Abstract
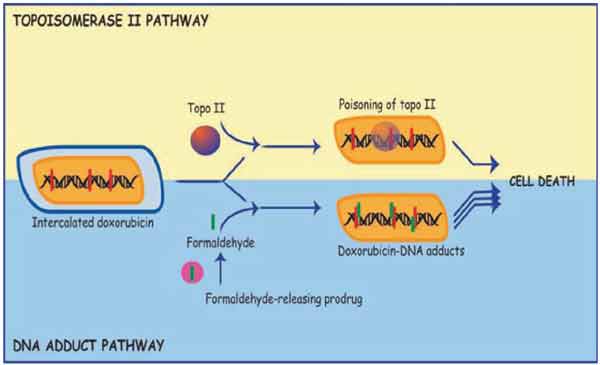
Doxorubicin has been in use as a key anticancer drug for forty years, either as a single agent or in combination chemotherapy. It functions primarily by interfering with topoisomerase II activity but in the presence of formaldehyde, it forms adducts with DNA, mainly with the exocyclic amine of guanine at GpC sites and these adducts are more cytotoxic than topoisomerase II induced damage. High levels of adducts form spontaneously from the endogenous level of formaldehyde in tumour cells (1,300 adducts per cell after a 4 hr treatment with doxorubicin), but substantially higher levels form with the addition of exogenous sources of formaldehyde, such as formaldehyde releasing prodrugs. The enhanced cytotoxicity of adducts has been confirmed in mouse models, with adduct-forming conditions resulting in much improved inhibition of tumour growth, as well as cardioprotection. Doxorubicin cardiotoxicity has been attributed to topoisomerase II poisoning, and the cardioprotection is consistent with a mechanism switch from topoisomerase II poisoning to covalent adduct formation. Although the adducts have a half-life of less than one day, a population remains as essentially permanent lesions. The capacity of doxorubicin to form adducts offers a range of potential advantages over the conventional use of doxorubicin (as a topoisomerase II poison), including: enhanced cell kill; tumour-selective activation, hence tumour-selective cell kill; decreased cardiotoxicity; decreased resistance to prolonged doxorubicin treatment. There is therefore enormous potential to improve clinical responses to doxorubicin by using conditions which favour the formation of doxorubicin-DNA adducts.
Keywords: AN-7, AN-9, Cardiotoxicity, Doxorubicin, Doxorubicin-DNA adduct, Enhanced cytotoxicity, Formaldehyde activation, Formaldehyde-releasing prodrugs, Resistance mechanisms.
Current Topics in Medicinal Chemistry
Title:Potential Therapeutic Advantages of Doxorubicin when Activated by Formaldehyde to Function as a DNA Adduct-Forming Agent
Volume: 15 Issue: 14
Author(s): Suzanne M. Cutts, Ada Rephaeli, Abraham Nudelman, Michal Ugarenko and Don R. Phillips
Affiliation:
Keywords: AN-7, AN-9, Cardiotoxicity, Doxorubicin, Doxorubicin-DNA adduct, Enhanced cytotoxicity, Formaldehyde activation, Formaldehyde-releasing prodrugs, Resistance mechanisms.
Abstract: Doxorubicin has been in use as a key anticancer drug for forty years, either as a single agent or in combination chemotherapy. It functions primarily by interfering with topoisomerase II activity but in the presence of formaldehyde, it forms adducts with DNA, mainly with the exocyclic amine of guanine at GpC sites and these adducts are more cytotoxic than topoisomerase II induced damage. High levels of adducts form spontaneously from the endogenous level of formaldehyde in tumour cells (1,300 adducts per cell after a 4 hr treatment with doxorubicin), but substantially higher levels form with the addition of exogenous sources of formaldehyde, such as formaldehyde releasing prodrugs. The enhanced cytotoxicity of adducts has been confirmed in mouse models, with adduct-forming conditions resulting in much improved inhibition of tumour growth, as well as cardioprotection. Doxorubicin cardiotoxicity has been attributed to topoisomerase II poisoning, and the cardioprotection is consistent with a mechanism switch from topoisomerase II poisoning to covalent adduct formation. Although the adducts have a half-life of less than one day, a population remains as essentially permanent lesions. The capacity of doxorubicin to form adducts offers a range of potential advantages over the conventional use of doxorubicin (as a topoisomerase II poison), including: enhanced cell kill; tumour-selective activation, hence tumour-selective cell kill; decreased cardiotoxicity; decreased resistance to prolonged doxorubicin treatment. There is therefore enormous potential to improve clinical responses to doxorubicin by using conditions which favour the formation of doxorubicin-DNA adducts.
Export Options
About this article
Cite this article as:
M. Cutts Suzanne, Rephaeli Ada, Nudelman Abraham, Ugarenko Michal and R. Phillips Don, Potential Therapeutic Advantages of Doxorubicin when Activated by Formaldehyde to Function as a DNA Adduct-Forming Agent, Current Topics in Medicinal Chemistry 2015; 15 (14) . https://dx.doi.org/10.2174/1568026615666150413154512
| DOI https://dx.doi.org/10.2174/1568026615666150413154512 |
Print ISSN 1568-0266 |
| Publisher Name Bentham Science Publisher |
Online ISSN 1873-4294 |
Call for Papers in Thematic Issues
Chemistry Based on Natural Products for Therapeutic Purposes
The development of new pharmaceuticals for a wide range of medical conditions has long relied on the identification of promising natural products (NPs). There are over sixty percent of cancer, infectious illness, and CNS disease medications that include an NP pharmacophore, according to the Food and Drug Administration. Since NP ...read more
Current Trends in Drug Discovery Based on Artificial Intelligence and Computer-Aided Drug Design
Drug development discovery has faced several challenges over the years. In fact, the evolution of classical approaches to modern methods using computational methods, or Computer-Aided Drug Design (CADD), has shown promising and essential results in any drug discovery campaign. Among these methods, molecular docking is one of the most notable ...read more
Drug Discovery in the Age of Artificial Intelligence
In the age of artificial intelligence (AI), we have witnessed a significant boom in AI techniques for drug discovery. AI techniques are increasingly integrated and accelerating the drug discovery process. These developments have not only attracted the attention of academia and industry but also raised important questions regarding the selection ...read more
From Biodiversity to Chemical Diversity: Focus of Flavonoids
Flavonoids are the largest group of polyphenols, plant secondary metabolites arising from the essential aromatic amino acid phenylalanine (or more rarely from tyrosine) via the phenylpropanoid pathway. The flavan nucleus is the basic 15-carbon skeleton of flavonoids (C6-C3-C6), which consists of two phenyl rings (A and B) and a heterocyclic ...read more
 49
49 5
5 1
1
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
- Announcements
Related Articles
-
Targeting Apelinergic System in Cardiometabolic Disease
Current Drug Targets Respiratory Syncytial Virus (RSV) Prevention and Treatment: Past, Present, and Future
Cardiovascular & Hematological Agents in Medicinal Chemistry Modulation of k-Ras Signaling by Natural Products
Current Medicinal Chemistry Potential Role of Rho Kinase Inhibitors in Combating Diabetes-Related Complications Including Diabetic Neuropathy-A Review
Current Diabetes Reviews Impact of Aging on the Angiogenic Potential of the Myocardium: Implications for Angiogenic Therapies with Emphasis on Sirtuin Agonists
Recent Patents on Cardiovascular Drug Discovery SHP-2 Regulates Growth Factor Dependent Vascular Signalling and Function
Mini-Reviews in Medicinal Chemistry Emerging Role for Antioxidant Therapy in Protection Against Diabetic Cardiac Complications: Experimental and Clinical Evidence for Utilization of Classic and New Antioxidants
Current Cardiology Reviews Does A Subclinical Cardiotoxic Effect of Clozapine Exist? Results from a Follow-up Pilot Study
Cardiovascular & Hematological Agents in Medicinal Chemistry Stable Gastric Pentadecapeptide BPC 157-NO-system Relation
Current Pharmaceutical Design Impact of Angiotensin Converting Enzyme Inhibitors/Angiotensin Receptors Blockers on Mortality in Acute Heart Failure Patients with Left Ventricular Systolic Dysfunction in the Middle East: Observations from the Gulf Acute Heart Failure Registry (Gulf CARE)
Current Vascular Pharmacology Criteria for Creating and Assessing Mouse Models of Diabetic Neuropathy
Current Drug Targets Pharmacological Modulations of the Renin-Angiotensin-Aldosterone System in Human Congestive Heart Failure: Effects on Peripheral Vascular Endothelial Function
Current Vascular Pharmacology Molecular Diagnosis and Treatment of Multiple Endocrine Neoplasia Type 2B in Ethnic Han Chinese
Endocrine, Metabolic & Immune Disorders - Drug Targets Cardiovascular Pharmacogenetics of Antihypertensive and Lipid- Lowering Therapies
Current Molecular Medicine Targeting Mitochondrial Dysfunction in Chronic Heart Failure: Current Evidence and Potential Approaches
Current Pharmaceutical Design Relationship of High-Sensitivity C-Reactive Protein Concentrations and Systolic Heart Failure
Current Vascular Pharmacology The Cardiovascular and Cardiac Actions of Ecstasy and its Metabolites
Current Pharmaceutical Biotechnology Fetal Risks of Maternal Diabetes
Current Women`s Health Reviews Targeting the Chemokines in Cardiac Repair
Current Pharmaceutical Design Angiogenesis as a therapeutic target in breast cancer
Mini-Reviews in Medicinal Chemistry