Abstract
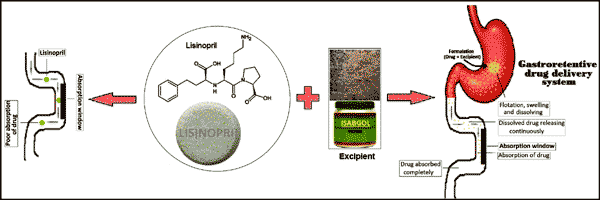
The gastroretentive drug delivery system is site-specific and allows the drug to remain in the stomach for a prolonged period of time so that it can be released in a controlled manner in gastrointestinal tract. The present study was carried out to develop a gastroretentive drug delivery system using isabgol as an excipient to prolong the residence time of the model drug lisinopril in the stomach. The gastroretentive ability of isabgol was increased by addition of NaHCO3 as a gas-generating agent while its mucoadhesive property was enhanced by incorporation of HPMC-K4M. The drug, NaHCO3 and HPMC-K3M were imbibed on isabgol-husk as per entrapment efficiency of the isabgol-husk. After drying, the product was filled in a hard gelatin capsule and evaluated for its buoyancy, mucoadhesive properties, swelling index and in vitro drug release. The lisinopril released through isabgol was delayed by 12 hours when compared to a preparation available on the market which released the complete drug in 0.5 hours. The drug release study of lisinopril from the formulation follows first order kinetics using a diffusion controlled mechanism. The results from the present study revealed that isabgol can be used as a potential excipient for the formulation of gastroretentive drug delivery systems in the near future.
Keywords: Detachment force, Gastroretentive drug delivery, Lisinopril, Mucoadhesive, Swelling power.
Current Drug Delivery
Title:A Gastroretentive Drug Delivery System of Lisinopril Imbibed on Isabgol- Husk
Volume: 11 Issue: 3
Author(s): Ravindra Semwal, Ruchi Badoni Semwal and Deepak Kumar Semwal
Affiliation:
Keywords: Detachment force, Gastroretentive drug delivery, Lisinopril, Mucoadhesive, Swelling power.
Abstract: The gastroretentive drug delivery system is site-specific and allows the drug to remain in the stomach for a prolonged period of time so that it can be released in a controlled manner in gastrointestinal tract. The present study was carried out to develop a gastroretentive drug delivery system using isabgol as an excipient to prolong the residence time of the model drug lisinopril in the stomach. The gastroretentive ability of isabgol was increased by addition of NaHCO3 as a gas-generating agent while its mucoadhesive property was enhanced by incorporation of HPMC-K4M. The drug, NaHCO3 and HPMC-K3M were imbibed on isabgol-husk as per entrapment efficiency of the isabgol-husk. After drying, the product was filled in a hard gelatin capsule and evaluated for its buoyancy, mucoadhesive properties, swelling index and in vitro drug release. The lisinopril released through isabgol was delayed by 12 hours when compared to a preparation available on the market which released the complete drug in 0.5 hours. The drug release study of lisinopril from the formulation follows first order kinetics using a diffusion controlled mechanism. The results from the present study revealed that isabgol can be used as a potential excipient for the formulation of gastroretentive drug delivery systems in the near future.
Export Options
About this article
Cite this article as:
Semwal Ravindra, Semwal Badoni Ruchi and Semwal Kumar Deepak, A Gastroretentive Drug Delivery System of Lisinopril Imbibed on Isabgol- Husk, Current Drug Delivery 2014; 11 (3) . https://dx.doi.org/10.2174/15672018113106660065
| DOI https://dx.doi.org/10.2174/15672018113106660065 |
Print ISSN 1567-2018 |
| Publisher Name Bentham Science Publisher |
Online ISSN 1875-5704 |
Call for Papers in Thematic Issues
Advances of natural products, bio-actives and novel drug delivery system against emerging viral infections
Due to the increasing prevalence of viral infections and the ability of these human pathogens to develop resistance to current treatment strategies, there is a great need to find and develop new compounds to combat them. These molecules must have low toxicity, specific activity and high bioavailability. The most suitable ...read more
Electrospun Fibers as Drug Delivery Systems
In recent years, electrospun fibers have attracted considerable attention as potential platforms for drug delivery due to their distinctive properties and adaptability. These fibers feature a notable surface area-to-volume ratio and can be intentionally designed with high porosity, facilitating an increased capacity for drug loading and rendering them suitable for ...read more
Emerging Nanotherapeutics for Mitigation of Neurodegenerative Disorders
Conditions affecting the central nervous system (CNS) present a significant hurdle due to limited access of both treatments and diagnostic tools for the brain. The blood-brain barrier (BBB) acts as a barrier, restricting the passage of molecules from the bloodstream into the brain. The most formidable challenge facing scientists is ...read more
Nanotechnology Based Chemotherapy for the treatment of Head & Neck Cancer
The escalating recurrence rates observed in Head and Neck cancer, particularly within the chemo-therapeutically treated cohort (50-60%), can be attributed to the non-selective nature of current anticancer drug delivery modalities. In this context, nanotechnology-based drug delivery systems emerge as a promising avenue for achieving precise localization of therapeutic agents to ...read more
 37
37
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
Related Articles
-
Recent Advancement and Technological Aspects of Pulsatile Drug Delivery System - A Laconic Review
Current Drug Targets The Use of Dexmedetomidine in Anesthesia and Intensive Care: A Review
Current Pharmaceutical Design NAD+, Sirtuins, and Cardiovascular Disease
Current Pharmaceutical Design New Antihypertensive Drugs Under Development
Current Medicinal Chemistry Polymorphism in Endothelin-1 Gene: An Overview
Current Clinical Pharmacology Patents on Technologies of Human Tissue and Organ Regeneration from Pluripotent Human Embryonic Stem Cells
Recent Patents on Regenerative Medicine Editorial [Hot topic: The Inflammation Paradigm in Cardiovascular Disease (Guest Editor: Guha Krishnaswamy)]
Cardiovascular & Hematological Disorders-Drug Targets The Effects of Glucose-Lowering Therapies on Diabetic Kidney Disease
Current Diabetes Reviews 23Na Magnetic Resonance Imaging for the Determination of Myocardial Viability: The Status and the Challenges
Current Vascular Pharmacology Sp/KLF Family and Tumor Angiogenesis in Pancreatic Cancer
Current Pharmaceutical Design Microbial / Enzymatic Synthesis of Chiral Intermediates for Pharmaceuticals: Case Studies from BMS
Current Organic Chemistry Apoptosis: Potential Therapeutic Targets for New Drug Discovery
Current Medicinal Chemistry Human Induced Pluripotent Stem Cells for Inherited Cardiovascular Diseases Modeling
Current Stem Cell Research & Therapy Meet Our Editorial Board Member
Current Drug Safety Neprilysin: A Potential Therapeutic Target of Arterial Hypertension?
Current Cardiology Reviews Vascular Endothelial Growth Factor Inhibitor Therapy and Cardiovascular and Renal Damage in Renal Cell Carcinoma
Current Vascular Pharmacology The Long-Term Effect of Medically Enhancing Melanin Intrinsic Bioenergetics Capacity in Prematurity
Current Genomics L-Arginine Modulates Glucose and Lipid Metabolism in Obesity and Diabetes
Current Protein & Peptide Science Modulation of Cell Death in Age-Related Diseases
Current Pharmaceutical Design Diagnostic Cardiac Catheterization in the Pediatric Population
Current Cardiology Reviews