Abstract
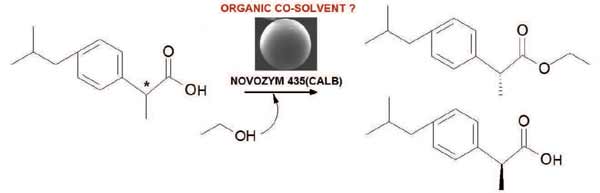
The commercial biocatalyst Novozym ® 435 was used for the kinetic resolution of (R/S)-ibuprofen through the esterification with short chain alcohols (ethanol, 1-propanol and 2-propanol) in the absence of organic co-solvent. The best enzymatic performance was obtained by employing ethanol as reagent and solvent. Due to the deleterious effect of this alcohol on the integrity of the commercial biocatalyst previously reported different organic co-solvents (isooctane, nhexane, carbon tetrachloride, ethyl acetate, acetonitrile and tetrahydrofuran) were screened in order to minimize the volume of ethanol to be used. Thus, the effect of the chemical nature of the co-solvent on enantioselective esterification of (R/S)-ibuprofen with ethanol was evaluated. The results show that the best performance was obtained with the reaction system without co-solvent added. Additionally, this investigation demonstrated the need to address multiple physicochemical properties of the solvents to analyze their effects on biocatalysis.
Keywords: Ibuprofen, Novozym435, Ethanol, Organic solvents.
Current Catalysis
Title:Effect of Co-solvents in the Enantioselective Esterification of (R/S)- ibuprofen with Ethanol
Volume: 3 Issue: 2
Author(s): Carla Jose, M. Victoria Toledo, Jaiver Osorio Grisales and Laura E. Briand
Affiliation:
Keywords: Ibuprofen, Novozym435, Ethanol, Organic solvents.
Abstract: The commercial biocatalyst Novozym ® 435 was used for the kinetic resolution of (R/S)-ibuprofen through the esterification with short chain alcohols (ethanol, 1-propanol and 2-propanol) in the absence of organic co-solvent. The best enzymatic performance was obtained by employing ethanol as reagent and solvent. Due to the deleterious effect of this alcohol on the integrity of the commercial biocatalyst previously reported different organic co-solvents (isooctane, nhexane, carbon tetrachloride, ethyl acetate, acetonitrile and tetrahydrofuran) were screened in order to minimize the volume of ethanol to be used. Thus, the effect of the chemical nature of the co-solvent on enantioselective esterification of (R/S)-ibuprofen with ethanol was evaluated. The results show that the best performance was obtained with the reaction system without co-solvent added. Additionally, this investigation demonstrated the need to address multiple physicochemical properties of the solvents to analyze their effects on biocatalysis.
Export Options
About this article
Cite this article as:
Jose Carla, Toledo Victoria M., Grisales Osorio Jaiver and Briand E. Laura, Effect of Co-solvents in the Enantioselective Esterification of (R/S)- ibuprofen with Ethanol, Current Catalysis 2014; 3 (2) . https://dx.doi.org/10.2174/2211544702666131230234058
| DOI https://dx.doi.org/10.2174/2211544702666131230234058 |
Print ISSN 2211-5447 |
| Publisher Name Bentham Science Publisher |
Online ISSN 2211-5455 |
 30
30
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
- Announcements
Related Articles
-
Ganglioside Mimicry as a Cause of Guillain-Barre Syndrome
CNS & Neurological Disorders - Drug Targets Blockade of Ser16-Hsp20 Phosphorylation Attenuates Neuroprotection Dependent Upon Bcl-2 and Bax
Current Neurovascular Research Aspartic Proteinases in Disease: A Structural Perspective
Current Drug Targets Nanoparticle Systems Modulating Myeloid-Derived Suppressor Cells for Cancer Immunotherapy
Current Topics in Medicinal Chemistry ADME Optimization and Toxicity Assessment in Early- and Late-Phase Drug Discovery
Current Topics in Medicinal Chemistry Immobilization-Induced Behavioral Deficits are Attenuated But Coping with Repeated Stress Impaired in Apomorphine Injected Rats
Current Psychopharmacology Is There a Role for Cytokine Based Therapies in Fibromyalgia
Current Pharmaceutical Design Synthesis of Salinosporamide A and Its Analogs as 20S Proteasome Inhibitors and SAR Summarization
Current Topics in Medicinal Chemistry Inflammatory Cytokine Interleukin-6 (IL-6) Promotes the Proangiogenic Ability of Adipose Stem Cells from Obese Subjects <i>via</i> the IL-6 Signaling Pathway
Current Stem Cell Research & Therapy Synthesis, Characterization & Screening for Anti-Inflammatory & Analgesic Activity of Quinoline Derivatives Bearing Azetidinones Scaffolds
Anti-Inflammatory & Anti-Allergy Agents in Medicinal Chemistry Cellular Senescence in the Development and Treatment of Cancer
Current Pharmaceutical Design Anti-Amyloid-β Immunotherapy: A Leading Novel Avenue for Alzheimer's Disease
Mini-Reviews in Medicinal Chemistry Apoptosis in the Homeostasis of the Immune System and in Human Immune Mediated Diseases
Current Pharmaceutical Design Chemokines and Brain Functions
Current Drug Targets - Inflammation & Allergy Identification of Novel Anti-inflammatory Agents from Ayurvedic Medicine for Prevention of Chronic Diseases: “Reverse Pharmacology” and “Bedside to Bench” Approach
Current Drug Targets Therapeutic Potential of Vitamin D for Multiple Sclerosis
Current Medicinal Chemistry Other Potential Therapeutic Targets in Thyroid Orbitopathy
Immunology, Endocrine & Metabolic Agents in Medicinal Chemistry (Discontinued) Ethanol Withdrawal and Hyperalgesia
Current Drug Abuse Reviews Glycogen Synthase Kinase-3 in Neurodegeneration and Neuroprotection:Lessons from Lithium
Current Alzheimer Research Homochiral Drugs: A Demanding Tendency of the Pharmaceutical Industry
Current Medicinal Chemistry