Abstract
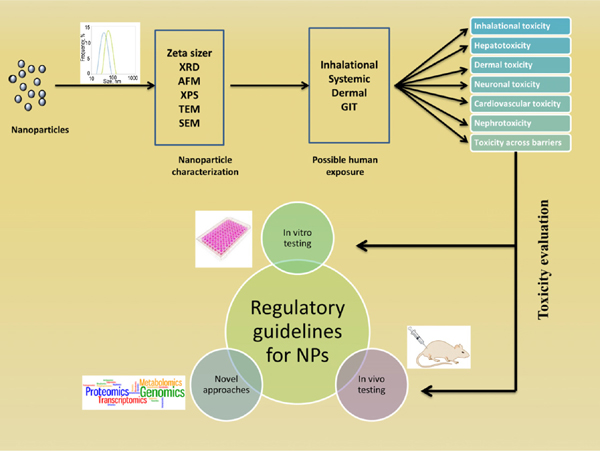
Background: The rapid progress in the application of different types of nanoparticles in various biomedical applications resulted in substantial increase in the use of these materials. The special physicochemical properties like small size and increased surface area offered by nanoparticles may pose unexpected toxicologic threats despite of offering promising biological effects.
Objective: The intended as well as unintended exposure of nanoparticles to humans is a major challenge for the expanding field of nanoparticle therapy. In this review, an attempt has been made to briefly discuss the different factors affecting nanoparticle mediated toxicity, mechanism of toxicity, and interference of NPs with bioassays. A comprehensive overview has been provided for the in vitro and in vivo toxicologic assessment of NPs and what and how framing guidelines should be used for global unification of nanotoxicologic studies.
Conclusion: The present review entails a discussion of the toxicity of nanoparticles along with interferences inherent to nanoparticles in toxicity studies. The lack of availability of regulatory guidelines for nanoparticles is one of the biggest issues. Our understanding of the mechanisms of NP-induced toxicity is superficial; thus, it is too early to come to a general consensus on the toxicity of NPs. There is a need, therefore, to standardize and validate the toxicity testing assays based on NPs properties.
Keywords: Cytotoxicity, nanoparticles, nanotoxicology, omics, regulatory guidelines, reticuloendothelial system.