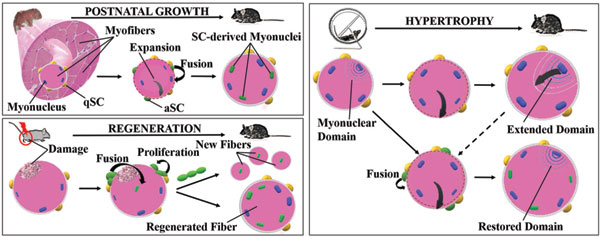
[1]
Tajbakhsh, S.; Cossu, G. Establishing myogenic identity during somitogenesis. Curr. Opin. Genet. Dev., 1997, 7(5), 634-641.
[2]
Buckingham, M.; Bajard, L.; Chang, T.; Daubas, P.; Hadchouel, J.; Meilhac, S.; Montarras, D.; Rocancourt, D.; Relaix, F. The formation of skeletal muscle: from somite to limb. J. Anat., 2003, 202(1), 59-68.
[3]
Musumeci, G.; Castrogiovanni, P.; Coleman, R.; Szychlinska, M.A.; Salvatorelli, L.; Parenti, R.; Magro, G.; Imbesi, R. Somitogenesis: From somite to skeletal muscle. Acta Histochem., 2015, 117(4-5), 313-328.
[4]
Chargè, S.B.P.; Rudnicki, M.A. Cellular and molecular regulation of muscle regeneration. Physiol. Rev., 2004, 84(1), 209-238.
[5]
Kuang, S.; Kuroda, K.; Le Grand, F.; Rudnicki, M.A. Asymmetric self-renewal and commitment of satellite stem cells in muscle. Cell, 2007, 129(5), 999-1010.
[6]
Dumont, N.A.; Bentzinger, C.F.; Sincennes, M-C.; Rudnicki, M.A. Satellite cells and skeletal muscle regeneration. Compr. Physiol., 2015, 5(3), 1027-1059.
[7]
Feige, P.; Brun, C.E.; Ritso, M.; Rudnicki, M.A. Orienting muscle stem cells for regeneration in homeostasis, aging, and disease. Cell Stem Cell, 2018, 23(5), 653-664.
[8]
Mauro, A. Satellite cell of skeletal muscle fibers. J. Biophys. Biochem. Cytol., 1961, 9(2), 493-495.
[9]
Katz, B. The terminations of the afferent nerve fibre in the muscle spindle of the frog. Philos. Trans. R. Soc. London . B Biol. Sci., 1961, 243(703), 221-240.
[10]
Seale, P.; Sabourin, L.A.; Girgis-Gabardo, A.; Mansouri, A.; Gruss, P.; Rudnicki, M.A. Pax7 is required for the specification of myogenic satellite cells. Cell, 2000, 102(6), 777-786.
[11]
Relaix, F.; Montarras, D.; Zaffran, S.; Gayraud-Morel, B.; Rocancourt, D.; Tajbakhsh, S.; Mansouri, A.; Cumano, A.; Buckingham, M. Pax3 and Pax7 have distinct and overlapping functions in adult muscle progenitor cells. J. Cell Biol., 2006, 172(1), 91-102.
[12]
Mechtersheimer, G.; Staudter, M.; Möller, P. Expression of the natural killer cell-associated antigens CD56 and CD57 in human neural and striated muscle cells and in their tumors. Cancer Res., 1991, 51(4), 1300-1307.
[13]
Irintchev, A.; Zeschnigk, M.; Starzinski-Powitz, A.; Wernig, A. Expression pattern of M-cadherin in normal, denervated, and regenerating mouse muscles. Dev. Dyn., 1994, 199(4), 326-337.
[14]
Garry, D.J.; Yang, Q.; Bassel-Duby, R.; Williams, R.S. Persistent expression of MNF identifies myogenic stem cells in postnatal muscles. Dev. Biol., 1997, 188(2), 280-294.
[15]
Tatsumi, R.; Anderson, J.E.; Nevoret, C.J.; Halevy, O.; Allen, R.E. HGF/SF is present in normal adult skeletal muscle and is capable of activating satellite cells. Dev. Biol., 1998, 194(1), 114-128.
[16]
Jesse, T.L.; LaChance, R.; Iademarco, M.F.; Dean, D.C. Interferon regulatory factor-2 is a transcriptional activator in muscle where It regulates expression of vascular cell adhesion molecule-1. J. Cell Biol., 1998, 140(5), 1265-1276.
[17]
Beauchamp, J.R.; Heslop, L.; Yu, D.S.; Tajbakhsh, S.; Kelly, R.G.; Wernig, A.; Buckingham, M.E.; Partridge, T.A.; Zammit, P.S. Expression of CD34 and Myf5 defines the majority of quiescent adult skeletal muscle satellite cells. J. Cell Biol., 2000, 151(6), 1221-1234.
[18]
Cornelison, D.D.W.; Filla, M.S.; Stanley, H.M.; Rapraeger, A.C.; Olwin, B.B. Syndecan-3 and syndecan-4 specifically mark skeletal muscle satellite cells and are implicated in satellite cell maintenance and muscle regeneration. Dev. Biol., 2001, 239(1), 79-94.
[19]
Schmidt, K.; Glaser, G.; Wernig, A.; Wegner, M.; Rosorius, O. Sox8 is a specific marker for muscle satellite cells and inhibits myogenesis. J. Biol. Chem., 2003, 278(32), 29769-29775.
[20]
Lee, H-J.; Göring, W.; Ochs, M.; Mühlfeld, C.; Steding, G.; Paprotta, I.; Engel, W.; Adham, I.M. Sox15 is required for skeletal muscle regeneration. Mol. Cell. Biol., 2004, 24(19), 8428-8436.
[21]
Sherwood, R.I.; Christensen, J.L.; Conboy, I.M.; Conboy, M.J.; Rando, T.A.; Weissman, I.L.; Wagers, A.J. Isolation of adult mouse myogenic progenitors. Cell, 2004, 119(4), 543-554.
[22]
Volonte, D.; Liu, Y.; Galbiati, F. The modulation of caveolin-1 expression controls satellite cell activation during muscle repair. FASEB J., 2005, 19(2), 237-239.
[23]
Fukada, S.; Uezumi, A.; Ikemoto, M.; Masuda, S.; Segawa, M.; Tanimura, N.; Yamamoto, H.; Miyagoe-Suzuki, Y.; Takeda, S. Molecular signature of quiescent satellite cells in adult skeletal muscle. Stem Cells, 2007, 25(10), 2448-2459.
[24]
Gnocchi, V.F.; White, R.B.; Ono, Y.; Ellis, J.A.; Zammit, P.S. Further characterisation of the molecular signature of quiescent and activated mouse muscle satellite cells. PLoS One, 2009, 4(4), e5205.
[25]
Fukada, S-i.; Yamaguchi, M.; Kokubo, H.; Ogawa, R.; Uezumi, A.; Yoneda, T.; Matev, M.M.; Motohashi, N.; Ito, T.; Zolkiewska, A.; Johnson, R.L.; Saga, Y.; Miyagoe-Suzuki, Y.; Tsujikawa, K.; Takeda, S.; Yamamoto, H. Hesr1 and Hesr3 are essential to generate undifferentiated quiescent satellite cells and to maintain satellite cell numbers. Development, 2011, 138(21), 4609-4619.
[26]
Dumont, N.A.; Wang, Y.X.; von Maltzahn, J.; Pasut, A.; Bentzinger, C.F.; Brun, C.E.; Rudnicki, M.A. Dystrophin expression in muscle stem cells regulates their polarity and asymmetric division. Nat. Med., 2015, 21(12), 1455-1463.
[27]
Bischoff, R.; Heintz, C. Enhancement of skeletal muscle regeneration. Dev. Dyn., 1994, 201(1), 41-54.
[28]
Blaauw, B.; Reggiani, C. The role of satellite cells in muscle hypertrophy. J. Muscle Res. Cell Motil., 2014, 35(1), 3-10.
[29]
Murach, K.A.; Englund, D.A.; Dupont-Versteegden, E.E.; McCarthy, J.J.; Peterson, C.A. Myonuclear domain flexibility challenges rigid assumptions on satellite cell contribution to skeletal muscle fiber hypertrophy. Front. Physiol., 2018, 9, 635.
[30]
Fry, C.S.; Lee, J.D.; Jackson, J.R.; Kirby, T.J.; Stasko, S.A.; Liu, H.; Dupont-Versteegden, E.E.; McCarthy, J.J.; Peterson, C.A. Regulation of the muscle fiber microenvironment by activated satellite cells during hypertrophy. FASEB J., 2014, 28(4), 1654-1665.
[31]
McCarthy, J.J.; Mula, J.; Miyazaki, M.; Erfani, R.; Garrison, K.; Farooqui, A.B.; Srikuea, R.; Lawson, B.A.; Grimes, B.; Keller, C.; Van Zant, G.; Campbell, K.S.; Esser, K.A.; Dupont-Versteegden, E.E.; Peterson, C.A. Effective fiber hypertrophy in satellite cell-depleted skeletal muscle. Development, 2011, 138(17), 3657-3666.
[32]
Murach, K.A.; White, S.H.; Wen, Y.; Ho, A.; Dupont-Versteegden, E.E.; McCarthy, J.J.; Peterson, C.A. Differential requirement for satellite cells during overload-induced muscle hypertrophy in growing versus mature mice. Skelet. Muscle, 2017, 7(1), 14.
[33]
Lee, J.D.; Fry, C.S.; Mula, J.; Kirby, T.J.; Jackson, J.R.; Liu, F.; Yang, L.; Dupont-Versteegden, E.E.; McCarthy, J.J.; Peterson, C.A. Aged muscle demonstrates fiber-type adaptations in response to mechanical overload, in the absence of myofiber hypertrophy, independent of satellite cell abundance. J. Gerontol.Ser A Biol. Sci. Med. Sci., 2016, 71(4), 461-467.
[34]
Shefer, G.; Van de Mark, D.P.; Richardson, J.B.; Yablonka-Reuveni, Z. Satellite-cell pool size does matter: Defining the myogenic potency of aging skeletal muscle. Dev. Biol., 2006, 294(1), 50-66.
[35]
Scicchitano, B.M.; Sica, G.; Musarò, A. Stem Cells and Tissue Niche: Two Faces of the Same Coin of Muscle Regeneration. Eur. J. Transl. Myol., 2016, 26(4), 6125.
[36]
Barberi, L.; Scicchitano, B.M.; De Rossi, M.; Bigot, A.; Duguez, S.; Wielgosik, A.; Stewart, C.; McPhee, J.; Conte, M.; Narici, M.; Franceschi, C.; Mouly, V.; Butler-Browne, G.; Musarò, A. Age-dependent alteration in muscle regeneration: the critical role of tissue niche. Biogerontology, 2013, 14(3), 273-292.
[37]
Bischoff, R. Interaction between satellite cells and skeletal muscle fibers. Development, 1990, 109(4), 943-952.
[38]
Xu, X.; Wilschut, K.J.; Kouklis, G.; Tian, H.; Hesse, R.; Garland, C.; Sbitany, H.; Hansen, S.; Seth, R.; Knott, P.D.; Hoffman, W.Y.; Pomerantz, J.H. Human satellite cell transplantation and regeneration from diverse skeletal muscles. Stem Cell Rep, 2015, 5(3), 419-434.
[39]
Murphy, M.M.; Lawson, J.A.; Mathew, S.J.; Hutcheson, D.A.; Kardon, G. Satellite cells, connective tissue fibroblasts and their interactions are crucial for muscle regeneration. Development, 2011, 138(17), 3625-3637.
[40]
Boonsanay, V.; Zhang, T.; Georgieva, A.; Kostin, S.; Qi, H.; Yuan, X.; Zhou, Y.; Braun, T. Regulation of skeletal muscle stem cell quiescence by Suv4-20h1-dependent facultative heterochromatin formation. Cell Stem Cell, 2016, 18(2), 229-242.
[41]
Yamaguchi, M.; Watanabe, Y.; Ohtani, T.; Uezumi, A.; Mikami, N.; Nakamura, M.; Sato, T.; Ikawa, M.; Hoshino, M.; Tsuchida, K.; Miyagoe-Suzuki, Y.; Tsujikawa, K.; Takeda, S.; Yamamoto, H.; Fukada, S. Calcitonin receptor signaling inhibits muscle stem cells from escaping the quiescent state and the niche. Cell Rep, 2015, 13(2), 302-314.
[42]
Quarta, M.; Brett, J.O.; DiMarco, R.; De Morree, A.; Boutet, S.C.; Chacon, R.; Gibbons, M.C.; Garcia, V.A.; Su, J.; Shrager, J.B.; Heilshorn, S.; Rando, T.A. An artificial niche preserves the quiescence of muscle stem cells and enhances their therapeutic efficacy. Nat. Biotechnol., 2016, 34(7), 752-759.
[43]
Sato, T.; Yamamoto, T.; Sehara-Fujisawa, A. miR-195/497 induce postnatal quiescence of skeletal muscle stem cells. Nat. Commun., 2014, 5(1), 4597.
[44]
Cheung, T.H.; Quach, N.L.; Charville, G.W.; Liu, L.; Park, L.; Edalati, A.; Yoo, B.; Hoang, P.; Rando, T.A. Maintenance of muscle stem-cell quiescence by microRNA-489. Nature, 2012, 482(7386), 524-528.
[45]
Westholm, J.O.; Lai, E.C. Mirtrons: microRNA biogenesis via splicing. Biochimie, 2011, 93(11), 1897-1904.
[46]
Baghdadi, M.B.; Firmino, J.; Soni, K.; Evano, B.; Di Girolamo, D.; Mourikis, P.; Castel, D.; Tajbakhsh, S. Notch-Induced miR-708 Antagonizes satellite cell migration and maintains quiescence. Cell Stem Cell, 2018, 23, 1-10.
[47]
Scharner, J.; Zammit, P.S. The muscle satellite cell at 50: the formative years. Skelet. Muscle, 2011, 1(1), 28.
[48]
Creuzet, S.; Lescaudron, L.; Li, Z.; Fontaine-Pérus, J.; Myo, D. Myogenin, and Desmin-nls-lacZ Transgene emphasize the distinct patterns of satellite cell activation in growth and regeneration. Exp. Cell Res., 1998, 243(2), 241-253.
[49]
Yablonka-Reuveni, Z.; Rivera, A.J. Temporal expression of regulatory and structural muscle proteins during myogenesis of satellite cells on isolated adult rat fibers. Dev. Biol., 1994, 164(2), 588-603.
[50]
van Velthoven, C.T.J.; de Morree, A.; Egner, I.M.; Brett, J.O.; Rando, T.A. Transcriptional profiling of quiescent muscle stem cells in vivo. Cell Rep, 2017, 21(7), 1994-2004.
[51]
Fu, X.; Wang, H.; Hu, P. Stem cell activation in skeletal muscle regeneration. Cell. Mol. Life Sci., 2015, 72(9), 1663-1677.
[52]
Zhang, K.; Sha, J.; Harter, M.L. Activation of Cdc6 by MyoD is associated with the expansion of quiescent myogenic satellite cells. J. Cell Biol., 2010, 188(1), 39-48.
[53]
Relaix, F.; Zammit, P.S. Satellite cells are essential for skeletal muscle regeneration: the cell on the edge returns centre stage. Development, 2012, 139(16), 2845-2856.
[54]
Chang, N.C.; Sincennes, M-C.; Chevalier, F.P.; Brun, C.E.; Lacaria, M.; Segalés, J.; Muñoz-Cánoves, P.; Ming, H.; Rudnicki, M.A. The dystrophin glycoprotein complex regulates the epigenetic activation of muscle stem cell commitment. Cell Stem Cell, 2018, 22(5), 755-768.
[55]
Palacios, D.; Mozzetta, C.; Consalvi, S.; Caretti, G.; Saccone, V.; Proserpio, V.; Marquez, V.E.; Valente, S.; Mai, A.; Forcales, S.V.; Sartorelli, V.; Puri, P.L. TNF/p38α/polycomb signaling to Pax7 locus in satellite cells links inflammation to the epigenetic control of muscle regeneration. Cell Stem Cell, 2010, 7(4), 455-469.
[56]
Mozzetta, C.; Consalvi, S.; Saccone, V.; Forcales, S.V.; Puri, P.L.; Palacios, D. Selective control of Pax7 expression by TNF-activated p38α/polycomb repressive complex 2 (PRC2) signaling during muscle satellite cell differentiation. Cell Cycle, 2011, 10(2), 191-198.
[57]
Ding, S.; Swennen, G.N.M.; Messmer, T.; Gagliardi, M.; Molin, D.G.M.; Li, C.; Zhou, G.; Post, M.J. Maintaining bovine satellite cells stemness through p38 pathway. Sci. Rep., 2018, 8(1), 10808.
[58]
Chen, J-F.; Mandel, E.M.; Thomson, J.M.; Wu, Q.; Callis, T.E.; Hammond, S.M.; Conlon, F.L.; Wang, D-Z. The role of microRNA-1 and microRNA-133 in skeletal muscle proliferation and differentiation. Nat. Genet., 2006, 38(2), 228-233.
[59]
Kim, H.K.; Lee, Y.S.; Sivaprasad, U.; Malhotra, A.; Dutta, A. Muscle-specific microRNA miR-206 promotes muscle differentiation. J. Cell Biol., 2006, 174(5), 677-687.
[60]
Baghdadi, M.B.; Castel, D.; Machado, L.; Fukada, S.; Birk, D.E.; Relaix, F.; Tajbakhsh, S.; Mourikis, P. Reciprocal signalling by Notch-Collagen V-CALCR retains muscle stem cells in their niche. Nature, 2018, 557(7707), 714-718.
[61]
Fujimaki, S.; Seko, D.; Kitajima, Y.; Yoshioka, K.; Tsuchiya, Y.; Masuda, S.; Ono, Y. Notch1 and Notch2 coordinately regulate stem cell function in the quiescent and activated states of muscle satellite cells. Stem Cells, 2018, 36(2), 278-285.
[62]
Chen, Z.; Bu, N.; Qiao, X.; Zuo, Z.; Shu, Y.; Liu, Z.; Qian, Z.; Chen, J.; Hou, Y. Forkhead box M1 transcriptionally regulates the expression of long noncoding RNAs Snhg8 and Gm26917 to promote proliferation and survival of muscle satellite cells. Stem Cells, 2018, 36(7), 1097-1108.
[63]
Yan, Z.; Choi, S.; Liu, X.; Zhang, M.; Schageman, J.J.; Lee, S.Y.; Hart, R.; Lin, L.; Thurmond, F.A.; Williams, R.S. Highly coordinated gene regulation in mouse skeletal muscle regeneration. J. Biol. Chem., 2003, 278(10), 8826-8836.
[64]
Musarò, A. The basis of muscle regeneration. Adv. Biol., 2014, 1-16.
[65]
Halevy, O.; Novitch, B.G.; Spicer, D.B.; Skapek, S.X.; Rhee, J.; Hannon, G.J.; Beach, D.; Lassar, A.B. Correlation of terminal cell cycle arrest of skeletal muscle with induction of p21 by MyoD. Science, 1995, 267(5200), 1018-1021.
[66]
Dey, B.K.; Gagan, J.; Dutta, A. miR-206 and -486 induce myoblast differentiation by downregulating Pax7. Mol. Cell. Biol., 2011, 31(1), 203-214.
[67]
Yu, X.; Zhang, Y.; Li, T.; Ma, Z.; Jia, H.; Chen, Q.; Zhao, Y.; Zhai, L.; Zhong, R.; Li, C.; Zou, X.; Meng, J.; Chen, A.K.; Puri, P.L.; Chen, M.; Zhu, D. Long non-coding RNA Linc-RAM enhances myogenic differentiation by interacting with MyoD. Nat. Commun., 2017, 8, 14016.
[68]
Hernández-Hernández, J.M.; García-González, E.G.; Brun, C.E.; Rudnicki, M.A. The myogenic regulatory factors, determinants of muscle development, cell identity and regeneration. Semin. Cell Dev. Biol., 2017, 72, 10-18.
[69]
Teixeira, C.F.P.; Chaves, F.; Zamunér, S.R.; Fernandes, C.M.; Zuliani, J.P.; Cruz-Hofling, M.A.; Fernandes, I.; Gutiérrez, J.M. Effects of neutrophil depletion in the local pathological alterations and muscle regeneration in mice injected with Bothrops jararaca snake venom. Int. J. Exp. Pathol., 2005, 86(2), 107-115.
[70]
Huang, W-C.; Sala-Newby, G.B.; Susana, A.; Johnson, J.L.; Newby, A.C. Classical macrophage activation up-regulates several matrix metalloproteinases through mitogen activated protein kinases and nuclear factor-κB. PLoS One, 2012, 7(8), e42507.
[71]
Chen, S-E.; Jin, B.; Li, Y-P. TNF-alpha regulates myogenesis and muscle regeneration by activating p38 MAPK. Am. J. Physiol. Cell Physiol., 2007, 292(5), C1660-C1671.
[72]
Peterson, J.M.; Bakkar, N.; Guttridge, D.C. NF-κB signaling in skeletal muscle health and disease. Curr. Top. Dev. Biol., 2011, 96, 85-119.
[73]
Muñoz-Cánoves, P.; Scheele, C.; Pedersen, B.K.; Serrano, A.L. Interleukin-6 myokine signaling in skeletal muscle: a double-edged sword? FEBS J., 2013, 280(17), 4131-4148.
[74]
Deng, B.; Wehling-Henricks, M.; Villalta, S.A.; Wang, Y.; Tidball, J.G. IL-10 triggers changes in macrophage phenotype that promote muscle growth and regeneration. J. Immunol., 2012, 189(7), 3669-3680.
[75]
Arnold, L.; Henry, A.; Poron, F.; Baba-Amer, Y.; van Rooijen, N.; Plonquet, A.; Gherardi, R.K.; Chazaud, B. Inflammatory monocytes recruited after skeletal muscle injury switch into antiinflammatory macrophages to support myogenesis. J. Exp. Med., 2007, 204(5), 1057-1069.
[76]
Ruffell, D.; Mourkioti, F.; Gambardella, A.; Kirstetter, P.; Lopez, R.G.; Rosenthal, N.; Nerlov, C.A. CREB-C/EBPbeta cascade induces M2 macrophage-specific gene expression and promotes muscle injury repair. Proc. Natl. Acad. Sci. USA, 2009, 106(41), 17475-17480.
[77]
Liu, X.; Liu, Y.; Zhao, L.; Zeng, Z.; Xiao, W.; Chen, P. Macrophage depletion impairs skeletal muscle regeneration: The roles of regulatory factors for muscle regeneration. Cell Biol. Int., 2017, 41(3), 228-238.
[78]
Engert, J.C.; Berglund, E.B.; Rosenthal, N. Proliferation precedes differentiation in IGF-I-stimulated myogenesis. J. Cell Biol., 1996, 135(2), 431-440.
[79]
Musarò, A.; Rosenthal, N. Maturation of the myogenic program is induced by postmitotic expression of insulin-like growth factor I. Mol. Cell. Biol., 1999, 19(4), 3115-3124.
[80]
Tonkin, J.; Temmerman, L.; Sampson, R.D.; Gallego-Colon, E.; Barberi, L.; Bilbao, D.; Schneider, M.D.; Musarò, A.; Rosenthal, N. Monocyte/Macrophage-derived IGF-1 Orchestrates murine skeletal muscle regeneration and modulates autocrine polarization. Mol. Ther., 2015, 23(7), 1189-1200.
[81]
Zhang, C.; Li, Y.; Wu, Y.; Wang, L.; Wang, X.; Du, J. Interleukin-6/STAT3 pathway is essential for macrophage infiltration and myoblast proliferation during muscle regeneration. J. Biol. Chem., 2013, 288(3), 1489-1499.
[82]
Forcina, L.; Miano, C.; Musarò, A. The physiopathologic interplay between stem cells and tissue niche in muscle regeneration and the role of IL-6 on muscle homeostasis and diseases. Cytokine Growth Factor Rev., 2018, 41, 1-9.
[83]
Serrano, A.L.; Baeza-Raja, B.; Perdiguero, E.; Jardí, M.; Muñoz-Cánoves, P. Interleukin-6 Is an essential regulator of satellite cell-mediated skeletal muscle hypertrophy. Cell Metab., 2008, 7(1), 33-44.
[84]
Joe, A.W.B.; Yi, L.; Natarajan, A.; Le Grand, F.; So, L.; Wang, J.; Rudnicki, M.A.; Rossi, F.M.V. Muscle injury activates resident fibro/adipogenic progenitors that facilitate myogenesis. Nat. Cell Biol., 2010, 12(2), 153-163.
[85]
Formicola, L.; Marazzi, G.; Sassoon, D.A. The extraocular muscle stem cell niche is resistant to ageing and disease. Front. Aging Neurosci., 2014, 6, 328.
[86]
Uezumi, A.; Fukada, S.; Yamamoto, N.; Takeda, S.; Tsuchida, K. Mesenchymal progenitors distinct from satellite cells contribute to ectopic fat cell formation in skeletal muscle. Nat. Cell Biol., 2010, 12(2), 143-152.
[87]
Lemos, D.R.; Babaeijandaghi, F.; Low, M.; Chang, C-K.; Lee, S.T.; Fiore, D.; Zhang, R-H.; Natarajan, A.; Nedospasov, S.A.; Rossi, F.M.V. Nilotinib reduces muscle fibrosis in chronic muscle injury by promoting TNF-mediated apoptosis of fibro/adipogenic progenitors. Nat. Med., 2015, 21(7), 786-794.
[88]
Shin, J.; McFarland, D.C.; Velleman, S.G. Heparan sulfate proteoglycans, syndecan-4 and glypican-1, differentially regulate myogenic regulatory transcription factors and paired box 7 expression during turkey satellite cell myogenesis: Implications for muscle growth. Poult. Sci., 2012, 91(1), 201-207.
[89]
Harthan, L.B.; McFarland, D.C.; Velleman, S.G. The effect of syndecan-4 and glypican-1 expression on age-related changes in myogenic satellite cell proliferation, differentiation, and fibroblast growth factor 2 responsiveness. Comp. Biochem. Physiol.Part A Mol. Integr. Physiol., 2013, 166(4), 590-602.
[90]
Yin, H.; Price, F.; Rudnicki, M.A. Satellite cells and the muscle stem cell niche. Physiol. Rev., 2013, 93(1), 23-67.
[91]
Fiore, D.; Judson, R.N.; Low, M.; Lee, S.; Zhang, E.; Hopkins, C.; Xu, P.; Lenzi, A.; Rossi, F.M.V.; Lemos, D.R. Pharmacological blockage of fibro/adipogenic progenitor expansion and suppression of regenerative fibrogenesis is associated with impaired skeletal muscle regeneration. Stem Cell Res. , 2016, 17(1), 161-169.
[92]
Urciuolo, A.; Quarta, M.; Morbidoni, V.; Gattazzo, F.; Molon, S.; Grumati, P.; Montemurro, F.; Tedesco, F.S.; Blaauw, B.; Cossu, G.; Vozzi, G.; Rando, T.A.; Bonaldo, P. Collagen VI regulates satellite cell self-renewal and muscle regeneration. Nat. Commun., 2013, 4(1), 1964.
[93]
Hardy, D.; Besnard, A.; Latil, M.; Jouvion, G.; Briand, D.; Thépenier, C.; Pascal, Q.; Guguin, A.; Gayraud-Morel, B.; Cavaillon, J-M.; Tajbakhsh, S.; Rocheteau, P.; Chrétien, F. Comparative study of injury models for studying muscle regeneration in mice. PLoS One, 2016, 11(1), e0147198.
[94]
Latroche, C.; Weiss-Gayet, M.; Muller, L.; Gitiaux, C.; Leblanc, P.; Liot, S.; Ben-Larbi, S.; Abou-Khalil, R.; Verger, N.; Bardot, P.; Magnan, M.; Chrétien, F.; Mounier, R.; Germain, S.; Chazaud, B. Coupling between myogenesis and angiogenesis during skeletal muscle regeneration is stimulated by restorative macrophages. Stem Cell Rep, 2017, 9(6), 2018-2033.
[95]
Qahar, M.; Takuma, Y.; Mizunoya, W.; Tatsumi, R.; Ikeuchi, Y.; Nakamura, M. Semaphorin 3A promotes activation of Pax7, Myf5, and MyoD through inhibition of emerin expression in activated satellite cells. FEBS Open Bio, 2016, 6(6), 529-539.
[96]
Tatsumi, R.; Sankoda, Y.; Anderson, J.E.; Sato, Y.; Mizunoya, W.; Shimizu, N.; Suzuki, T.; Yamada, M.; Rhoads, R.P.; Ikeuchi, Y.; Allen, R.E.; Furuse, M.; Ikcuchi, Y.; Nishimura, T.; Yagi, T. Possible implication of satellite cells in regenerative motoneuritogenesis: HGF upregulates neural chemorepellent Sema3A during myogenic differentiation. Am. J. Physiol., 2009, 297(2), C238-C252.
[97]
Tatsumi, R.; Suzuki, T.; Do, M.Q.; Ohya, Y.; Anderson, J.E.; Shibata, A.; Kawaguchi, M.; Ohya, S.; Ohtsubo, H.; Mizunoya, W.; Sawano, S.; Komiya, Y.; Ichitsubo, R.; Ojima, K.; Nishimatsu, S.I.; Nohno, T.; Ohsawa, Y.; Sunada, Y.; Nakamura, M.; Furuse, M.; Ikeuchi, Y.; Nishimura, T.; Yagi, T.; Allen, R.E. Slow-myofiber commitment by semaphorin 3A secreted from myogenic stem cells. Stem Cells, 2017, 35(7), 1815-1834.
[98]
De Angelis, L.; Berghella, L.; Coletta, M.; Lattanzi, L.; Zanchi, M.; Cusella-De Angelis, M.G.; Ponzetto, C.; Cossu, G. Skeletal myogenic progenitors originating from embryonic dorsal aorta coexpress endothelial and myogenic markers and contribute to postnatal muscle growth and regeneration. J. Cell Biol., 1999, 147(4), 869-878.
[99]
Tamaki, T.; Akatsuka, A.; Ando, K.; Nakamura, Y.; Matsuzawa, H.; Hotta, T.; Roy, R.R.; Edgerton, V.R. Identification of myogenic-endothelial progenitor cells in the interstitial spaces of skeletal muscle. J. Cell Biol., 2002, 157(4), 571-577.
[100]
Polesskaya, A.; Seale, P.; Rudnicki, M.A. Wnt signaling induces the myogenic specification of resident CD45+ adult stem cells during muscle regeneration. Cell, 2003, 113(7), 841-852.
[101]
Kuang, S.; Chargé, S.B.; Seale, P.; Huh, M.; Rudnicki, M.A. Distinct roles for Pax7 and Pax3 in adult regenerative myogenesis. J. Cell Biol., 2006, 172(1), 103-113.
[102]
Minasi, M.G.; Riminucci, M.; De Angelis, L.; Borello, U.; Berarducci, B.; Innocenzi, A.; Caprioli, A.; Sirabella, D.; Baiocchi, M.; De Maria, R.; Boratto, R.; Jaffredo, T.; Broccoli, V.; Bianco, P.; Cossu, G. The meso-angioblast: a multipotent, self-renewing cell that originates from the dorsal aorta and differentiates into most mesodermal tissues. Development, 2002, 129(11), 2773-2783.
[103]
Tonlorenzi, R.; Dellavalle, A.; Schnapp, E.; Cossu, G.; Sampaolesi, M. Isolation and characterization of mesoangioblasts from mouse, dog, and human tissues. Curr. Protoc. Stem Cell Biol., 2007, 2, 2B.1..
[104]
Gussoni, E.; Soneoka, Y.; Strickland, C.D.; Buzney, E.A.; Khan, M.K.; Flint, A.F.; Kunkel, L.M.; Mulligan, R.C. Dystrophin expression in the mdx mouse restored by stem cell transplantation. Nature, 1999, 401(6751), 390-394.
[105]
Asakura, A.; Rudnicki, M.A. Side population cells from diverse adult tissues are capable of in vitro hematopoietic differentiation. Exp. Hematol., 2002, 30(11), 1339-1345.
[106]
Torrente, Y.; Belicchi, M.; Sampaolesi, M.; Pisati, F.; Meregalli, M.; D’Antona, G.; Tonlorenzi, R.; Porretti, L.; Gavina, M.; Mamchaoui, K.; Pellegrino, M.A.; Furling, D.; Mouly, V.; Butler-Browne, G.S.; Bottinelli, R.; Cossu, G.; Bresolin, N. Human circulating AC133+ stem cells restore dystrophin expression and ameliorate function in dystrophic skeletal muscle. J. Clin. Invest., 2004, 114(2), 182-195.
[107]
Meeson, A.P.; Hawke, T.J.; Graham, S.; Jiang, N.; Elterman, J.; Hutcheson, K.; DiMaio, J.M.; Gallardo, T.D.; Garry, D.J. Cellular and molecular regulation of skeletal muscle side population cells. Stem Cells, 2004, 22(7), 1305-1320.
[108]
Montanaro, F.; Liadaki, K.; Schienda, J.; Flint, A.; Gussoni, E.; Kunkel, L.M. Demystifying SP cell purification: viability, yield, and phenotype are defined by isolation parameters. Exp. Cell Res., 2004, 298(1), 144-154.
[109]
Rivier, F.; Alkan, O.; Flint, A.F.; Muskiewicz, K.; Allen, P.D.; Leboulch, P.; Gussoni, E. Role of bone marrow cell trafficking in replenishing skeletal muscle SP and MP cell populations. J. Cell Sci., 2004, 117(10), 1979-1988.
[110]
Tanaka, K.K.; Hall, J.K.; Troy, A.A.; Cornelison, D.D.W.; Majka, S.M.; Olwin, B.B. Syndecan-4-expressing muscle progenitor cells in the SP engraft as satellite cells during muscle regeneration. Cell Stem Cell, 2009, 4(3), 217-225.
[111]
Sancricca, C.; Mirabella, M.; Gliubizzi, C.; Broccolini, A.; Gidaro, T.; Morosetti, R. Vessel-associated stem cells from skeletal muscle: From biology to future uses in cell therapy. World J. Stem Cells, 2010, 2(3), 39.
[112]
Quattrocelli, M.; Palazzolo, G.; Perini, I.; Crippa, S.; Cassano, M.; Sampaolesi, M. Mouse and human mesoangioblasts: Isolation and characterization from adult skeletal muscles. Methods Mol. Biol., 2012, 798, 65-76.
[113]
Sampaolesi, M.; Torrente, Y.; Innocenzi, A.; Tonlorenzi, R.; D’Antona, G.; Pellegrino, M.A.; Barresi, R.; Bresolin, N.; De Angelis, M.G.C.; Campbell, K.P.; Bottinelli, R.; Cossu, G. Cell therapy of -sarcoglycan null dystrophic mice through intra-arterial delivery of mesoangioblasts. Science, 2003, 301(5632), 487-492.
[114]
Sampaolesi, M.; Blot, S.; D’Antona, G.; Granger, N.; Tonlorenzi, R.; Innocenzi, A.; Mognol, P.; Thibaud, J-L.; Galvez, B.G.; Barthélémy, I.; Perani, L.; Mantero, S.; Guttinger, M.; Pansarasa, O.; Rinaldi, C.; Cusella De Angelis, M.G.; Torrente, Y.; Bordignon, C.; Bottinelli, R.; Cossu, G. Mesoangioblast stem cells ameliorate muscle function in dystrophic dogs. Nature, 2006, 444(7119), 574-579.
[115]
Cossu, G.; Previtali, S.C.; Napolitano, S.; Cicalese, M.P.; Tedesco, F.S.; Nicastro, F.; Noviello, M.; Roostalu, U.; Natali Sora, M.G.; Scarlato, M.; De Pellegrin, M.; Godi, C.; Giuliani, S.; Ciotti, F.; Tonlorenzi, R.; Lorenzetti, I.; Rivellini, C.; Benedetti, S.; Gatti, R.; Marktel, S.; Mazzi, B.; Tettamanti, A.; Ragazzi, M.; Imro, M.A.; Marano, G.; Ambrosi, A.; Fiori, R.; Sormani, M.P.; Bonini, C.; Venturini, M.; Politi, L.S.; Torrente, Y.; Ciceri, F. Intra-arterial transplantation of HLA-matched donor mesoangioblasts in Duchenne muscular dystrophy. EMBO Mol. Med., 2015, 7(12), 1513-1528.
[116]
Dellavalle, A.; Sampaolesi, M.; Tonlorenzi, R.; Tagliafico, E.; Sacchetti, B.; Perani, L.; Innocenzi, A.; Galvez, B.G.; Messina, G.; Morosetti, R.; Li, S.; Belicchi, M.; Peretti, G.; Chamberlain, J.S.; Wright, W.E.; Torrente, Y.; Ferrari, S.; Bianco, P.; Cossu, G. Pericytes of human skeletal muscle are myogenic precursors distinct from satellite cells. Nat. Cell Biol., 2007, 9(3), 255-267.
[117]
Dellavalle, A.; Maroli, G.; Covarello, D.; Azzoni, E.; Innocenzi, A.; Perani, L.; Antonini, S.; Sambasivan, R.; Brunelli, S.; Tajbakhsh, S.; Cossu, G. Pericytes resident in postnatal skeletal muscle differentiate into muscle fibres and generate satellite cells. Nat. Commun., 2011, 2(1), 499.
[118]
Crisan, M.; Yap, S.; Casteilla, L.; Chen, C-W.; Corselli, M.; Park, T.S.; Andriolo, G.; Sun, B.; Zheng, B.; Zhang, L.; Norotte, C.; Teng, P-N.; Traas, J.; Schugar, R.; Deasy, B.M.; Badylak, S.; Bűhring, H-J.; Giacobino, J-P.; Lazzari, L.; Huard, J.; Péault, B. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell, 2008, 3(3), 301-313.
[119]
Birbrair, A.; Zhang, T.; Wang, Z-M.; Messi, M.L.; Enikolopov, G.N.; Mintz, A.; Delbono, O. Role of pericytes in skeletal muscle regeneration and fat accumulation. Stem Cells Dev., 2013, 22(16), 2298-2314.
[120]
Scicchitano, B.M.; Dobrowolny, G.; Sica, G.; Musaro, A. Molecular insights into muscle homeostasis, atrophy and wasting. Curr. Genomics, 2018, 19(5), 356-369.
[121]
Scicchitano, B.M.; Pelosi, L.; Sica, G.; Musarò, A. The physiopathologic role of oxidative stress in skeletal muscle. Mech. Ageing Dev., 2018, 170, 37-44.
[122]
Franco, I.; Johansson, A.; Olsson, K.; Vrtačnik, P.; Lundin, P.; Helgadottir, H.T.; Larsson, M.; Revêchon, G.; Bosia, C.; Pagnani, A.; Provero, P.; Gustafsson, T.; Fischer, H.; Eriksson, M. Somatic mutagenesis in satellite cells associates with human skeletal muscle aging. Nat. Commun., 2018, 9(1), 800.
[123]
Hayflick, L. The limited in vitro lifetime of human diploid cell strains. Exp. Cell Res., 1965, 37(3), 614-636.
[124]
George, T.; Velloso, C.P.; Alsharidah, M.; Lazarus, N.R.; Harridge, S.D.R. Sera from young and older humans equally sustain proliferation and differentiation of human myoblasts. Exp. Gerontol., 2010, 45(11), 875-881.
[125]
Hikida, R.S. Aging changes in satellite cells and their functions. Curr. Aging Sci., 2011, 4(3), 279-297.
[126]
Tichy, E.D.; Sidibe, D.K.; Tierney, M.T.; Stec, M.J.; Sharifi-Sanjani, M.; Hosalkar, H.; Mubarak, S.; Johnson, F.B.; Sacco, A.; Mourkioti, F. Single stem cell imaging and analysis reveals telomere length differences in diseased human and mouse skeletal muscles. Stem Cell Reports, 2017, 9(4), 1328-1341.
[127]
Decary, S.; Hamida, C.B.; Mouly, V.; Barbet, J.P.; Hentati, F.; Butler-Browne, G.S. Shorter telomeres in dystrophic muscle consistent with extensive regeneration in young children. Neuromuscul. Disord., 2000, 10(2), 113-120.
[128]
Pelosi, L.; Berardinelli, M.G.; Forcina, L.; Spelta, E.; Rizzuto, E.; Nicoletti, C.; Camilli, C.; Testa, E.; Catizone, A.; De Benedetti, F.; Musarò, A. Increased levels of interleukin-6 exacerbate the dystrophic phenotype in mdx mice. Hum. Mol. Genet., 2015, 24(21), 6041-6053.
[129]
Bernet, J.D.; Doles, J.D.; Hall, J.K.; Kelly Tanaka, K.; Carter, T.A.; Olwin, B.B. p38 MAPK signaling underlies a cell-autonomous loss of stem cell self-renewal in skeletal muscle of aged mice. Nat. Med., 2014, 20(3), 265-271.
[130]
Price, F.D.; von Maltzahn, J.; Bentzinger, C.F.; Dumont, N.A.; Yin, H.; Chang, N.C.; Wilson, D.H.; Frenette, J.; Rudnicki, M.A. Inhibition of JAK-STAT signaling stimulates adult satellite cell function. Nat. Med., 2014, 20(10), 1174-1181.
[131]
Sousa-Victor, P.; Gutarra, S.; García-Prat, L.; Rodriguez-Ubreva, J.; Ortet, L.; Ruiz-Bonilla, V.; Jardí, M.; Ballestar, E.; González, S.; Serrano, A.L.; Perdiguero, E.; Muñoz-Cánoves, P. Geriatric muscle stem cells switch reversible quiescence into senescence. Nature, 2014, 506(7488), 316-321.
[132]
Carlson, B.M.; Faulkner, J.A. Muscle transplantation between young and old rats: age of host determines recovery. Am. J. Physiol., 1989, 256(6 Pt 1), C1262-C1266.
[133]
Carlson, B.M.; Dedkov, E.I.; Borisov, A.B.; Faulkner, J.A. Skeletal muscle regeneration in very old rats. J. Gerontol. A Biol. Sci. Med. Sci., 2001, 56(5), B224-B233.
[134]
Conboy, I.M.; Conboy, M.J.; Wagers, A.J.; Girma, E.R.; Weissman, I.L.; Rando, T.A. Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature, 2005, 433(7027), 760-764.
[135]
Daynes, R.A.; Araneo, B.A.; Ershler, W.B.; Maloney, C.; Li, G.Z.; Ryu, S.Y. Altered regulation of IL-6 production with normal aging. Possible linkage to the age-associated decline in dehydroepiandrosterone and its sulfated derivative. J. Immunol., 1993, 150(12), 5219-5230.
[136]
Paliwal, P.; Pishesha, N.; Wijaya, D.; Conboy, I.M. Age dependent increase in the levels of osteopontin inhibits skeletal muscle regeneration. Aging (Albany N.Y.), 2012, 4(8), 553-566.
[137]
McKay, B.R.; Ogborn, D.I.; Baker, J.M.; Toth, K.G.; Tarnopolsky, M.A.; Parise, G. Elevated SOCS3 and altered IL-6 signaling is associated with age-related human muscle stem cell dysfunction. Am. J. Physiol. Cell Physiol., 2013, 304(8), C717-C728.
[138]
Barbieri, M.; Ferrucci, L.; Ragno, E.; Corsi, A.; Bandinelli, S.; Bonafè, M.; Olivieri, F.; Giovagnetti, S.; Franceschi, C.; Guralnik, J.M.; Paolisso, G. Chronic inflammation and the effect of IGF-I on muscle strength and power in older persons. Am. J. Physiol. Endocrinol. Metab., 2003, 284(3), E481-E487.
[139]
Hirata, A.; Masuda, S.; Tamura, T.; Kai, K.; Ojima, K.; Fukase, A.; Motoyoshi, K.; Kamakura, K.; Miyagoe-Suzuki, Y.; Takeda, S. Expression profiling of cytokines and related genes in regenerating skeletal muscle after cardiotoxin injection. Am. J. Pathol., 2003, 163(1), 203-215.
[140]
Uaesoontrachoon, K.; Yoo, H-J.; Tudor, E.M.; Pike, R.N.; Mackie, E.J.; Pagel, C.N. Osteopontin and skeletal muscle myoblasts: Association with muscle regeneration and regulation of myoblast function in vitro. Int. J. Biochem. Cell Biol., 2008, 40(10), 2303-2314.
[141]
Vetrone, S.A.; Montecino-Rodriguez, E.; Kudryashova, E.; Kramerova, I.; Hoffman, E.P.; Liu, S.D.; Miceli, M.C.; Spencer, M.J. Osteopontin promotes fibrosis in dystrophic mouse muscle by modulating immune cell subsets and intramuscular TGF-beta. J. Clin. Invest., 2009, 119(6), 1583-1594.
[142]
Kuswanto, W.; Burzyn, D.; Panduro, M.; Wang, K.K.; Jang, Y.C.; Wagers, A.J.; Benoist, C.; Mathis, D. Poor repair of skeletal muscle in aging mice reflects a defect in local, interleukin-33-dependent accumulation of regulatory T cells. Immunity, 2016, 44(2), 355-367.
[143]
Uezumi, A.; Fukada, S.; Yamamoto, N.; Takeda, S.; Tsuchida, K. Mesenchymal progenitors distinct from satellite cells contribute to ectopic fat cell formation in skeletal muscle. Nat. Cell Biol., 2010, 12(2), 143-152.
[144]
Uezumi, A.; Ito, T.; Morikawa, D.; Shimizu, N.; Yoneda, T.; Segawa, M.; Yamaguchi, M.; Ogawa, R.; Matev, M.M.; Miyagoe-Suzuki, Y.; Takeda, S.; Tsujikawa, K.; Tsuchida, K.; Yamamoto, H.; Fukada, S-i. Fibrosis and adipogenesis originate from a common mesenchymal progenitor in skeletal muscle. J. Cell Sci., 2011, 124(21), 3654-3664.
[145]
Pelosi, L.; Coggi, A.; Forcina, L.; Musarò, A. MicroRNAs modulated by local mIGF-1 expression in mdx dystrophic mice. Front. Aging Neurosci., 2015, 7, 69.
[146]
Barton, E.R.; Morris, L.; Musaro, A.; Rosenthal, N.; Sweeney, H.L. Muscle-specific expression of insulin-like growth factor I counters muscle decline in mdx mice. J. Cell Biol., 2002, 157(1), 137-148.
[147]
Petrillo, S.; Pelosi, L.; Piemonte, F.; Travaglini, L.; Forcina, L.; Catteruccia, M.; Petrini, S.; Verardo, M.; D’Amico, A.; Musarò, A.; Bertini, E. Oxidative stress in Duchenne muscular dystrophy: focus on the NRF2 redox pathway. Hum. Mol. Genet., 2017, 26(14), 2781-2790.
[148]
Pelosi, L.; Forcina, L.; Nicoletti, C.; Scicchitano, B.M.; Musarò, A. Increased circulating levels of interleukin-6 induce perturbation in redox-regulated signaling cascades in muscle of dystrophic mice. Oxid. Med. Cell. Longev., 2017, 2017, 1-10.
[149]
Forcina, L.; Pelosi, L.; Miano, C.; Musarò, A.; Forcina, L.; Pelosi, L.; Miano, C.; Musarò, A. Insights into the pathogenic secondary symptoms caused by the primary loss of dystrophin. J. Funct. Morphol. Kinesiol., 2017, 2(4), 44.
[150]
Pelosi, L.; Berardinelli, M.G.; De Pasquale, L.; Nicoletti, C.; D’Amico, A.; Carvello, F.; Moneta, G.M.; Catizone, A.; Bertini, E.; De Benedetti, F.; Musarò, A. Functional and morphological improvement of dystrophic muscle by interleukin 6 receptor blockade. EBioMed, 2015, 2(4), 285-293.