Abstract
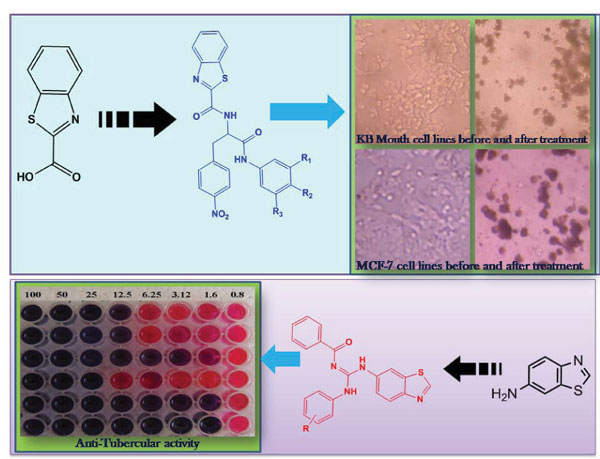
Background: Two series of Guanidinyl benzothiazole and benzothiazole diamide derivatives were synthesized and screened for their anti-mycobacterial activity and cytotoxicity on cancer cell lines.
Methods: The anti-mycobacterium study indicates that all the synthesized benzothiazole compounds were appreciably active and some of the compounds have MIC values lower than the standard drugs. Benzothiazoleguanidinyl derivatives (13a, 13b and 13f) showed the excellent activity with MIC values 1.6 µg/mL. The guanidinyl group and electron donating group present in the molecule interacts with the microorganism and arrest the further growth, indicating excellent activity by these compounds. These benzothiazole derivatives were also tested for cytotoxicity against MCF-7 and KB Mouth cell lines by MTT assay and they were found to be moderately active.
Results: For the KB-Mouth cell lines, diamide compounds (9a-9h) have remarkable activity and they showed IC50 values at 10 µg/mL. Compared to benzothiazole diamides, Benzothiazole guanidinyl compounds selectively acted as a good anti-mycobacterium agent.
Conclusion:In order to rationalize the in-vitro anti-tuberculosis activity, we carried out molecular docking studies with enoyl acyl carrier reductase (InhA) of M. tuberculosis and they exhibited remarkable docking scores from -5.85 to -9.27, which was comparable with the positive control Isoniazid (INH) with -6.61 as the docking score and showed less affinity towards the DprE1 protein.
Keywords: Guanidinyl benzothiazole, benzothiazole amide, anti-mycobacterial activity, IC50 value, MCF-7, KB- mouth cell lines.