressor Gene Cap43 and the Y-box Binding Protein-1
Current Medicinal Chemistry Role of Mechanoinsensitive Nociceptors in Painful Diabetic Peripheral Neuropathy
Current Diabetes Reviews Fetal Mesenchymal Stem Cells in Cancer Therapy
Current Stem Cell Research & Therapy Regulatable Gene Expression Systems for Gene Therapy
Current Gene Therapy The Important Roles of miR-205 in Normal Physiology, Cancers and as a Potential Therapeutic Target
Current Cancer Drug Targets Implications of Photophysical and Physicochemical Factors on Successful Application of Photodynamic Therapy
Current Pharmaceutical Design Nitric Oxide: State of the Art in Drug Design
Current Medicinal Chemistry Cancer Drug Development Using Glucose Metabolism Radiopharmaceuticals
Current Pharmaceutical Design Inhibition of Apoptosis in Pediatric Cancer by Survivin
Current Pediatric Reviews