Abstract
Introduction: Histone deacetylase (HDAC) enzymes control the acetylation status of transcription factors that regulate chromatin structure and gene function. The transcriptional regulatory factors that distinguish histone acetylation and deacetylation patterns by pharmacological HDAC inhibition (HDACi) have not yet been studied.
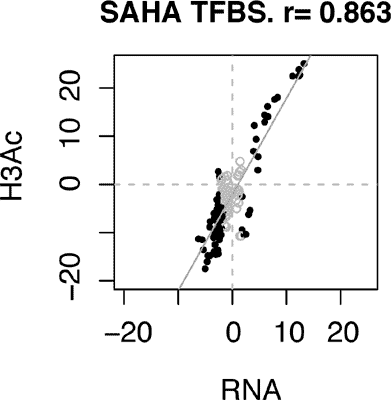
Methods: We analysed sequencing datasets derived from human aortic endothelial cells (HAECs) stimulated with the HDAC inhibitors, Trichostatin A (TSA) and suberoylanilide hydroxamic acid (SAHA). We integrated gene expression and histone acetylation profiles with the transcription factor binding site (TFBS) database derived from the Encyclopedia of DNA Elements (ENCODE) project. Results: Overall, TFBS signatures observed in SAHA and TSA stimulated cells were analogous. Histone acetylation was observed at transcription factor binding sites of target genes associated with the silencing factors NRSF, EZH2 and SUZ12. Histone deacetylation was a prominent property of HDACi and correlated with changes in the expression of genes regulated by proteins in transcriptional control such as histone acetyltransferase P300 and lysine demethylase JARID1A, as well as the regulatory factors cMYC, YY1 and STAT family proteins. Conclusion: We identified several transcription factors and coregulators implicated in the regulation of histone modification at target genes mediated by pharmacological HDAC inhibition.Keywords: Epigenetics, Deacetylation, Histone Acetylation, Transcription factor, Suberoylanilide Hydroxamic acid, Trichostatin A.
Current Topics in Medicinal Chemistry
Title:Pharmacological Histone Deacetylation Distinguishes Transcriptional Regulators
Volume: 17 Issue: 14
Author(s): Haloom Rafehi, Tom C. Karagiannis and Assam El-Osta*
Affiliation:
- Epigenetics in Human Health and Disease, Baker IDI Heart and Diabetes Institute, 75 Commercial Road, Melbourne, VIC,Australia
Keywords: Epigenetics, Deacetylation, Histone Acetylation, Transcription factor, Suberoylanilide Hydroxamic acid, Trichostatin A.
Abstract: Introduction: Histone deacetylase (HDAC) enzymes control the acetylation status of transcription factors that regulate chromatin structure and gene function. The transcriptional regulatory factors that distinguish histone acetylation and deacetylation patterns by pharmacological HDAC inhibition (HDACi) have not yet been studied.
Methods: We analysed sequencing datasets derived from human aortic endothelial cells (HAECs) stimulated with the HDAC inhibitors, Trichostatin A (TSA) and suberoylanilide hydroxamic acid (SAHA). We integrated gene expression and histone acetylation profiles with the transcription factor binding site (TFBS) database derived from the Encyclopedia of DNA Elements (ENCODE) project. Results: Overall, TFBS signatures observed in SAHA and TSA stimulated cells were analogous. Histone acetylation was observed at transcription factor binding sites of target genes associated with the silencing factors NRSF, EZH2 and SUZ12. Histone deacetylation was a prominent property of HDACi and correlated with changes in the expression of genes regulated by proteins in transcriptional control such as histone acetyltransferase P300 and lysine demethylase JARID1A, as well as the regulatory factors cMYC, YY1 and STAT family proteins. Conclusion: We identified several transcription factors and coregulators implicated in the regulation of histone modification at target genes mediated by pharmacological HDAC inhibition.Export Options
About this article
Cite this article as:
Rafehi Haloom, Karagiannis C. Tom and El-Osta Assam*, Pharmacological Histone Deacetylation Distinguishes Transcriptional Regulators, Current Topics in Medicinal Chemistry 2017; 17 (14) . https://dx.doi.org/10.2174/1568026617666161104104341
| DOI https://dx.doi.org/10.2174/1568026617666161104104341 |
Print ISSN 1568-0266 |
| Publisher Name Bentham Science Publisher |
Online ISSN 1873-4294 |
Call for Papers in Thematic Issues
Chemistry Based on Natural Products for Therapeutic Purposes
The development of new pharmaceuticals for a wide range of medical conditions has long relied on the identification of promising natural products (NPs). There are over sixty percent of cancer, infectious illness, and CNS disease medications that include an NP pharmacophore, according to the Food and Drug Administration. Since NP ...read more
Current Trends in Drug Discovery Based on Artificial Intelligence and Computer-Aided Drug Design
Drug development discovery has faced several challenges over the years. In fact, the evolution of classical approaches to modern methods using computational methods, or Computer-Aided Drug Design (CADD), has shown promising and essential results in any drug discovery campaign. Among these methods, molecular docking is one of the most notable ...read more
Drug Discovery in the Age of Artificial Intelligence
In the age of artificial intelligence (AI), we have witnessed a significant boom in AI techniques for drug discovery. AI techniques are increasingly integrated and accelerating the drug discovery process. These developments have not only attracted the attention of academia and industry but also raised important questions regarding the selection ...read more
From Biodiversity to Chemical Diversity: Focus of Flavonoids
Flavonoids are the largest group of polyphenols, plant secondary metabolites arising from the essential aromatic amino acid phenylalanine (or more rarely from tyrosine) via the phenylpropanoid pathway. The flavan nucleus is the basic 15-carbon skeleton of flavonoids (C6-C3-C6), which consists of two phenyl rings (A and B) and a heterocyclic ...read more
 46
46 3
3 1
1
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
- Announcements
Related Articles
-
Mitochondrially-Targeted Therapeutic Strategies for Alzheimer’s Disease
Current Alzheimer Research CT-negative Subarachnoid Hemorrhage Caused by Telangiectasia: A Case Report
Current Medical Imaging Biochemical, Enzymatic and Molecular Properties of Renin/Prorenin Mediated by (Pro)Renin Receptor
Immunology, Endocrine & Metabolic Agents in Medicinal Chemistry (Discontinued) Update on Pharmacological Treatment of Progressive Myoclonus Epilepsies
Current Pharmaceutical Design Anticancer Mammalian Target of Rapamycin (mTOR) Signaling Pathway Inhibitors: Current Status, Challenges and Future Prospects in Management of Epilepsy
CNS & Neurological Disorders - Drug Targets <i>Ajuga</i> L.: A Systematic Review on Chemical Composition, Phytopharmacological and Biological Potential
Current Bioactive Compounds Valproate Induced Acute Pancreatitis - A Unique Case Report
Current Drug Safety Design, Synthesis and Biological Evaluation of Benzimidazole-pyridine- Piperidine Hybrids as a New Class of Potent Antimicrobial Agents
Letters in Drug Design & Discovery SkQ-1 Regulates Xanthine Oxidase Activity in the Settings of Epilepsic Seizures in Rats
Neuroscience and Biomedical Engineering (Discontinued) Increased Risk of Falls, Fall-related Injuries and Fractures in People with Type 1 and Type 2 Diabetes - A Nationwide Cohort Study
Current Drug Safety Therapeutic Potential of Metabotropic Glutamate Receptor Modulators
Current Neuropharmacology Xenobiotic Acyl Glucuronides and Acyl CoA Thioesters as Protein- Reactive Metabolites With the Potential to Cause Idiosyncratic Drug Reactions
Current Drug Metabolism Extracellular Level of GABA and Glu: In Vivo Microdialysis-HPLC Measurements
Current Topics in Medicinal Chemistry Zebrafish as a Platform for Studies on Seizures and Epilepsy
Current Psychopharmacology Role of Gap Junction Channel in the Development of Beat-to-Beat Action Potential Repolarization Variability and Arrhythmias
Current Pharmaceutical Design Patent Selections
Recent Patents on CNS Drug Discovery (Discontinued) From Cannabis to Cannabidiol to Treat Epilepsy, Where Are We?
Current Pharmaceutical Design Pharmacogenomics of Cardiovascular Complications in Diabetes and Obesity
Recent Patents on Biotechnology Localised Delivery of Therapeutic Agents to CNS Malignancies: Old and New Approaches
Current Pharmaceutical Biotechnology synthesis and Structure-Activity Relationships of 2,3-Benzodiazepines as AMPA Receptor Antagonists
Mini-Reviews in Medicinal Chemistry