Abstract
Background: Nimotuzumab is shown to be efficacious in advanced pancreatic cancer treatment, but its predictive marker has not been established.
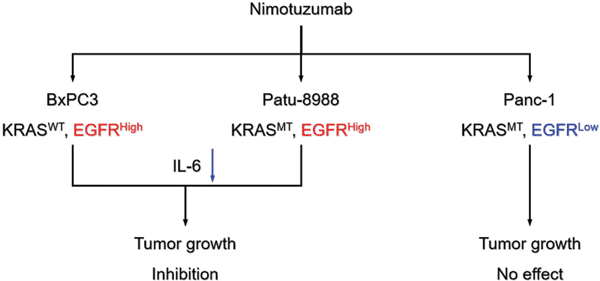
Objective: To investigate the impact of EGFR and KRAS status on antitumor efficacy of nimotuzumab and to explore its underlying mechanism. Methods: EGFR expressions of pancreatic cancer cell lines, BxPC3, Panc-1, and Patu-8988, were analyzed by Western blot and immunocytochemistry, and KRAS status was determined by gene sequencing. Anti-tumor effect of nimotuzumab were evaluated in vitro and in vivo. The expressions of related molecules in EGFR pathway and IL-6 was analyzed by Western blot, immunohistochemistry, and/or real-time PCR. Results: BxPC3 cells had wild type KRAS and high-level EGFR; Panc-1 cells had mutant KRAS (G13A) and low-level EGFR; Patu-8988 cells had mutant KRAS (G12V) and high-level EGFR. Nimotuzumab did not affect cell proliferation or apoptosis in vitro. Growth of BxPC3 and Patu-8988 xenografts were significantly inhibited by nimotuzumab, but not Panc-1 xenografts, compared with that of the control group. Expression of EGFR in BxPC3 and Patu-8988 xenografts was significantly reduced by nimotuzumab. The IL-6 expression in BxPC3 and Patu-8988 xenografts was higher than that in Panc-1 xenografts in the control group, and was significantly reduced by nimotuzumab. Conclusion: Pancreatic cancer cells with EGFR high expression were more sensitive to nimotuzumab in vivo. KRAS status had no impact on anti-tumor efficacy of nimotuzumab in pancreatic cancer cells.Keywords: Pancreatic cancer, nimotuzumab, predictive marker, EGFR, KRAS mutation.
Current Cancer Drug Targets
Title:EGFR High Expression, but not KRAS Status, Predicts Sensitivity of Pancreatic Cancer Cells to Nimotuzumab Treatment In Vivo
Volume: 17 Issue: 1
Author(s): Chenfei Zhou, Liangjun Zhu, Jun Ji, Fangmi Ding, Chao Wang, Qu Cai, Yingyan Yu, Zhenggang Zhu and Jun Zhang
Affiliation:
Keywords: Pancreatic cancer, nimotuzumab, predictive marker, EGFR, KRAS mutation.
Abstract: Background: Nimotuzumab is shown to be efficacious in advanced pancreatic cancer treatment, but its predictive marker has not been established.
Objective: To investigate the impact of EGFR and KRAS status on antitumor efficacy of nimotuzumab and to explore its underlying mechanism. Methods: EGFR expressions of pancreatic cancer cell lines, BxPC3, Panc-1, and Patu-8988, were analyzed by Western blot and immunocytochemistry, and KRAS status was determined by gene sequencing. Anti-tumor effect of nimotuzumab were evaluated in vitro and in vivo. The expressions of related molecules in EGFR pathway and IL-6 was analyzed by Western blot, immunohistochemistry, and/or real-time PCR. Results: BxPC3 cells had wild type KRAS and high-level EGFR; Panc-1 cells had mutant KRAS (G13A) and low-level EGFR; Patu-8988 cells had mutant KRAS (G12V) and high-level EGFR. Nimotuzumab did not affect cell proliferation or apoptosis in vitro. Growth of BxPC3 and Patu-8988 xenografts were significantly inhibited by nimotuzumab, but not Panc-1 xenografts, compared with that of the control group. Expression of EGFR in BxPC3 and Patu-8988 xenografts was significantly reduced by nimotuzumab. The IL-6 expression in BxPC3 and Patu-8988 xenografts was higher than that in Panc-1 xenografts in the control group, and was significantly reduced by nimotuzumab. Conclusion: Pancreatic cancer cells with EGFR high expression were more sensitive to nimotuzumab in vivo. KRAS status had no impact on anti-tumor efficacy of nimotuzumab in pancreatic cancer cells.Export Options
About this article
Cite this article as:
Zhou Chenfei, Zhu Liangjun, Ji Jun, Ding Fangmi, Wang Chao, Cai Qu, Yu Yingyan, Zhu Zhenggang and Zhang Jun, EGFR High Expression, but not KRAS Status, Predicts Sensitivity of Pancreatic Cancer Cells to Nimotuzumab Treatment In Vivo, Current Cancer Drug Targets 2017; 17 (1) . https://dx.doi.org/10.2174/1568009616666161013101657
| DOI https://dx.doi.org/10.2174/1568009616666161013101657 |
Print ISSN 1568-0096 |
| Publisher Name Bentham Science Publisher |
Online ISSN 1873-5576 |
Call for Papers in Thematic Issues
Advances in Cancer Biomarkers and Potential Drug Targets: From Diagnosis to Therapy
Cancer biomarkers play a crucial role in the diagnosis, prognosis, and treatment of cancer. They provide valuable information for cancer detection, risk assessment, treatment selection, and monitoring response to therapy. With advancements in molecular biology and high-throughput technologies, there has been an increasing interest in identifying and characterizing cancer biomarkers ...read more
Novel Therapeutic Approaches to Target Drug Resistant Tumors
With the development of disciplines such as chemical biology and molecular biology, the genes or proteins closely related to tumor occurrence and development have gradually become clear. Targeted therapies targeting these genes or proteins provide more effective methods for tumor treatment. Tumor targeted drugs generally only act on specific targets ...read more
ROLE OF IMMUNE AND GENOTOXIC RESPONSE BIOMARKERS IN TUMOR MICROENVIRONMENT IN CANCER DIAGNOSIS AND TREATMENT
Biological biomarkers have been used in medical research as an indicator of a normal or abnormal process inside the body, or of a disease. Nowadays, various researchers are in process to explore and investigate the biological markers for the early assessment of cancer. DNA Damage response (DDR) pathways and immune ...read more
Targeting the battlefield between host and tumor: basic research and clinical practice on reshaping tumor immune microenvironment
Immune system protects host against malignant tumors through effector cells and molecules. Cancer development and its response to therapy are regulated by inflammation, which either promotes or suppresses cancer progression. Chronic inflammation facilitates cancer progression and treatment resistance, whereas induction of acute inflammatory reactions often lead to anti-cancer immune responses. ...read more
 104
104 8
8
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
Related Articles
-
Natural Polyphenols and their Synthetic Analogs as Emerging Anticancer Agents
Current Drug Targets Maxillary Metastasis of Esophageal Cancer: Report of the First Case and Literature Review
Combinatorial Chemistry & High Throughput Screening Manipulation of the Immune System for Cancer Defeat: A Focus on the T Cell Inhibitory Checkpoint Molecules
Current Medicinal Chemistry The Application of Natural Products in Cancer Therapy by Targeting Apoptosis Pathways
Current Drug Metabolism Clinical Analysis Methods of Voice Disorders
Current Bioinformatics Perinatal Management of Fetal Tumors
Current Pediatric Reviews Recent Advances in PUVA Photochemotherapy and PDT for the Treatment of Cancer
Current Pharmaceutical Design Promotion of Apoptosis in Cancer Cells by Selective Purine-Derived Pharmacological CDK Inhibitors: One Outcome, Many Mechanisms
Current Pharmaceutical Design HIF-1 Inhibitors for Cancer Therapy: From Gene Expression to Drug Discovery
Current Pharmaceutical Design Phenolic Compounds: the Role of Redox Regulation in Neurodegenerative Disease and Cancer
Mini-Reviews in Medicinal Chemistry Gut-liver Axis and Microbiota in NAFLD: Insight Pathophysiology for Novel Therapeutic Target
Current Pharmaceutical Design Update on COX-2 Selective Inhibitors: Chemical Classification, Side Effects and their Use in Cancers and Neuronal Diseases
Current Topics in Medicinal Chemistry Anlotinib-Induced Hypertension: Current Concepts and Future Prospects
Current Pharmaceutical Design Molecular Docking Studies and Inhibition Properties of Some Antineoplastic Agents against Paraoxonase-I
Anti-Cancer Agents in Medicinal Chemistry Towards Understanding the Role of Cancer-Associated Inflammation in Chemoresistance
Current Pharmaceutical Design CypA: A Potential Target of Tumor Radiotherapy and/or Chemotherapy
Current Medicinal Chemistry Wnt/beta-Catenin Signaling and Small Molecule Inhibitors
Current Pharmaceutical Design Epidemiology and Pathophysiology of Chronic Cough
Current Respiratory Medicine Reviews Polysaccharide-K (PSK) in Cancer - Old Story, New Possibilities?
Current Medicinal Chemistry WNT Signaling in Stem Cell Biology and Regenerative Medicine
Current Drug Targets