Abstract
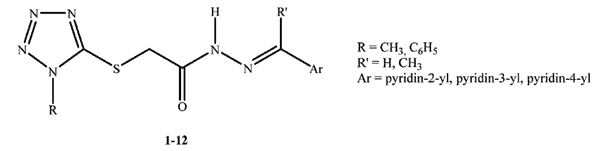
2-[(1-Methyl-1H-tetrazol-5-yl)thio]-N'-[(aryl)methylidene/ethylidene]acetohydrazides were synthesized and investigatedin vitroagainst pathogenic bacteriaand Candidaspecies for their antimicrobial activityusing CLSI broth microdilution method. MTT assay was also carried out to evaluate their cytotoxic effects on NIH/3T3 mouse embryonic fibroblast cell lines.2-((1-Phenyl-1H-tetrazol-5-yl)thio)-N'-(1-(pyridin-3-yl)ethylidene)acetohydrazide can be considered as the most promising antibacterial agent againstEnterococcusfaecalis (ATCC 29212) with a MIC value of 100 µg/mL when compared with chloramphenicol (MIC= 200 µg/mL) and low toxicity against NIH/3T3 cells(IC50>500µg/mL).
Keywords: Antimicrobial activity, cytotoxicity, hydrazone, pyridine, tetrazole.
Letters in Drug Design & Discovery
Title:Synthesis and Evaluation of Tetrazole-BasedHydrazone Derivatives Bearing a Pyridine Moiety as Antimicrobial Agents
Volume: 12 Issue: 8
Author(s): Ahmet Ozdemir, Mehlika Dilek Altıntop, Belgin Sever, Zerrin Canturk and Zafer Asım Kaplancıkl
Affiliation:
Keywords: Antimicrobial activity, cytotoxicity, hydrazone, pyridine, tetrazole.
Abstract: 2-[(1-Methyl-1H-tetrazol-5-yl)thio]-N'-[(aryl)methylidene/ethylidene]acetohydrazides were synthesized and investigatedin vitroagainst pathogenic bacteriaand Candidaspecies for their antimicrobial activityusing CLSI broth microdilution method. MTT assay was also carried out to evaluate their cytotoxic effects on NIH/3T3 mouse embryonic fibroblast cell lines.2-((1-Phenyl-1H-tetrazol-5-yl)thio)-N'-(1-(pyridin-3-yl)ethylidene)acetohydrazide can be considered as the most promising antibacterial agent againstEnterococcusfaecalis (ATCC 29212) with a MIC value of 100 µg/mL when compared with chloramphenicol (MIC= 200 µg/mL) and low toxicity against NIH/3T3 cells(IC50>500µg/mL).
Export Options
About this article
Cite this article as:
Ozdemir Ahmet, Dilek Altıntop Mehlika, Sever Belgin, Canturk Zerrin and Asım Kaplancıkl Zafer, Synthesis and Evaluation of Tetrazole-BasedHydrazone Derivatives Bearing a Pyridine Moiety as Antimicrobial Agents, Letters in Drug Design & Discovery 2015; 12 (8) . https://dx.doi.org/10.2174/1570180812666150309235217
| DOI https://dx.doi.org/10.2174/1570180812666150309235217 |
Print ISSN 1570-1808 |
| Publisher Name Bentham Science Publisher |
Online ISSN 1875-628X |
 25
25 1
1
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
Related Articles
-
Transforming Cancer Epigenetics Using Nutritive Approaches and Noncoding RNAs
Current Cancer Drug Targets Computational Studies Applied to Anti-inflammatory Drug Discovery: A Review
Current Organic Chemistry Synthesis, Antiproliferative Activity and Molecular Docking of New Thiazole/Benzothiazole Fused Pyranopyrimidine Derivatives
Letters in Organic Chemistry Exogenous Hormonal Regulation in Breast Cancer Cells by Phytoestrogens and Endocrine Disruptors
Current Medicinal Chemistry Protective Effect of NSAIDs on Cancer and Influence of COX-2 C G Genotype
Current Cancer Drug Targets Drug Interaction Potential of Trastuzumab Emtansine (T-DM1) Combined with Pertuzumab in Patients With HER2-Positive Metastatic Breast Cancer
Current Drug Metabolism Toward the Identification of Novel Carbonic Anhydrase XIV Inhibitors using 3D-QSAR Pharmacophore Model, Virtual Screening and Molecular Docking Study
Letters in Drug Design & Discovery Host Pharmacogenetics in the Treatment of HIV and Cancer
Current Drug Safety Nonprofit Pharma: Solutions to What Ails the Industry
Current Medicinal Chemistry Metabolomics
Current Drug Metabolism Nanomedical Applications of Amphiphilic Dendrimeric Micelles
Current Medicinal Chemistry <i>In Vitro</i> Anti-proliferative Properties of Flavonoids Isolated from <i>Artocarpus Heterophyllus</i> on Cancer Cell Lines
The Natural Products Journal Natural Products Targeting Cancer Stem Cells: A Revisit
Current Medicinal Chemistry Cell-Selective Mitochondrial Targeting: Progress in Mitochondrial Medicine
Current Drug Delivery Increased Expression of Matrix Metalloproteinases Mediates Thromboxane A2-Induced Invasion in Lung Cancer Cells
Current Cancer Drug Targets Some Thiazole Derivatives Combined with Different Heterocycles: Cytotoxicity Evaluation and Apoptosis Inducing Studies
Anti-Cancer Agents in Medicinal Chemistry Vitamin D Analogs- Drug Design Based on Proteins Involved in Vitamin D Signal Transduction
Current Drug Targets - Immune, Endocrine & Metabolic Disorders Anticancer Properties of Asian Water Monitor Lizard (Varanus salvator), Python (Malayopython reticulatus) and Tortoise (Cuora kamaroma amboinensis)
Anti-Cancer Agents in Medicinal Chemistry Implication of Medical Treatment for Surgical Strategies in IBD
Current Drug Targets Plant-Made Antibodies: Properties and Therapeutic Applications
Current Medicinal Chemistry