Abstract
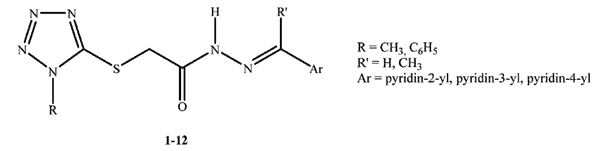
2-[(1-Methyl-1H-tetrazol-5-yl)thio]-N'-[(aryl)methylidene/ethylidene]acetohydrazides were synthesized and investigatedin vitroagainst pathogenic bacteriaand Candidaspecies for their antimicrobial activityusing CLSI broth microdilution method. MTT assay was also carried out to evaluate their cytotoxic effects on NIH/3T3 mouse embryonic fibroblast cell lines.2-((1-Phenyl-1H-tetrazol-5-yl)thio)-N'-(1-(pyridin-3-yl)ethylidene)acetohydrazide can be considered as the most promising antibacterial agent againstEnterococcusfaecalis (ATCC 29212) with a MIC value of 100 µg/mL when compared with chloramphenicol (MIC= 200 µg/mL) and low toxicity against NIH/3T3 cells(IC50>500µg/mL).
Keywords: Antimicrobial activity, cytotoxicity, hydrazone, pyridine, tetrazole.
Letters in Drug Design & Discovery
Title:Synthesis and Evaluation of Tetrazole-BasedHydrazone Derivatives Bearing a Pyridine Moiety as Antimicrobial Agents
Volume: 12 Issue: 8
Author(s): Ahmet Ozdemir, Mehlika Dilek Altıntop, Belgin Sever, Zerrin Canturk and Zafer Asım Kaplancıkl
Affiliation:
Keywords: Antimicrobial activity, cytotoxicity, hydrazone, pyridine, tetrazole.
Abstract: 2-[(1-Methyl-1H-tetrazol-5-yl)thio]-N'-[(aryl)methylidene/ethylidene]acetohydrazides were synthesized and investigatedin vitroagainst pathogenic bacteriaand Candidaspecies for their antimicrobial activityusing CLSI broth microdilution method. MTT assay was also carried out to evaluate their cytotoxic effects on NIH/3T3 mouse embryonic fibroblast cell lines.2-((1-Phenyl-1H-tetrazol-5-yl)thio)-N'-(1-(pyridin-3-yl)ethylidene)acetohydrazide can be considered as the most promising antibacterial agent againstEnterococcusfaecalis (ATCC 29212) with a MIC value of 100 µg/mL when compared with chloramphenicol (MIC= 200 µg/mL) and low toxicity against NIH/3T3 cells(IC50>500µg/mL).
Export Options
About this article
Cite this article as:
Ozdemir Ahmet, Dilek Altıntop Mehlika, Sever Belgin, Canturk Zerrin and Asım Kaplancıkl Zafer, Synthesis and Evaluation of Tetrazole-BasedHydrazone Derivatives Bearing a Pyridine Moiety as Antimicrobial Agents, Letters in Drug Design & Discovery 2015; 12 (8) . https://dx.doi.org/10.2174/1570180812666150309235217
| DOI https://dx.doi.org/10.2174/1570180812666150309235217 |
Print ISSN 1570-1808 |
| Publisher Name Bentham Science Publisher |
Online ISSN 1875-628X |
 25
25 1
1
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
Related Articles
-
Nitric Oxide-Releasing Biomaterials for Biomedical Applications
Current Medicinal Chemistry Genetics of Cancer Susceptibility
Current Genomics Recent Progress in Phosphoinositide 3-Kinases: Oncogenic Properties and Prognostic and Therapeutic Implications
Current Protein & Peptide Science Aldehyde Dehydrogenase as a Marker for Stem Cells
Current Stem Cell Research & Therapy Transporters at CNS Barrier Sites: Obstacles or Opportunities for Drug Delivery?
Current Pharmaceutical Design Therapeutic Potential of Plant Extracts and Phytochemicals Against Brain Ischemia-Reperfusion Injury: A Review
The Natural Products Journal Computer-Aided Drug Design of Bioactive Natural Products
Current Topics in Medicinal Chemistry Chromogranin A and the Endothelial Barrier Function
Current Medicinal Chemistry Doripenem: A New Addition to the Carbapenem Class of Antimicrobials
Recent Patents on Anti-Infective Drug Discovery Macromolecular Drug Targets in Cancer Treatment and Thiosemicarbazides as Anticancer Agents
Anti-Cancer Agents in Medicinal Chemistry Approaches to Gastrointestinal Cytoprotection: From Isolated Cells, Via Animal Experiments to Healthy Human Subjects and Patients with Different Gastrointestinal Disorders
Current Pharmaceutical Design The Discovery of Antiangiogenic Molecules: A Historical Review
Current Pharmaceutical Design Targeting ErbB Receptors in High-Grade Glioma
Current Pharmaceutical Design Tribbles-Related Protein Family Members as Regulators or Substrates of the Ubiquitin-Proteasome System in Cancer Development
Current Cancer Drug Targets Synthesis and Anti-Breast Cancer Activity of Tetrasubstituted Pyrrole Derivatives
Letters in Drug Design & Discovery Cancer Cell-derived Secretory Factors in Breast Cancer-associated Lung Metastasis: Their Mechanism and Future Prospects
Current Cancer Drug Targets Environmental Chemicals and Breast Cancer Risk – A Structural Chemistry Perspective
Current Medicinal Chemistry 1,3,4-oxadiazole-2-thione Derivatives; Novel Approach for Anticancer and Tubulin Polymerization Inhibitory Activities
Anti-Cancer Agents in Medicinal Chemistry Inhibition of Sulfotransferases by Xenobiotics
Current Drug Metabolism Prediction of the Estrogen Receptor Binding Affinity for both hER<sub>α</sub> and hER<sub>β</sub> by QSAR Approaches
Letters in Drug Design & Discovery