Abstract
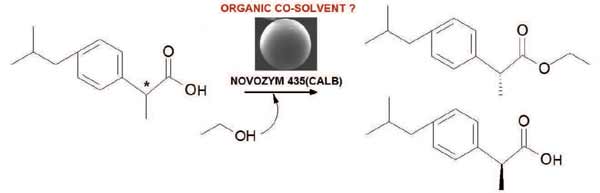
The commercial biocatalyst Novozym ® 435 was used for the kinetic resolution of (R/S)-ibuprofen through the esterification with short chain alcohols (ethanol, 1-propanol and 2-propanol) in the absence of organic co-solvent. The best enzymatic performance was obtained by employing ethanol as reagent and solvent. Due to the deleterious effect of this alcohol on the integrity of the commercial biocatalyst previously reported different organic co-solvents (isooctane, nhexane, carbon tetrachloride, ethyl acetate, acetonitrile and tetrahydrofuran) were screened in order to minimize the volume of ethanol to be used. Thus, the effect of the chemical nature of the co-solvent on enantioselective esterification of (R/S)-ibuprofen with ethanol was evaluated. The results show that the best performance was obtained with the reaction system without co-solvent added. Additionally, this investigation demonstrated the need to address multiple physicochemical properties of the solvents to analyze their effects on biocatalysis.
Keywords: Ibuprofen, Novozym435, Ethanol, Organic solvents.