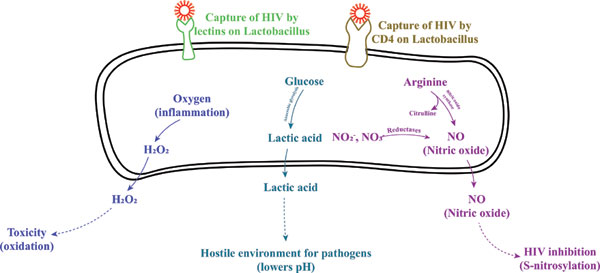
[1]
Rowland, I.; Gibson, G.; Heinken, A.; Scott, K.; Swann, J.; Thiele, I.; Tuohy, K. Gut microbiota functions: Metabolism of nutrients and other food components. Eur. J. Nutr., 2017, 1-24.
[2]
Peterson, J.; Garges, S.; Giovanni, M.; McInnes, P.; Wang, L.; Schloss, J.A.; Bonazzi, V.; McEwen, J.E.; Wetterstrand, K.A.; Deal, C.; Baker, C.C.; Di Francesco, V.; Howcroft, T.K.; Karp, R.W.; Lunsford, R.D.; Wellington, C.R.; Belachew, T.; Wright, M.; Giblin, C.; David, H.; Mills, M.; Salomon, R.; Mullins, C.; Akolkar, B.; Begg, L.; Davis, C.; Grandison, L.; Humble, M.; Khalsa, J.; Little, A.R.; Peavy, H.; Pontzer, C.; Portnoy, M.; Sayre, M.H.; Starke-Reed, P.; Zakhari, S.; Read, J.; Watson, B.; Guyer, M. The NIH Human Microbiome Project. Genome Res., 2009, 19(12), 2317-2323.
[3]
Backhed, F.; Ley, R.E.; Sonnenburg, J.L.; Peterson, D.A.; Gordon, J.I. Host-bacterial mutualism in the human intestine. Science, 2005, 307(5717), 1915-1920.
[4]
Turnbaugh, P.J.; Ley, R.E.; Hamady, M.; Fraser-Liggett, C.M.; Knight, R.; Gordon, J.I. The human microbiome project. Nature, 2007, 449(7164), 804-810.
[5]
Shepherd, E.S.; DeLoache, W.C.; Pruss, K.M.; Whitaker, W.R.; Sonnenburg, J.L. An exclusive metabolic niche enables strain engraftment in the gut microbiota. Nature, 2018, 557(7705), 434.
[6]
Sender, R.; Fuchs, S.; Milo, R. Revised estimates for the number of human and bacteria cells in the body. PLoS Biol., 2016, 14(8), e1002533.
[7]
Maeda, Y.; Takeda, K. Role of gut microbiota in rheumatoid arthritis. J. Clin. Med., 2017, 6(6), 60.
[8]
Berer, K.; Gerdes, L.A.; Cekanaviciute, E.; Jia, X.; Xiao, L.; Xia, Z.; Liu, C.; Klotz, L.; Stauffer, U.; Baranzini, S.E. Gut microbiota from multiple sclerosis patients enables spontaneous autoimmune encephalomyelitis in mice. Proc. Natl. Acad. Sci., 2017, 201711233.
[9]
Hathaway-Schrader, J.D.; Steinkamp, H.M.; Chavez, M.B.; Poulides, N.A.; Kirkpatrick, J.E.; Chew, M.E.; Huang, E.; Alekseyenko, A.V.; Aguirre, J.I.; Novince, C.M. Antibiotic perturbation of gut microbiota dysregulates osteoimmune cross talk in postpubertal skeletal development. Am. J. Pathol., 2019, 89(2), 370-390.
[10]
Chen, G.Y. The role of the gut microbiome in colorectal cancer. Clin. Colon Rectal Surg., 2018.
[11]
Zou, S.; Fang, L.; Lee, M-H. Dysbiosis of gut microbiota in promoting the development of colorectal cancer. Gastroenterol. Rep., 2017, 6(1), 1-12.
[12]
Wang, B.; Yao, M.; Lv, L.; Ling, Z.; Li, L. The human microbiota in health and disease. Engineering, 2017, 3(1), 71-82.
[13]
Anukam, K.C.; Osazuwa, E.O.; Osadolor, H.B.; Bruce, A.W.; Reid, G. Yogurt containing probiotic Lactobacillus rhamnosus GR-1 and L. reuteri RC-14 helps resolve moderate diarrhea and increases CD4 count in HIV/AIDS patients. J. Clin. Gastroenterol., 2008, 42(3), 239-243.
[14]
Reid, G. Is bacterial vaginosis a disease? Appl. Microbiol. Biotechnol., 2018, 102(2), 553-558.
[15]
Durchschein, F.; Petritsch, W.; Hammer, H.F. Diet therapy for inflammatory bowel diseases: The established and the new. World J. Gastroenterol., 2016, 22(7), 2179-2194.
[16]
Bull, M.J.; Plummer, N.T. Part 2: Treatments for chronic gastrointestinal disease and gut dysbiosis. Integr. Med. (Encinitas), 2015, 14(1), 25-33.
[17]
Gamallat, Y.; Meyiah, A.; Kuugbee, E.D.; Hago, A.M.; Chiwala, G.; Awadasseid, A.; Bamba, D.; Zhang, X.; Shang, X.; Luo, F.; Xin, Y. Lactobacillus rhamnosus induced epithelial cell apoptosis, ameliorates inflammation and prevents colon cancer development in an animal model. Biomed. Pharmacother., 2016, 83, 536-541.
[18]
Taha, T.E.; Hoover, D.R.; Dallabetta, G.A.; Kumwenda, N.I.; Mtimavalye, L.A.; Yang, L.P.; Liomba, G.N.; Broadhead, R.L.; Chiphangwi, J.D.; Miotti, P.G. Bacterial vaginosis and disturbances of vaginal flora: Association with increased acquisition of HIV. AIDS, 1998, 12(13), 1699-1706.
[19]
Martin, H.L.; Richardson, B.A.; Nyange, P.M.; Lavreys, L.; Hillier, S.L.; Chohan, B.; Mandaliya, K.; Ndinya-Achola, J.O.; Bwayo, J.; Kreiss, J. Vaginal lactobacilli, microbial flora, and risk of human immunodeficiency virus type 1 and sexually transmitted disease acquisition. J. Infect. Dis., 1999, 180(6), 1863-1868.
[20]
Taha, T.E.; Gray, R.H.; Kumwenda, N.I.; Hoover, D.R.; Mtimavalye, L.A.; Liomba, G.N.; Chiphangwi, J.D.; Dallabetta, G.A.; Miotti, P.G. HIV infection and disturbances of vaginal flora during pregnancy. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol., 1999, 20(1), 52-59.
[21]
Ledru, S.; Meda, N.; Ledru, E.; Bazie, A.J.; Chiron, J.P. HIV-1 infection associated with abnormal vaginal flora morphology and bacterial vaginosis. Lancet, 1997, 350(9086), 1251-1252.
[22]
Schwebke, J.R. Abnormal vaginal flora as a biological risk factor for acquisition of HIV infection and sexually transmitted diseases. J. Infect. Dis., 2005, 192(8), 1315-1317.
[23]
Martin, D.H. The microbiota of the vagina and its influence on women’s health and disease. Am. J. Med. Sci., 2012, 343(1), 2.
[24]
Ravel, J.; Brotman, R.M. Translating the vaginal microbiome: Gaps and challenges. Genome Med., 2016, 8(1), 35.
[25]
Zabihollahi, R.; Motevaseli, E.; Sadat, S.M.; Azizi-Saraji, A.R.; Asaadi-Dalaie, S.; Modarressi, M.H. Inhibition of HIV and HSV infection by vaginal lactobacilli in vitro and in vivo. Daru, 2012, 20(1), 53.
[26]
Irvine, S.L.; Hummelen, R.; Hekmat, S. Probiotic yogurt consumption may improve gastrointestinal symptoms, productivity, and nutritional intake of people living with human immunodeficiency virus in Mwanza, Tanzania. Nutr. Res., 2011, 31(12), 875-881.
[27]
Hummelen, R.; Changalucha, J.; Butamanya, N.L.; Koyama, T.E.; Cook, A.; Habbema, J.D.; Reid, G. Effect of 25 weeks probiotic supplementation on immune function of HIV patients. Gut Microbes, 2011, 2(2), 80-85.
[28]
Anukam, K.C.; Osazuwa, E.O.; Osadolor, H.B.; Bruce, A.W.; Reid, G. Yogurt containing probiotic Lactobacillus rhamnosus GR-1 and L. reuteri RC-14 helps resolve moderate diarrhea and increases CD4 count in HIV/AIDS patients. J. Clin. Gastroenterol., 2008, 42(3), 239-243.
[29]
Bolton, M.; van der Straten, A.; Cohen, C.R. Probiotics: Potential to prevent HIV and sexually transmitted infections in women. Sex. Transm. Dis., 2008, 35(3), 214-225.
[30]
Genç, M.R.; Delaney, M.L.; Onderdonk, A.B.; Witkin, S.S. Microbiology; Group, P.S. Vaginal nitric oxide in pregnant women with bacterial vaginosis. Am. J. Reprod. Immunol., 2006, 56(2), 86-90.
[31]
Kroncke, K.D.; Fehsel, K.; Kolb-Bachofen, V. Nitric oxide: Cytotoxicity versus cytoprotection--how, why, when, and where? Nitric Oxide, 1997, 1(2), 107-120.
[32]
Rao, D.R.; Phipatanaku, W. An overview of fractional exhaled nitric oxide and children with asthma. Expert Rev. Clin. Immunol., 2016.
[33]
Ali, G.; Mohsin, S.; Khan, M.; Nasir, G.A.; Shams, S.; Khan, S.N.; Riazuddin, S. Nitric oxide augments mesenchymal stem cell ability to repair liver fibrosis. J. Transl. Med., 2012, 10, 75.
[34]
Acharya, G.; Hopkins, R.A.; Lee, C.H. Advanced polymeric matrix for valvular complications. J. Biomed. Mater. Res. A, 2012, 100(5), 1151-1159.
[35]
Torre, D.; Pugliese, A.; Speranza, F. Role of nitric oxide in HIV-1 infection: Friend or foe? Lancet Infect. Dis., 2002, 2(5), 273-280.
[36]
Kim, T.K.; Thomas, S.M.; Ho, M.; Sharma, S.; Reich, C.I.; Frank, J.A.; Yeater, K.M.; Biggs, D.R.; Nakamura, N.; Stumpf, R.; Leigh, S.R.; Tapping, R.I.; Blanke, S.R.; Slauch, J.M.; Gaskins, H.R.; Weisbaum, J.S.; Olsen, G.J.; Hoyer, L.L.; Wilson, B.A. Heterogeneity of vaginal microbial communities within individuals. J. Clin. Microbiol., 2009, 47, 1181-1189.
[37]
Borody, T.J.; Khoruts, A. Fecal microbiota transplantation and emerging applications. Nat. Rev. Gastroenterol. Hepatol., 2012, 9(2), 88-96.
[38]
Fox, C.; Eichelberger, K. Maternal microbiome and pregnancy outcomes. Fertil. Steril., 2015, 104(6), 1358-1363.
[39]
Cobb, C.M.; Kelly, P.J.; Williams, K.B.; Babbar, S.; Angolkar, M.; Derman, R.J. The oral microbiome and adverse pregnancy outcomes. Int. J. Womens Health, 2017, 9, 551-559.
[40]
Miller, E.A.; Beasley, D.E.; Dunn, R.R.; Archie, E.A. Lactobacilli dominance and vaginal pH: Why is the human vaginal microbiome unique? Front. Microbiol., 2016, 7, 1936.
[41]
Huttenhower, C.; Gevers, D.; Knight, R.; Abubucker, S.; Badger, J.H.; Chinwalla, A.T.; Creasy, H.H.; Earl, A.M.; FitzGerald, M.G.; Fulton, R.S. Structure, function and diversity of the healthy human microbiome. Nature, 2012, 486(7402), 207.
[42]
Ma, B.; Forney, L.J.; Ravel, J. Vaginal microbiome: Rethinking health and disease. Annu. Rev. Microbiol., 2012, 66, 371-389.
[43]
Zozaya-Hinchliffe, M.; Lillis, R.; Martin, D.H.; Ferris, M.J. Quantitative PCR assessments of bacterial species in women with and without bacterial vaginosis. J. Clin. Microbiol., 2010, 48(5), 1812-1819.
[44]
Ravel, J.; Gajer, P.; Abdo, Z.; Schneider, G.M.; Koenig, S.S.; McCulle, S.L.; Karlebach, S.; Gorle, R.; Russell, J.; Tacket, C.O. Vaginal microbiome of reproductive-age women. Proc. Natl. Acad. Sci., 2011, 108(Supplement. 1), 4680-4687.
[45]
Methé, B.A.; Nelson, K.E.; Pop, M.; Creasy, H.H.; Giglio, M.G.; Huttenhower, C.; Gevers, D.; Petrosino, J.F.; Abubucker, S.; Badger, J.H. A framework for human microbiome research. Nature, 2012, 486(7402), 215.
[46]
Ravel, J.; Gajer, P.; Abdo, Z.; Schneider, G.M.; Koenig, S.S.; McCulle, S.L.; Karlebach, S.; Gorle, R.; Russell, J.; Tacket, C.O.; Brotman, R.M.; Davis, C.C.; Ault, K.; Peralta, L.; Forney, L.J. Vaginal microbiome of reproductive-age women. Proc. Natl. Acad. Sci. USA, 2011, 108(Suppl. 1), 4680-4687.
[47]
Gajer, P.; Brotman, R.M.; Bai, G.; Sakamoto, J.; Schütte, U.M.; Zhong, X.; Koenig, S.S.; Fu, L.; Ma, Z.S.; Zhou, X. Temporal dynamics
of the human vaginal microbiota. Sci. Transl. Med, 2012, 4(132), 132ra52-132ra52.
[48]
Fettweis, J.M.; Brooks, J.P.; Serrano, M.G.; Sheth, N.U.; Girerd, P.H.; Edwards, D.J.; Strauss, J.F., III; Jefferson, K.K.; Buck, G.A.; Consortium, V.M. Differences in vaginal microbiome in African American women versus women of European ancestry. Microbiology, 2014, 160(10), 2272-2282.
[49]
Steel, J.H.; Malatos, S.; Kennea, N.; Edwards, A.D.; Miles, L.; Duggan, P.; Reynolds, P.R.; Feldman, R.G.; Sullivan, M.H. Bacteria and inflammatory cells in fetal membranes do not always cause preterm labor. Pediatr. Res., 2005, 57(3), 404-411.
[50]
Stout, M.J.; Conlon, B.; Landeau, M.; Lee, I.; Bower, C.; Zhao, Q.; Roehl, K.A.; Nelson, D.M.; Macones, G.A.; Mysorekar, I.U. Identification of intracellular bacteria in the basal plate of the human placenta in term and preterm gestations. Am. J. Obstet. Gynecol., 2013, 208(3), 226.e1-226.e7.
[51]
Fortner, K.B.; Grotegut, C.A.; Ransom, C.E.; Bentley, R.C.; Feng, L.; Lan, L.; Heine, R.P.; Seed, P.C.; Murtha, A.P. Bacteria localization and chorion thinning among preterm premature rupture of membranes. PLoS One, 2014, 9(1), e83338.
[52]
Combs, C.A.; Garite, T.J.; Lapidus, J.A.; Lapointe, J.P.; Gravett, M.; Rael, J.; Amon, E.; Baxter, J.K.; Brady, K.; Clewell, W.; Eddleman, K.A.; Fortunato, S.; Franco, A.; Haas, D.M.; Heyborne, K.; Hickok, D.E.; How, H.Y.; Luthy, D.; Miller, H.; Nageotte, M.; Pereira, L.; Porreco, R.; Robilio, P.A.; Simhan, H.; Sullivan, S.A.; Trofatter, K.; Westover, T. Detection of microbial invasion of the amniotic cavity by analysis of cervicovaginal proteins in women with preterm labor and intact membranes. Am. J. Obstet. Gynecol., 2015, 212(4), 482.e1-482.e12.
[53]
Combs, C.A.; Gravett, M.; Garite, T.J.; Hickok, D.E.; Lapidus, J.; Porreco, R.; Rael, J.; Grove, T.; Morgan, T.K.; Clewell, W.; Miller, H.; Luthy, D.; Pereira, L.; Nageotte, M.; Robilio, P.A.; Fortunato, S.; Simhan, H.; Baxter, J.K.; Amon, E.; Franco, A.; Trofatter, K.; Heyborne, K. Amniotic fluid infection, inflammation, and colonization in preterm labor with intact membranes. Am. J. Obstet. Gynecol., 2014, 210(2), 125.e1-125.e15.
[54]
Aagaard, K.; Ma, J.; Antony, K.M.; Ganu, R.; Petrosino, J.; Versalovic, J. The placenta harbors a unique microbiome. Sci. Transl. Med., 2014, 6(237), 237ra65.
[55]
Pelzer, E.; Gomez-Arango, L.F.; Barrett, H.L.; Nitert, M.D. Review: Maternal health and the placental microbiome. Placenta, 2017, 54, 30-37.
[56]
Janda, J.M.; Abbott, S.L. 16S rRNA gene sequencing for bacterial identification in the diagnostic laboratory: Pluses, perils, and pitfalls. J. Clin. Microbiol., 2007, 45(9), 2761-2764.
[57]
Janda, J.M. Clinical Decisions: How Relevant is Modern Bacterial Taxonomy for Clinical Microbiologists? Clin. Microbiol. Newsl., 2018, 40(7), 51-57.
[58]
Blekhman, R.; Goodrich, J.K.; Huang, K.; Sun, Q.; Bukowski, R.; Bell, J.T.; Spector, T.D.; Keinan, A.; Ley, R.E.; Gevers, D.; Clark, A.G. Host genetic variation impacts microbiome composition across human body sites. Genome Biol., 2015, 16, 191.
[59]
Green, K.A.; Zarek, S.M.; Catherino, W.H. Gynecologic health and disease in relation to the microbiome of the female reproductive tract. Fertil. Steril., 2015, 104(6), 1351-1357.
[60]
Prince, A.L.; Chu, D.M.; Seferovic, M.D.; Antony, K.M.; Ma, J.; Aagaard, K.M. The perinatal microbiome and pregnancy: moving beyond the vaginal microbiome. Cold Spring Harb. Perspect. Med., 2015, 5(6)
[61]
Relman, D.A. ‘Til death do us part’: Coming to terms with symbiotic relationships. Forward. Nat. Rev. Microbiol., 2008, 6(10), 721-724.
[62]
Ley, R.E.; Hamady, M.; Lozupone, C.; Turnbaugh, P.; Ramey, R.R.; Bircher, J.S.; Schlegel, M.L.; Tucker, T.A.; Schrenzel, M.D.; Knight, R.; Gordon, J.I. Evolution of mammals and their gut microbes. Science, 2008, 320(5883), 1647-1651.
[63]
Sewankambo, N.; Gray, R.H.; Wawer, M.J.; Paxton, L.; McNaim, D.; Wabwire-Mangen, F.; Serwadda, D.; Li, C.; Kiwanuka, N.; Hillier, S.L.; Rabe, L.; Gaydos, C.A.; Quinn, T.C.; Konde-Lule, J. HIV-1 infection associated with abnormal vaginal flora morphology and bacterial vaginosis. Lancet, 1997, 350(9077), 546-550.
[64]
Lamont, R.F.; Sobel, J.D.; Akins, R.A.; Hassan, S.S.; Chaiworapongsa, T.; Kusanovic, J.P.; Romero, R. The vaginal microbiome: New information about genital tract flora using molecular based techniques. Bjog, 2011, 118(5), 533-549.
[65]
Marshall, J.C. An endotoxin or an exogenous
hormone? Clin. Infect. Dis, 2005, 41(Supplement_7), S470-S480.
[66]
Pålsson‐McDermott, E.M.; O’neill, L.A. Signal transduction by the lipopolysaccharide receptor, Toll‐like receptor‐4. Immunology, 2004, 113(2), 153-162.
[67]
Roumen, R.; Frieling, J.; Van Tits, H.; Van der Vliet, J.; Goris, R. Endotoxemia after major vascular operations. J. Vasc. Surg., 1993, 18(5), 853-857.
[68]
Hasday, J.D.; Bascom, R.; Costa, J.J.; Fitzgerald, T.; Dubin, W. Bacterial endotoxin is an active component of cigarette smoke. Chest J., 1999, 115(3), 829-835.
[69]
Iarŭmov, N.; Evtimov, R.; Argirov, D. The role of bacterial translocation and endotoxemia in pathogenesis of obturation ileus, caused by colorectal carcinoma. Limulus test--a method for quick diagnostics of endotoxemia. Khirurgiia, 2004, 60(3), 48-55.
[70]
Erridge, C.; Attina, T.; Spickett, C.M.; Webb, D.J. A high-fat meal induces low-grade endotoxemia: Evidence of a novel mechanism of postprandial inflammation. Am. J. Clin. Nutr., 2007, 86(5), 1286-1292.
[71]
Aoki, K. A study of endotoxemia in ulcerative colitis and Crohn’s disease. Clinical study. Acta Med. Okayama, 1978, 32(2), 147-158.
[72]
Nahum, A.; Hoyt, J.; Schmitz, L.; Moody, J.; Shapiro, R.; Marini, J.J. Effect of mechanical ventilation strategy on dissemination of intratracheally instilled Escherichia coli in dogs. Crit. Care Med., 1997, 25(10), 1733-1743.
[73]
Schietroma, M.; Carlei, F.; Cappelli, S.; Amicucci, G. Intestinal permeability and systemic endotoxemia after laparotomic or laparoscopic cholecystectomy. Ann. Surg., 2006, 243(3), 359.
[74]
Schietroma, M.; Pessia, B.; Carlei, F.; Cecilia, E.M.; Amicucci, G. Intestinal permeability, systemic endotoxemia, and bacterial translocation after open or laparoscopic resection for colon cancer: A prospective randomized study. Int. J. Colorectal Dis., 2013, 28(12), 1651-1660.
[75]
Bhandari, P.; Rishi, P.; Prabha, V. Positive effect of probiotic Lactobacillus plantarum in reversing LPS-induced infertility in a mouse model. J. Med. Microbiol., 2016, 65(5), 345-350.
[76]
Zhang, L.; Li, N.; des Robert, C.; Fang, M.; Liboni, K.; McMahon, R.; Caicedo, R.A.; Neu, J. Lactobacillus rhamnosus GG decreases lipopolysaccharide-induced systemic inflammation in a gastrostomy-fed infant rat model. J. Pediatr. Gastroenterol. Nutr., 2006, 42(5), 545-552.
[77]
Abbas, A.K.; Lichtman, A.H.; Pillai, S. Basic immunology: functions and disorders of the immune system., 2016,
[78]
Kato, I.; Yokokura, T.; Mutai, M. Macrophage activation by Lactobacillus casei in mice. Microbiol. Immunol., 1983, 27(7), 611-618.
[79]
Kirjavainen, P.V.; El-Nezami, H.S.; Salminen, S.J.; Ahokas, J.T.; Wright, P.F. The effect of orally administered viable probiotic and dairy lactobacilli on mouse lymphocyte proliferation. FEMS Immunol. Med. Microbiol., 1999, 26(2), 131-135.
[80]
Perdigon, G.; de Macias, M.E.; Alvarez, S.; Oliver, G.; de Ruiz Holgado, A.A. Effect of perorally administered lactobacilli on macrophage activation in mice. Infect. Immun., 1986, 53(2), 404-410.
[81]
Maassen, C.B.; van Holten-Neelen, C.; Balk, F.; den Bak-Glashouwer, M.J.; Leer, R.J.; Laman, J.D.; Boersma, W.J.; Claassen, E. Strain-dependent induction of cytokine profiles in the gut by orally administered Lactobacillus strains. Vaccine, 2000, 18(23), 2613-2623.
[82]
Shimizu, K.; Muranaka, Y.; Fujimura, R.; Ishida, H.; Tazume, S.; Shimamura, T. Normalization of reproductive function in germfree mice following bacterial contamination. Exp. Anim., 1998, 47(3), 151-158.
[83]
Minami, M.; Oowada, T.; Ozaki, A.; Yamamoto, T.; Mizutani, T.; Saito, K.; Adachi, J.; Fujisawa, T.; Mitsuoka, T. Estrous cycle and vaginal flora in conventionalized and gnotobiotic BALB/c mice. Bifidus-Flores Et Fructus, 1987, 1(1), 25-32.
[84]
Di Cerbo, A.; Palmieri, B.; Aponte, M.; Morales-Medina, J.C.; Iannitti, T. Mechanisms and therapeutic effectiveness of lactobacilli. J. Clin. Pathol., 2016, 69(3), 187-203.
[85]
Petrova, M.I.; Lievens, E.; Malik, S.; Imholz, N.; Lebeer, S. Lactobacillus species as biomarkers and agents that can promote various aspects of vaginal health. Front. Physiol., 2015, 6, 81.
[86]
Wilck, N.; Matus, M.G.; Kearney, S.M.; Olesen, S.W.; Forslund, K.; Bartolomaeus, H.; Haase, S.; Mähler, A.; Balogh, A.; Markó, L. Salt-responsive gut commensal modulates T H 17 axis and disease. Nature, 2017, 551(7682), 585.
[87]
Madhur, M.S.; Lob, H.E.; McCann, L.A.; Iwakura, Y.; Blinder, Y.; Guzik, T.J.; Harrison, D.G. Interleukin 17 promotes angiotensin II-induced hypertension and vascular dysfunction. Hypertension, 2010, 55(2), 500-507.
[88]
Norlander, A.E.; Saleh, M.A.; Kamat, N.V.; Ko, B.; Gnecco, J.; Zhu, L.; Dale, B.L.; Iwakura, Y.; Hoover, R.S.; McDonough, A.A. Interleukin-17A regulates renal sodium transporters and renal injury in angiotensin II-induced hypertension novelty and significance. Hypertension, 2016, 68(1), 167-174.
[89]
Lowe, P.P.; Gyongyosi, B.; Satishchandran, A.; Iracheta-Vellve, A.; Ambade, A.; Cho, Y.; Kodys, K.; Catalano, D.; Ward, D.V.; Szabo, G. Correction: Alcohol-related changes in the intestinal microbiome influence neutrophil infiltration, inflammation and steatosis in early alcoholic hepatitis in mice. PLoS One, 2017, 12(5), e0179070.
[90]
Dodd, D.; Spitzer, M.H.; Van Treuren, W.; Merrill, B.D.; Hryckowian, A.J.; Higginbottom, S.K.; Le, A.; Cowan, T.M.; Nolan, G.P.; Fischbach, M.A. A gut bacterial pathway metabolizes aromatic amino acids into nine circulating metabolites. Nature, 2017, 551(7682), 648.
[91]
Antonio, M.A.; Meyn, L.A.; Murray, P.J.; Busse, B.; Hillier, S.L. Vaginal colonization by probiotic Lactobacillus crispatus CTV-05 is decreased by sexual activity and endogenous Lactobacilli. J. Infect. Dis., 2009, 199(10), 1506-1513.
[92]
O’Hanlon, D.E.; Moench, T.R.; Cone, R.A. In vaginal fluid, bacteria associated with bacterial vaginosis can be suppressed with lactic acid but not hydrogen peroxide. BMC Infect. Dis., 2011, 11(1), 200.
[93]
Redondo-Lopez, V.; Cook, R.L.; Sobel, J.D. Emerging role of lactobacilli in the control and maintenance of the vaginal bacterial microflora. Rev. Infect. Dis., 1990, 12(5), 856-872.
[94]
Witkin, S.S.; Alvi, S.; Bongiovanni, A.M.; Linhares, I.M.; Ledger, W.J. Lactic acid stimulates interleukin-23 production by peripheral blood mononuclear cells exposed to bacterial lipopolysaccharide. FEMS Immunol. Med. Microbiol., 2011, 61(2), 153-158.
[95]
Aroutcheva, A.; Gariti, D.; Simon, M.; Shott, S.; Faro, J.; Simoes, J.A.; Gurguis, A.; Faro, S. Defense factors of vaginal lactobacilli. Am. J. Obstet. Gynecol., 2001, 185(2), 375-379.
[96]
A, V. S.; E, A. K. Vaginal protection by H2O2-producing lactobacilli. Jundishapur J. Microbiol., 2015, 8(10), e22913.
[97]
Barrons, R.; Tassone, D. Use of Lactobacillus probiotics for bacterial genitourinary infections in women: A review. Clin. Ther., 2008, 30(3), 453-468.
[98]
O’Hanlon, D.E.; Moench, T.R.; Cone, R.A. In vaginal fluid, bacteria associated with bacterial vaginosis can be suppressed with lactic acid but not hydrogen peroxide. BMC Infect. Dis., 2011, 11, 200.
[99]
Workowski, K.A.; Bolan, G.A. Sexually transmitted diseases treatment guidelines (2015). Reproduct. Endocrinol., 2015, (24), 51-56.
[100]
Nunn, K.L.; Wang, Y.Y.; Harit, D.; Humphrys, M.S.; Ma, B.; Cone, R.; Ravel, J.; Lai, S.K. Enhanced trapping of HIV-1 by human cervicovaginal mucus is associated with Lactobacillus crispatus-dominant microbiota. MBio, 2015, 6(5), e01084-e01015.
[101]
Witkin, S.S.; Linhares, I.M. HIV Inhibition by Lactobacilli: Easier in a test tube than in real life. MBio, 2015, 6(5)
[102]
Su, Y.; Zhang, B.; Su, L. CD4 detected from Lactobacillus helps understand the interaction between Lactobacillus and HIV. Microbiol. Res., 2013, 168(5), 273-277.
[103]
Irvine, S.L.; Hummelen, R.; Hekmat, S.; Looman, C.W.; Habbema, J.D.; Reid, G. Probiotic yogurt consumption is associated with an increase of CD4 count among people living with HIV/AIDS. J. Clin. Gastroenterol., 2010, 44(9), e201-e205.
[104]
Gao, W.; Weng, J.; Gao, Y.; Chen, X. Comparison of the vaginal microbiota diversity of women with and without human papillomavirus infection: a cross-sectional study. BMC Infect. Dis., 2013, 13(1), 271.
[105]
Oh, H. Y.; Kim, B.-S.; Seo, S.-S.; Kong, J.-S.; Lee, J.-K.; Park, S.-Y.; Hong, K.-M.; Kim, H.-K.; Kim, M.K. The association of uterine
cervical microbiota with an increased risk for cervical intraepithelial
neoplasia in Korea. Clin. Microbiol. Infect, 2015, 21(7), 674. e1-674. e9.
[106]
Audirac-Chalifour, A.; Torres-Poveda, K.; Bahena-Román, M.; Téllez-Sosa, J.; Martínez-Barnetche, J.; Cortina-Ceballos, B.; López-Estrada, G.; Delgado-Romero, K.; Burguete-García, A.I.; Cantú, D. Cervical microbiome and cytokine profile at various stages of cervical cancer: A pilot study. PLoS One, 2016, 11(4), e0153274.
[107]
Yang, T-K.; Chung, C-J.; Chung, S-D.; Muo, C-H.; Chang, C-H.; Huang, C-Y. Risk of endometrial cancer in women with pelvic inflammatory disease: A nationwide population-based retrospective cohort study. Medicine, 2015, 94(34)
[108]
Walther-António, M.R.; Chen, J.; Multinu, F.; Hokenstad, A.; Distad, T.J.; Cheek, E.H.; Keeney, G.L.; Creedon, D.J.; Nelson, H.; Mariani, A. Potential contribution of the uterine microbiome in the development of endometrial cancer. Genome Med., 2016, 8(1), 122.
[109]
Reid, G.; Bruce, A.W.; Fraser, N.; Heinemann, C.; Owen, J.; Henning, B. Oral probiotics can resolve urogenital infections. FEMS Immunol. Med. Microbiol., 2001, 30(1), 49-52.
[110]
Beerepoot, M.; Ter Riet, G.; Nys, S. Lactobacilli versus antibiotics to prevent urinary tract infections. A randomized double-blind non-inferiority trial in postmenopausal women. Arch. Intern. Med., 2012, 172, 704-712.
[111]
Perez-Burgos, A.; Wang, B.; Mao, Y-K.; Mistry, B.; Neufeld, K-A.M.; Bienenstock, J.; Kunze, W. Psychoactive bacteria Lactobacillus rhamnosus (JB-1) elicits rapid frequency facilitation in vagal afferents. Am. J. Physiol. Gastrointest. Liver Physiol., 2012, 304(2), G211-G220.
[112]
Al-Ghazzewi, F.; Tester, R. Impact of prebiotics and probiotics on skin health. Benef. Microbes, 2014, 5(2), 99-107.
[113]
Ettinger, G.; MacDonald, K.; Reid, G.; Burton, J.P. The influence of the human microbiome and probiotics on cardiovascular health. Gut Microbes, 2014, 5(6), 719-728.
[114]
King, S.; Glanville, J.; Sanders, M.E.; Fitzgerald, A.; Varley, D. Effectiveness of probiotics on the duration of illness in healthy children and adults who develop common acute respiratory infectious conditions: A systematic review and meta-analysis. Br. J. Nutr., 2014, 112(1), 41-54.
[115]
Steenbergen, L.; Sellaro, R.; van Hemert, S.; Bosch, J.A.; Colzato, L.S. A randomized controlled trial to test the effect of multispecies probiotics on cognitive reactivity to sad mood. Brain Behav. Immun., 2015, 48, 258-264.
[116]
Reid, G.; Abrahamsson, T.; Bailey, M.; Bindels, L.B.; Bubnov, R.; Ganguli, K.; Martoni, C.; O’Neill, C.; Savignac, H.M.; Stanton, C. How do probiotics and prebiotics function at distant sites? Benef. Microbes, 2017, 8(4), 521-533.
[117]
Stilling, R.M.; Dinan, T.G.; Cryan, J.F. Microbial genes, brain & behavior-epigenetic regulation of the gut-brain axis. Genes Brain Behav., 2014, 13(1), 69-86.
[118]
Janssen, A.W.; Kersten, S. Potential mediators linking gut bacteria to metabolic health: A critical view. J. Physiol., 2017, 595(2), 477-487.
[119]
Ogbonnaya, E.S.; Clarke, G.; Shanahan, F.; Dinan, T.G.; Cryan, J.F.; O’Leary, O.F. Adult hippocampal neurogenesis is regulated by the microbiome. Biol. Psychiatry, 2015, 78(4), e7-e9.
[120]
Han, Y.W.; Wang, X. Mobile microbiome: Oral bacteria in extra-oral infections and inflammation. J. Dent. Res., 2013, 92(6), 485-491.
[121]
Prince, A.L.; Antony, K.M.; Ma, J.; Aagaard, K.M. The microbiome and development: a mother’s perspective. Semin. Reprod. Med., 2014, 32(1), 14-22.
[122]
Straka, M. Pregnancy and periodontal tissues. Neuroendocrinol. Lett., 2011, 32(1), 34-38.
[123]
Gonzales-Marin, C.; Spratt, D.A.; Allaker, R.P. Maternal oral origin of Fusobacterium nucleatum in adverse pregnancy outcomes as determined using the 16S-23S rRNA gene intergenic transcribed spacer region. J. Med. Microbiol., 2013, 62(Pt 1), 133-144.
[124]
Ide, M.; Papapanou, P.N. Epidemiology of association between maternal periodontal disease and adverse pregnancy outcomes-systematic review. J. Periodontol., 2013, 84(4)(Suppl.), S181-S194.
[125]
Kumar, A.; Basra, M.; Begum, N.; Rani, V.; Prasad, S.; Lamba, A.K.; Verma, M.; Agarwal, S.; Sharma, S. Association of maternal periodontal health with adverse pregnancy outcome. J. Obstet. Gynaecol. Res., 2013, 39(1), 40-45.
[126]
Han, Y.W. Fusobacterium nucleatum: A commensal-turned pathogen. Curr. Opin. Microbiol., 2015, 23, 141-147.
[127]
Ha, J.E.; Jun, J.K.; Ko, H.J.; Paik, D.I.; Bae, K.H. Association between periodontitis and preeclampsia in never-smokers: A prospective study. J. Clin. Periodontol., 2014, 41(9), 869-874.
[128]
Li, Y.; Guo, H.; Wang, X.; Lu, Y.; Yang, C.; Yang, P. Coinfection with Fusobacterium nucleatum can enhance the attachment and invasion of Porphyromonas gingivalis or Aggregatibacter actinomycetemcomitans to human gingival epithelial cells. Arch. Oral Biol., 2015, 60(9), 1387-1393.
[129]
Topcuoglu, N.; Kulekci, G. 16S rRNA based microarray analysis
of ten periodontal bacteria in patients with different forms of periodontitis. Anaerobe, 2015, 35(Pt A), 35-40.
[130]
Fardini, Y.; Wang, X.; Temoin, S.; Nithianantham, S.; Lee, D.; Shoham, M.; Han, Y.W. Fusobacterium nucleatum adhesin FadA binds vascular endothelial cadherin and alters endothelial integrity. Mol. Microbiol., 2011, 82(6), 1468-1480.
[131]
Han, Y.W.; Fardini, Y.; Chen, C.; Iacampo, K.G.; Peraino, V.A.; Shamonki, J.M.; Redline, R.W. Term stillbirth caused by oral Fusobacterium nucleatum. Obstet. Gynecol., 2010, 115(2 Pt 2), 442-445.
[132]
Han, Y.W.; Shen, T.; Chung, P.; Buhimschi, I.A.; Buhimschi, C.S. Uncultivated bacteria as etiologic agents of intra-amniotic inflammation leading to preterm birth. J. Clin. Microbiol., 2009, 47(1), 38-47.
[133]
Boggess, K.A.; Moss, K.; Madianos, P.; Murtha, A.P.; Beck, J.; Offenbacher, S. Fetal immune response to oral pathogens and risk of preterm birth. Am. J. Obstet. Gynecol., 2005, 193(3 Pt 2), 1121-1126.
[134]
Gogeneni, H.; Buduneli, N.; Ceyhan-Ozturk, B.; Gumus, P.; Akcali, A.; Zeller, I.; Renaud, D.E.; Scott, D.A.; Ozcaka, O. Increased infection with key periodontal pathogens during gestational diabetes mellitus. J. Clin. Periodontol., 2015, 42(6), 506-512.
[135]
Prince, A.L.; Antony, K.M.; Chu, D.M.; Aagaard, K.M. The microbiome, parturition, and timing of birth: more questions than answers. J. Reprod. Immunol., 2014, 104-105, 12-19.
[136]
Schneiderhan, J.; Master-Hunter, T.; Locke, A. Targeting gut flora to treat and prevent disease. J. Fam. Pract., 2016, 65(1), 33-39.
[137]
Saxena, R.; Sharma, V. A metagenomic insight into the human microbiome: Its implications in health and disease; Med. Health Genomics, 2016, pp. 107-119.
[138]
Hyland, N.; Stanton, C. The gut-brain axis: dietary, probiotic, and prebiotic interventions on the microbiota; Academic Press, 2016.
[139]
Petra, A.I.; Panagiotidou, S.; Hatziagelaki, E.; Stewart, J.M.; Conti, P.; Theoharides, T.C. Gut-microbiota-brain axis and its effect on neuropsychiatric disorders with suspected immune dysregulation. Clin. Therapeut., 2015, 37(5), 984-995.
[140]
Yeoman, C.J.; Thomas, S.M.; Miller, M.E.B.; Ulanov, A.V.; Torralba, M.; Lucas, S.; Gillis, M.; Cregger, M.; Gomez, A.; Ho, M. A multi-omic systems-based approach reveals metabolic markers of bacterial vaginosis and insight into the disease. PLoS One, 2013, 8(2), e56111.
[141]
Macklaim, J.M.; Fernandes, A.D.; Di Bella, J.M.; Hammond, J-A.; Reid, G.; Gloor, G.B. Comparative meta-RNA-seq of the vaginal microbiota and differential expression by Lactobacillus iners in health and dysbiosis. Microbiome, 2013, 1(1), 12.
[142]
Liu, T.; Li, J.; Liu, Y.; Xiao, N.; Suo, H.; Xie, K.; Yang, C.; Wu, C. Short-chain fatty acids suppress lipopolysaccharide-induced production of nitric oxide and proinflammatory cytokines through inhibition of NF-κB pathway in RAW264. 7 cells. Inflammation, 2012, 35(5), 1676-1684.
[143]
Tsavkelova, E.; Klimova, S.Y.; Cherdyntseva, T.; Netrusov, A. Hormones and hormone-like substances of microorganisms: A review. Appl. Biochem. Microbiol., 2006, 42(3), 229-235.
[144]
Rezzonico, F.; Smits, T.H.; Duffy, B. Detection of AI-2 receptors in genomes of Enterobacteriaceae suggests a role of type-2 quorum sensing in closed ecosystems. Sensors, 2012, 12(5), 6645-6665.
[145]
Kunc, M.; Gabrych, A.; Witkowski, J.M. Microbiome impact on metabolism and function of sex, thyroid, growth and parathyroid hormones. Acta Biochim. Pol., 2015, 63(2)
[146]
Carnevali, O.; Avella, M.A.; Gioacchini, G. Effects of probiotic administration on zebrafish development and reproduction. Gen. Comp. Endocrinol., 2013, 188, 297-302.
[147]
Antunes, L.C.; Arena, E.T.; Menendez, A.; Han, J.; Ferreira, R.B.; Buckner, M.M.; Lolic, P.; Madilao, L.L.; Bohlmann, J.; Borchers, C.H.; Finlay, B.B. Impact of salmonella infection on host hormone metabolism revealed by metabolomics. Infect. Immun., 2011, 79(4), 1759-1769.
[148]
Huang, C.T.; Woodward, W.E.; Hornick, R.B.; Rodriguez, J.T.; Nichols, B.L. Fecal steroids in diarrhea. I. Acute shigellosis. Am. J. Clin. Nutr., 1976, 29(9), 949-955.
[149]
Hickey, R.J.; Zhou, X.; Settles, M.L.; Erb, J.; Malone, K.; Hansmann, M.A.; Shew, M.L.; Van Der Pol, B.; Fortenberry, J.D.; Forney, L.J. Vaginal microbiota of adolescent girls prior to the onset of menarche resemble those of reproductive-age women. MBio, 2015, 6(2)
[150]
Kornman, K.S.; Loesche, W.J. Effects of estradiol and progesterone on Bacteroides melaninogenicus and Bacteroides gingivalis. Infect. Immun., 1982, 35(1), 256-263.
[151]
Menon, R.; Watson, S.E.; Thomas, L.N.; Allred, C.D.; Dabney, A.; Azcarate-Peril, M.A.; Sturino, J.M. Diet complexity and estrogen receptor beta status affect the composition of the murine intestinal microbiota. Appl. Environ. Microbiol., 2013, 79(18), 5763-5773.
[152]
Guo, Y.; Qi, Y.; Yang, X.; Zhao, L.; Wen, S.; Liu, Y.; Tang, L. Association between polycystic ovary syndrome and gut microbiota. PLoS One, 2016, 11(4), e0153196.
[153]
Cho, I.; Blaser, M.J. The human microbiome: At the interface of health and disease. Nat. Rev. Genet., 2012, 13(4), 260-270.
[154]
Clavel, T.; Desmarchelier, C.; Haller, D.; Gerard, P.; Rohn, S.; Lepage, P.; Daniel, H. Intestinal microbiota in metabolic diseases: from bacterial community structure and functions to species of pathophysiological relevance. Gut Microbes, 2014, 5(4), 544-551.
[155]
Hyman, R.W.; Fukushima, M.; Jiang, H.; Fung, E.; Rand, L.; Johnson, B.; Vo, K.C.; Caughey, A.B.; Hilton, J.F.; Davis, R.W.; Giudice, L.C. Diversity of the vaginal microbiome correlates with preterm birth. Reprod. Sci., 2014, 21(1), 32-40.
[156]
Koren, O.; Goodrich, J.K.; Cullender, T.C.; Spor, A.; Laitinen, K.; Backhed, H.K.; Gonzalez, A.; Werner, J.J.; Angenent, L.T.; Knight, R.; Backhed, F.; Isolauri, E.; Salminen, S.; Ley, R.E. Host remodeling of the gut microbiome and metabolic changes during pregnancy. Cell, 2012, 150(3), 470-480.
[157]
Laker, R.C.; Wlodek, M.E.; Connelly, J.J.; Yan, Z. Epigenetic origins of metabolic disease: the impact of the maternal condition to the offspring epigenome and later health consequences. Food S. Human Wellness, 2013, 2(1), 1-11.
[158]
Musso, G.; Gambino, R.; Cassader, M. Obesity, diabetes, and gut microbiota: The hygiene hypothesis expanded? Diabetes Care, 2010, 33(10), 2277-2284.
[159]
Thum, C.; Cookson, A.L.; Otter, D.E.; McNabb, W.C.; Hodgkinson, A.J.; Dyer, J.; Roy, N.C. Can nutritional modulation of maternal intestinal microbiota influence the development of the infant gastrointestinal tract? J. Nutr., 2012, 142(11), 1921-1928.
[160]
Väisänen‐Tommiska, M.R.H. Nitric oxide in the human uterine cervix: Endogenous ripening factor. Ann. Med., 2008, 40(1), 45-55.
[161]
Zdzisinska, B.; Kandefer-Szerszen, M. [The role of nitric oxide in normal and pathologic immunologic reactions]. Postepy Hig. Med. Dosw., 1998, 52(6), 621-636.
[162]
Salvemini, D.; Marino, M.H. Inducible nitric oxide synthase and inflammation. Expert Opin. Investig. Drugs, 1998, 7(1), 65-75.
[163]
Uehara, E.U.; Shida Bde, S.; de Brito, C.A. Role of nitric oxide in immune responses against viruses: Beyond microbicidal activity. Inflamm. Res., 2015, 64(11), 845-852.
[164]
Kelm, M. Nitric oxide metabolism and breakdown. Biochim. et Biophysic. Acta (BBA)-. Bioenergetics, 1999, 1411(2-3), 273-289.
[165]
Iarullina, D.R.; Il’inskaia, O.N.; Aganov, A.V.; Silkin, N.I.; Zverev, D.G. [Alternative pathways of nitric oxide formation in lactobacilli:
EPR evidence for nitric oxide synthase activity]. Mikrobiologiia, 2006, 75(6), 731-736.
[166]
Lundberg, J.O. Nitrate transport in salivary glands with implications for NO homeostasis. Proc. Natl. Acad. Sci. USA, 2012, 109(33), 13144-13145.
[167]
Lundberg, J.O.; Carlström, M.; Larsen, F.J.; Weitzberg, E. Roles of dietary inorganic nitrate in cardiovascular health and disease. Cardiovasc. Res., 2010, 89(3), 525-532.
[168]
Qu, X.; Wu, Z.; Pang, B.; Jin, L.; Qin, L.; Wang, S. From nitrate to nitric oxide: The role of salivary glands and oral bacteria. J. Dent. Res., 2016, 95(13), 1452-1456.
[169]
Mirvish, S.; Reimers, K.; Kutler, B.; Chen, S.; Haorah, J.; Morris, C.; Grandjean, A.; Lyden, E. Nitrate and nitrite concentrations in human saliva for men and women at different ages and times of the day and their consistency over time. Eur. J. Cancer Prev., 2000, 9(5), 335-342.
[170]
Duncan, C.; Dougall, H.; Johnston, P.; Green, S.; Brogan, R.; Leifert, C.; Smith, L.; Golden, M.; Benjamin, N. Chemical generation of nitric oxide in the mouth from the enterosalivary circulation of dietary nitrate. Nat. Med., 1995, 1(6), 546-551.
[171]
Li, H.; Duncan, C.; Townend, J.; Killham, K.; Smith, L.M.; Johnston, P.; Dykhuizen, R.; Kelly, D.; Golden, M.; Benjamin, N. Nitrate-reducing bacteria on rat tongues. Appl. Environ. Microbiol., 1997, 63(3), 924-930.
[172]
Sobko, T.; Reinders, C.; Norin, E.; Midtvedt, T.; Gustafsson, L.E.; Lundberg, J.O. Gastrointestinal nitric oxide generation in germ-free and conventional rats. Am. J.-. Gastrointest. Liver Physiol., 2004, 287(5), G993-G997.
[173]
Takahashi, N. Oral microbiome metabolism: From “who are they?” to “what are they doing?”. J. Dent. Res., 2015, 94(12), 1628-1637.
[174]
Qin, L.; Liu, X.; Sun, Q.; Fan, Z.; Xia, D.; Ding, G.; Ong, H.L.; Adams, D.; Gahl, W.A.; Zheng, C. Sialin (SLC17A5) functions as a nitrate transporter in the plasma membrane. Proc. Natl. Acad. Sci. USA, 2012, 109(33), 13434-13439.
[175]
Lundberg, J.O.; Weitzberg, E.; Gladwin, M.T. The nitrate-nitrite-nitric oxide pathway in physiology and therapeutics. Nat. Rev. Drug Discov., 2008, 7(2), 156.
[176]
Tejada-Simon, M.V.; Pestka, J.J. Proinflammatory cytokine and nitric oxide induction in murine macrophages by cell wall and cytoplasmic extracts of lactic acid bacteria. J. Food Prot., 1999, 62(12), 1435-1444.
[177]
Iarullina, D.R.; Il’inskaia, O.N. [Detection of NO-synthase activity of lactobacilli by fluorescent staining]. Mikrobiologiia, 2007, 76(4), 570-572.
[178]
Tiso, M.; Schechter, A.N. Correction: Nitrate reduction to nitrite, nitric oxide and ammonia by gut bacteria under physiological conditions. PLoS One, 2015, 10, e0127490.
[179]
Adawi, D.; Kasravi, F.B.; Molin, G.; Jeppsson, B. Effect of Lactobacillus supplementation with and without arginine on liver damage and bacterial translocation in an acute liver injury model in the rat. Hepatology, 1997, 25(3), 642-647.
[180]
Adawi, D.; Molin, G.; Jeppsson, B. Inhibition of nitric oxide production and the effects of arginine and Lactobacillus administration in an acute liver injury model. Ann. Surg., 1998, 228(6), 748-755.
[181]
Billiar, T.R.; Curran, R.D.; Harbrecht, B.G.; Stuehr, D.J.; Demetris, A.J.; Simmons, R.L. Modulation of nitrogen oxide synthesis in vivo: NG-monomethyl-L-arginine inhibits endotoxin-induced nitrate/nitrate biosynthesis while promoting hepatic damage. J. Leukoc. Biol., 1990, 48(6), 565-569.
[182]
Lamine, F.; Fioramonti, J.; Bueno, L.; Nepveu, F.; Cauquil, E.; Lobysheva, I.; Eutamene, H.; Theodorou, V. Nitric oxide released by Lactobacillus farciminis improves TNBS-induced colitis in rats. Scand. J. Gastroenterol., 2004, 39(1), 37-45.
[183]
Lamine, F.; Eutamene, H.; Fioramonti, J.; Bueno, L.; Theodorou, V. Colonic responses to Lactobacillus farciminis treatment in trinitrobenzene sulphonic acid-induced colitis in rats. Scand. J. Gastroenterol., 2004, 39(12), 1250-1258.
[184]
Nathan, C.F.; Hibbs, J.B., Jr Role of nitric oxide synthesis in macrophage antimicrobial activity. Curr. Opin. Immunol., 1991, 3(1), 65-70.
[185]
Croen, K.D. Evidence for antiviral effect of nitric oxide. Inhibition of herpes simplex virus type 1 replication. J. Clin. Invest., 1993, 91(6), 2446-2452.
[186]
Sarac, F.; Salman, T.; Gun, F.; Celik, A.; Gurler, N.; Dogru Abbasoglu, S.; Olgac, V.; Saygili, A. Effect of probiotic supplementation on bacterial translocation in common bile duct obstruction. Pediatr. Surg. Int., 2015, 31(2), 155-161.
[187]
Goldenberg, J.Z.; Lytvyn, L.; Steurich, J.; Parkin, P.; Mahant, S.; Johnston, B.C. Probiotics for the prevention of pediatric antibiotic-associated diarrhea. Cochrane Database Syst. Rev., 2015, 12, Cd004827.
[188]
Laskin, J.D.; Heck, D.E.; Laskin, D.L. Multifunctional role of nitric oxide in inflammation. Trends Endocrinol. Metab., 1994, 5(9), 377-382.
[189]
Moilanen, E.; Vapaatalo, H. Nitric oxide in inflammation and immune response. Ann. Med., 1995, 27(3), 359-367.
[190]
Akaike, T.; Suga, M.; Maeda, H. Free radicals in viral pathogenesis: Molecular mechanisms involving superoxide and NO. Proc. Soc. Exp. Biol. Med., 1998, 217(1), 64-73.
[191]
Sioutas, A.; Ehrén, I.; Lundberg, J.O.; Wiklund, N.P.; Gemzell-Danielsson, K. Intrauterine nitric oxide in pelvic inflammatory disease. Fertil. Steril., 2008, 89(4), 948-952.
[192]
Mannick, J.B.; Stamler, J.S.; Teng, E.; Simpson, N.; Lawrence, J.; Jordan, J.; Finberg, R.W. Nitric oxide modulates HIV-1 replication. J. Acquir. Immune Defic. Syndr., 1999, 22(1), 1-9.
[193]
Saura, M.; Zaragoza, C.; McMillan, A.; Quick, R.A.; Hohenadl, C.; Lowenstein, J.M.; Lowenstein, C.J. An antiviral mechanism of nitric oxide: Inhibition of a viral protease. Immunity, 1999, 10(1), 21-28.
[194]
Persichini, T.; Colasanti, M.; Fraziano, M.; Colizzi, V.; Medana, C.; Polticelli, F.; Venturini, G.; Ascenzi, P. Nitric oxide inhibits the HIV-1 reverse transcriptase activity. Biochem. Biophys. Res. Commun., 1999, 258(3), 624-627.
[195]
Persichini, T.; Colasanti, M.; Lauro, G.M.; Ascenzi, P. Cysteine nitrosylation inactivates the HIV-1 protease. Biochem. Biophys. Res. Commun., 1998, 250(3), 575-576.
[196]
Joo, H-M.; Kim, K-A.; Myoung, K-S.; Ahn, Y-T.; Lee, J-H.; Huh, C-S.; Han, M.J.; Kim, D-H. Lactobacillus helveticus HY7801 ameliorates vulvovaginal candidiasis in mice by inhibiting fungal growth and NF-κB activation. Int. Immunopharmacol., 2012, 14(1), 39-46.
[197]
Clayton, T.A.; Baker, D.; Lindon, J.C.; Everett, J.R.; Nicholson, J.K. Pharmacometabonomic identification of a significant host-microbiome metabolic interaction affecting human drug metabolism. Proc. Natl. Acad. Sci. USA, 2009, 106(34), 14728-14733.
[198]
Haiser, H.J.; Turnbaugh, P.J. Developing a metagenomic view of xenobiotic metabolism. Pharmacol. Res., 2013, 69(1), 21-31.
[199]
Viaud, S.; Saccheri, F.; Mignot, G.; Yamazaki, T.; Daillere, R.; Hannani, D.; Enot, D.P.; Pfirschke, C.; Engblom, C.; Pittet, M.J.; Schlitzer, A.; Ginhoux, F.; Apetoh, L.; Chachaty, E.; Woerther, P.L.; Eberl, G.; Berard, M.; Ecobichon, C.; Clermont, D.; Bizet, C.; Gaboriau-Routhiau, V.; Cerf-Bensussan, N.; Opolon, P.; Yessaad, N.; Vivier, E.; Ryffel, B.; Elson, C.O.; Dore, J.; Kroemer, G.; Lepage, P.; Boneca, I.G.; Ghiringhelli, F.; Zitvogel, L. The intestinal microbiota modulates the anticancer immune effects of cyclophosphamide. Science, 2013, 342(6161), 971-976.
[200]
Rao, M.; Gershon, M.D. The bowel and beyond: the enteric nervous system in neurological disorders. Nat. Rev. Gastroenterol. Hepatol., 2016, 13(9), 517.
[201]
Tse, J.K. Gut Microbiota, nitric oxide, and microglia as prerequisites for neurodegenerative disorders. ACS Chem. Neurosci., 2017, 8(7), 1438-1447.
[202]
Wo, Y.; Brisbois, E.J.; Wu, J.; Li, Z.; Major, T.C.; Mohammed, A.; Wang, X.; Colletta, A.; Bull, J.L.; Matzger, A.J. Reduction of thrombosis and bacterial infection via controlled Nitric Oxide (NO) release from S-Nitroso-N-acetylpenicillamine (SNAP) impregnated carbosil intravascular catheters. ACS Biomater. Sci. Eng., 2017, 3(3), 349-359.
[203]
Yoo, J.W.; Acharya, G.; Lee, C.H. In vivo evaluation of vaginal films for mucosal delivery of nitric oxide. Biomaterials, 2009, 30(23-24), 3978-3985.
[204]
Pfeiffer, S.; Leopold, E.; Schmidt, K.; Brunner, F.; Mayer, B. Inhibition of nitric oxide synthesis by NG-nitro-L-arginine methyl ester (L-NAME): Requirement for bioactivation to the free acid, NG-nitro-L-arginine. Br. J. Pharmacol., 1996, 118(6), 1433-1440.
[205]
Cani, P.D.; Bibiloni, R.; Knauf, C.; Waget, A.; Neyrinck, A.M.; Delzenne, N.M.; Burcelin, R. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes, 2008, 57(6), 1470-1481.
[206]
Carvalho, B.M.; Guadagnini, D.; Tsukumo, D.M.L.; Schenka, A.A.; Latuf-Filho, P.; Vassallo, J.; Dias, J.C.; Kubota, L.T.; Carvalheira, J.B.C.; Saad, M.J.A. Modulation of gut microbiota by antibiotics improves insulin signalling in high-fat fed mice. Diabetologia, 2012, 55(10), 2823-2834.
[207]
Membrez, M.; Blancher, F.; Jaquet, M.; Bibiloni, R.; Cani, P.D.; Burcelin, R.G.; Corthesy, I.; Mace, K.; Chou, C.J. Gut microbiota modulation with norfloxacin and ampicillin enhances glucose tolerance in mice. Faseb J., 2008, 22(7), 2416-2426.
[208]
Remus, D.M.; van Kranenburg, R. van, S. II; Taverne, N.; Bongers, R.S.; Wels, M.; Wells, J.M.; Bron, P.A.; Kleerebezem, M. Impact of 4 Lactobacillus plantarum capsular polysaccharide clusters on surface glycan composition and host cell signaling. Microb. Cell Fact., 2012, 11, 149.
[209]
Lee, I.C.; Caggianiello, G. van, S., II; Taverne, N.; Meijerink, M.; Bron, P.A.; Spano, G.; Kleerebezem, M. Strain-specific features of extracellular polysaccharides and their impact on Lactobacillus plantarum-Host Interactions. Appl. Environ. Microbiol., 2016, 82(13), 3959-3970.
[210]
Walsh, C.J.; Guinane, C.M.; O’Toole, P.W.; Cotter, P.D. Beneficial modulation of the gut microbiota. FEBS Lett., 2014, 588(22), 4120-4130.
[211]
Morgun, A.; Dzutsev, A.; Dong, X.; Greer, R.L.; Sexton, D.J.; Ravel, J.; Schuster, M.; Hsiao, W.; Matzinger, P.; Shulzhenko, N. Uncovering effects of antibiotics on the host and microbiota using transkingdom gene networks. Gut, 2015, 64(11), 1732-1743.
[212]
Mulligan, M.E.; Citron, D.M.; McNamara, B.T.; Finegold, S.M. Impact of cefoperazone therapy on fecal flora. Antimicrob. Agents Chemother., 1982, 22(2), 226-230.
[213]
Dollive, S.; Chen, Y.Y.; Grunberg, S.; Bittinger, K.; Hoffmann, C.; Vandivier, L.; Cuff, C.; Lewis, J.D.; Wu, G.D.; Bushman, F.D. Fungi of the murine gut: Episodic variation and proliferation during antibiotic treatment. PLoS One, 2013, 8(8), e71806.
[214]
Tauber, S.C.; Nau, R. Immunomodulatory properties of antibiotics. Curr. Mol. Pharmacol., 2008, 1(1), 68-79.
[215]
Steel, H.C.; Theron, A.J.; Cockeran, R.; Anderson, R.; Feldman, C. Pathogen- and host-directed anti-inflammatory activities of macrolide antibiotics. Mediators Inflamm., 2012, 2012, 584262.
[216]
Wang, X.; Ryu, D.; Houtkooper, R.H.; Auwerx, J. Antibiotic use and abuse: A threat to mitochondria and chloroplasts with impact on research, health, and environment. BioEssays, 2015, 37(10), 1045-1053.
[217]
Yoo, J.W.; Irvine, D.J.; Discher, D.E.; Mitragotri, S. Bio-inspired, bioengineered and biomimetic drug delivery carriers. Nat. Rev. Drug Discov., 2011, 10(7), 521-535.
[218]
Vilela, D.; Parmar, J.; Zeng, Y.; Zhao, Y.; Sanchez, S. Graphene-based microbots for toxic heavy metal removal and recovery from water. Nano Lett., 2016, 16(4), 2860-2866.
[219]
Clayton, T.A.; Baker, D.; Lindon, J.C.; Everett, J.R.; Nicholson, J.K. Pharmacometabonomic identification of a significant host-microbiome metabolic interaction affecting human drug metabolism. Proc. Natl. Acad. Sci., 2009, 106(34), 14728-14733.