Abstract
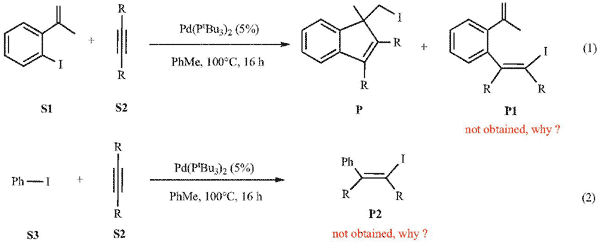
The reactions of 2-iodo-α-methyl styrene (S1) with diphenyl acetylene (S2) (equation 1) and iodo-benzene (S3) with diphenyl acetylene (S2) (equation 2) have been theoretically studied with the aid of density functional theory calculations. Equation 1 reaction involves four major steps, aryl-I oxidative addition, alkyne insertion, C=C bond insertion, and (sp3)C-I reductive elimination. Equation 2 reaction involves aryl-I oxidative addition, alkyne insertion, and further alkyne insertion. Based on the mechanistic study, we revealed why the indene product P could be obtained in equation 1 while the proposed product P2 could not be obtained in equation 2. In addition, the bulkiness of the ligand PtBu3 plays an important role for the steps involved undergoing mono-L or non-L paths.
Keywords: Alkyne insertion, aryl iodide, carbohelogenation, DFT calculation, steric effect.