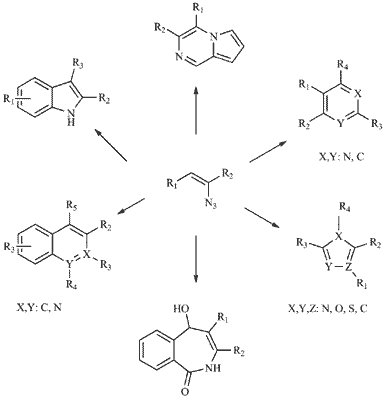
Abstract
Vinyl azides have emerged as important synthons in recent years and a variety of |useful synthetic methodologies have been achieved in the construction of the nitrogen heterocycles, such as indoles, pyrroles, imidazoles, pyrazoles, pyridines and pyrimidines. The unstable azide group and the presence of neighboring unsaturated double bond accentuate the high reactive ity of vinyl azides with other electrophiles and nucleophiles in various manners. One of the important features of vinyl azides is that they can decompose to vinyl nitrenes or highly strained three-membered cyclic imines, 2H-azirines. These two three-atom intermediates can easily undergo tandem reactions with 1,3-dipoles to generate the nitrogen-containing heterocycles. Vinyl azide can also act as a two-atom synthon while azide group serves as a leaving group in the cycloaddition reactions. What’s more, when vinyl azide is connected with a carbonyl group in the α-position of azide, the resulted α-azidovinylketone can react with the dinucleophiles, leading to the amino substituted ring. The purpose of this review is to present the synthetic approaches achieved both in our group and other groups.
Keywords: 2H-azirine, nitrene, nitrogen heterocycles, synthon, vinyl azides, α-azidovinylketone.
Current Organic Chemistry
Title:Vinyl Azides as Versatile Synthons for the Synthesis of Nitrogen-containing Heterocycles
Volume: 19 Issue: 9
Author(s): Hangcheng Ni, Guolin Zhang and Yongping Yu
Affiliation:
Keywords: 2H-azirine, nitrene, nitrogen heterocycles, synthon, vinyl azides, α-azidovinylketone.
Abstract: Vinyl azides have emerged as important synthons in recent years and a variety of |useful synthetic methodologies have been achieved in the construction of the nitrogen heterocycles, such as indoles, pyrroles, imidazoles, pyrazoles, pyridines and pyrimidines. The unstable azide group and the presence of neighboring unsaturated double bond accentuate the high reactive ity of vinyl azides with other electrophiles and nucleophiles in various manners. One of the important features of vinyl azides is that they can decompose to vinyl nitrenes or highly strained three-membered cyclic imines, 2H-azirines. These two three-atom intermediates can easily undergo tandem reactions with 1,3-dipoles to generate the nitrogen-containing heterocycles. Vinyl azide can also act as a two-atom synthon while azide group serves as a leaving group in the cycloaddition reactions. What’s more, when vinyl azide is connected with a carbonyl group in the α-position of azide, the resulted α-azidovinylketone can react with the dinucleophiles, leading to the amino substituted ring. The purpose of this review is to present the synthetic approaches achieved both in our group and other groups.
Export Options
About this article
Cite this article as:
Ni Hangcheng, Zhang Guolin and Yu Yongping, Vinyl Azides as Versatile Synthons for the Synthesis of Nitrogen-containing Heterocycles, Current Organic Chemistry 2015; 19 (9) . https://dx.doi.org/10.2174/1385272819666150320001329
| DOI https://dx.doi.org/10.2174/1385272819666150320001329 |
Print ISSN 1385-2728 |
| Publisher Name Bentham Science Publisher |
Online ISSN 1875-5348 |
Call for Papers in Thematic Issues
Advances of Heterocyclic Chemistry with Pesticide Activity
Global food safety and security will continue to be a global concern for the next 50 years and beyond. Plant diseases have had a significant impact on food safety and security throughout the entire food chain, from primary production to consumption. While conventional chemical pesticides have been traditionally used for ...read more
Calculation design of covalent/metal organic framework based catalysts
This research area combines theoretical computation and screening with machine learning for the design of covalent/metal organic framework-based catalysts, bridging the disciplines of organic chemistry, physical chemistry, computational chemistry, materials science, and machine learning. It covers several critical aspects: designing and synthesizing organic catalysts for improved performance, applying computational methods ...read more
Carbohydrates conversion in biofuels and bioproducts
Biomass pretreatment, hydrolysis, and saccharification of carbohydrates, and sugars bioconversion in biofuels and bioproducts within a biorefinery framework. Carbohydrates derived from woody biomass, agricultural wastes, algae, sewage sludge, or any other lignocellulosic feedstock are included in this issue. Simulation, techno-economic analysis, and life cycle analysis of a biorefinery process are ...read more
Catalytic C-H bond activation as a tool for functionalization of heterocycles
The major topic is the functionalization of heterocycles through catalyzed C-H bond activation. The strategies based on C-H activation not only provide straightforward formation of C-C or C-X bonds but, more importantly, allow for the avoidance of pre-functionalization of one or two of the cross-coupling partners. The beneficial impact of ...read more
Related Journals
 65
65 2
2
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
- Announcements
Related Articles
-
Noninvasive Diagnosis of Chemotherapy Related Cardiotoxicity
Current Cardiology Reviews Lentiviral Vectors: A Versatile Tool to Fight Cancer
Current Molecular Medicine Collateral Damage: Contribution of Peripheral Inflammation to Neurodegenerative Diseases
Current Topics in Medicinal Chemistry Ribonucleotide Reductase: A Mechanistic Portrait of Substrate Analogues Inhibitors
Current Medicinal Chemistry Cytochrome P450 Retinoic Acid 4-Hydroxylase Inhibitors: Potential Agents for Cancer Therapy
Mini-Reviews in Medicinal Chemistry Foamy Virus Vectors: An Awaited Alternative to Gammaretro- and Lentiviral Vectors
Current Gene Therapy Regulation of Cell Death and Survival by Resveratrol: Implications for Cancer Therapy
Anti-Cancer Agents in Medicinal Chemistry Role of Inflammatory Mediators in Angiogenesis
Current Drug Targets - Inflammation & Allergy Diagnosis and Treatment of Paraneoplastic Neurological Syndromes
Current Clinical Pharmacology A Brief Review of circRNA Biogenesis, Detection, and Function
Current Genomics Expression of Angiogenesis-related Genes in a Group of Iranian Cases of Breast Cancer
Current Pharmacogenomics and Personalized Medicine The Yin and Yang of Non-Neuronal α7-Nicotinic Receptors in Inflammation and Autoimmunity
Current Drug Targets Drug-Induced Thromboembolic Events in Patients with Malignancy
Cardiovascular & Hematological Disorders-Drug Targets Multi-target Activities of Selected Alkaloids and Terpenoids
Mini-Reviews in Organic Chemistry Inhibition of Autophagy by Targeting ATG4B: Promises and Challenges of An Emerging Anti-cancer Strategy
Clinical Cancer Drugs Hematotoxicity Testing by Cell Clonogenic Assay in Drug Development and Preclinical Trials
Current Pharmaceutical Design Sodium Selenite Decreased HDAC Activity, Cell Proliferation and Induced Apoptosis in Three Human Glioblastoma Cells
Anti-Cancer Agents in Medicinal Chemistry Leptin, Ciliary Neurotrophic Factor, Leukemia Inhibitory Factor and Interleukin- 6: Class-I Cytokines Involved in the Neuroendocrine Regulation of the Reproductive Function
Current Protein & Peptide Science Magnetic Nanoparticles: New Players in Antimicrobial Peptide Therapeutics.
Current Protein & Peptide Science Clinical Proteomics in Cancer Research
Current Proteomics