Abstract
Introduction: Geraniin has many biological activities including anti-osteoporotic and antihyperglycemic efficacies.
Materials and Methods: A rapid and simple method for the determination of geraniin in rat plasma using ultra performance liquid chromatography coupled to ultraviolet detector was developed. The plasma sample, spiked with epicatechin as an internal standard, was subjected to ethyl acetate extraction prior to analysis. Chromatographic separation was performed on the HSS T3 column and monitored at a wavelength of 280 nm. The limit of detection and lower limit of quantification was 0.07 µg/mL and 0.2 µg/mL in rat plasma, respectively.
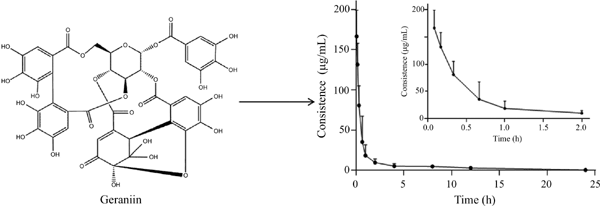
Conclusion: Good linearity was obtained in the range of 0.2 - 200 µg/mL, and the correlation coefficient was better than 0.997. The intra-day and inter-day precisions decreased 9.8%. The accuracy of QC samples ranged from 84.4% to 87.1%. The extraction recovery ranged from 88.4% to 90.3% and the matrix effect ranged from 84.4% to 87.2%. The analyte was stable in rat plasma when stored at room temperature for 12 hours, 4°C for 24 hours and -20°C for 15 days. t1/2 and t1/2 for i.v. was 0.21 ± 0.10 and 7.20 ± 2.20 h, respectively. Plasma clearance (CL) was 0.03 ± 0.02 L/h/kg and apparent volume of distribution (Vz) was 0.05 + 0.01 L/kg. The developed method was successfully applied to the pharmacokinetic study of geraniin in rats.
Keywords: Geraniin, UPLC, pharmacokinetics, epicatechin, rat, plasma.