Abstract
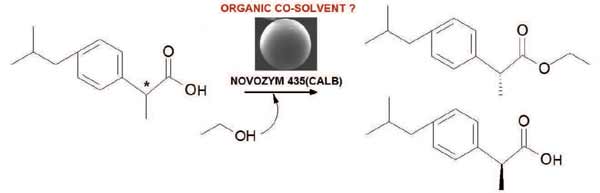
The commercial biocatalyst Novozym ® 435 was used for the kinetic resolution of (R/S)-ibuprofen through the esterification with short chain alcohols (ethanol, 1-propanol and 2-propanol) in the absence of organic co-solvent. The best enzymatic performance was obtained by employing ethanol as reagent and solvent. Due to the deleterious effect of this alcohol on the integrity of the commercial biocatalyst previously reported different organic co-solvents (isooctane, nhexane, carbon tetrachloride, ethyl acetate, acetonitrile and tetrahydrofuran) were screened in order to minimize the volume of ethanol to be used. Thus, the effect of the chemical nature of the co-solvent on enantioselective esterification of (R/S)-ibuprofen with ethanol was evaluated. The results show that the best performance was obtained with the reaction system without co-solvent added. Additionally, this investigation demonstrated the need to address multiple physicochemical properties of the solvents to analyze their effects on biocatalysis.
Keywords: Ibuprofen, Novozym435, Ethanol, Organic solvents.
Current Catalysis
Title:Effect of Co-solvents in the Enantioselective Esterification of (R/S)- ibuprofen with Ethanol
Volume: 3 Issue: 2
Author(s): Carla Jose, M. Victoria Toledo, Jaiver Osorio Grisales and Laura E. Briand
Affiliation:
Keywords: Ibuprofen, Novozym435, Ethanol, Organic solvents.
Abstract: The commercial biocatalyst Novozym ® 435 was used for the kinetic resolution of (R/S)-ibuprofen through the esterification with short chain alcohols (ethanol, 1-propanol and 2-propanol) in the absence of organic co-solvent. The best enzymatic performance was obtained by employing ethanol as reagent and solvent. Due to the deleterious effect of this alcohol on the integrity of the commercial biocatalyst previously reported different organic co-solvents (isooctane, nhexane, carbon tetrachloride, ethyl acetate, acetonitrile and tetrahydrofuran) were screened in order to minimize the volume of ethanol to be used. Thus, the effect of the chemical nature of the co-solvent on enantioselective esterification of (R/S)-ibuprofen with ethanol was evaluated. The results show that the best performance was obtained with the reaction system without co-solvent added. Additionally, this investigation demonstrated the need to address multiple physicochemical properties of the solvents to analyze their effects on biocatalysis.
Export Options
About this article
Cite this article as:
Jose Carla, Toledo Victoria M., Grisales Osorio Jaiver and Briand E. Laura, Effect of Co-solvents in the Enantioselective Esterification of (R/S)- ibuprofen with Ethanol, Current Catalysis 2014; 3 (2) . https://dx.doi.org/10.2174/2211544702666131230234058
| DOI https://dx.doi.org/10.2174/2211544702666131230234058 |
Print ISSN 2211-5447 |
| Publisher Name Bentham Science Publisher |
Online ISSN 2211-5455 |
 30
30
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
- Announcements
Related Articles
-
Pro- and Anti-Inflammatory Cytokine Expression Levels in Macrophages; An Approach to Develop Indazolpyridin-methanones as Novel Inflammation Medication
Anti-Inflammatory & Anti-Allergy Agents in Medicinal Chemistry Mucosal Healing in Ulcerative Colitis: Where do we Stand?
Current Drug Targets Fibroblast Growth Factors/Fibroblast Growth Factor Receptors as Targets for the Development of Anti-Angiogenesis Strategies
Current Pharmaceutical Design Insights Into the Role of microRNAs in Cardiac Diseases: From Biological Signalling to Therapeutic Targets
Cardiovascular & Hematological Agents in Medicinal Chemistry Genetics of Cholesterol and Lipoprotein Metabolism
Recent Patents on Cardiovascular Drug Discovery Correlation between MDSC and Immune Tolerance in Transplantation: Cytokines, Pathways and Cell-cell Interaction
Current Gene Therapy Inhibition of Matrix Metalloproteinases as a Feasible Therapeutic Target in Rheumatoid Arthritis
Current Rheumatology Reviews Foreword
Current Nutrition & Food Science Therapeutic Potential and Pharmaceutical Applications of <i>Cucurbita</i>
Current Nutrition & Food Science Targeting the Assembly of the Human Immunodeficiency Virus Type I
Current Pharmaceutical Design Molecular Pharmacological Approaches to Effects of Capsaicinoids and of Classical Antisecretory Drugs on Gastric Basal Acid Secretion and on Indomethacin-Induced Gastric Mucosal Damage in Human Healthy Subjects (Mini Review)
Current Pharmaceutical Design Synthesis and Biological Activity of 4-Amino-1-Methyl-5-Imidazolecarboxylic Acid Derivatives
Letters in Drug Design & Discovery The Impact of Polyphenolics in the Management of Breast Cancer: Mechanistic Aspects and Recent Patents
Recent Patents on Anti-Cancer Drug Discovery Neuroprotective Effects of Fisetin in Alzheimer’s and Parkinson’s Diseases: From Chemistry to Medicine
Current Topics in Medicinal Chemistry Alcohol and the Cardiovascular System: A Double-Edged Sword
Current Pharmaceutical Design Novel Therapeutic Agents for Hyperuricemia in Patients with Gout
Current Drug Therapy A Comprehensive Review on the Screening Models for the Pharmacological Assessment of Antiulcer Drugs
Current Clinical Pharmacology EDITOR’S PERSPECTIVE [The Challenges for Drug Development: Cytokines, Genes, and Stem Cells]
Current Neurovascular Research Targeted Vascular Drug Delivery in Cerebral Cancer
Current Pharmaceutical Design Src Inhibitors and Angiogenesis
Current Pharmaceutical Design